(Ph. Eur. 11.6 update)
(Ph. Eur. monograph 2132)
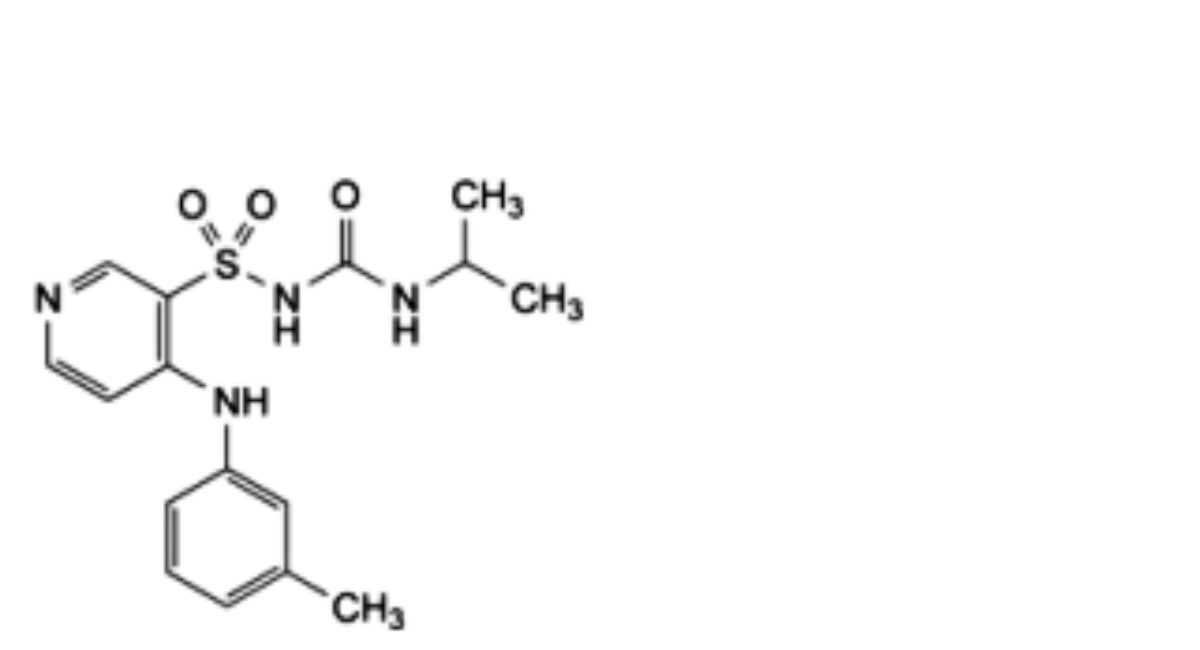
C16H20N4O3S 348.4 56211-40-6
Action and use
Thiazide-like diuretic.
DEFINITION
1-(1-Methylethyl)-3-[[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]urea.
Content
99.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white powder.
Solubility
Practically insoluble in water, slightly soluble in ethanol (96 per cent). It is sparingly soluble in dilute solutions of alkali hydroxides and slightly soluble in dilute acids.
It shows polymorphism (5.9).
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Comparison anhydrous torasemide CRS.
If the spectra obtained in the solid state show differences, dissolve the substance to be examined and the reference substance separately in methanol R, evaporate to dryness and record new spectra using the residues.
TESTS
Related substances
Liquid chromatography (2.2.29).
Solution A Dissolve 2.7 g of potassium dihydrogen phosphate R in 950 mL of water R, adjust to pH 3.5 with phosphoric acid R and dilute to 1000 mL with water R.
Test solution. Dissolve 20.0 mg of the substance to be examined in 15 mL of methanol R and sonicate for 15 min. Add 22.5 mL of solution A, cool to room temperature and dilute to 50.0 mL with the mobile phase.
Reference solution (a). Dissolve 2.0 mg of torasemide for system suitability CRS (containing impurities A, B, C and D) in 2.5 mL of methanol R and dilute to 5.0 mL with solution A.
Reference solution (b). Dilute 1.0 mL of the test solution to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (c) Dissolve the contents of a vial of torasemide impurity E CRS in 0.5 mL of methanol R. Add 0.5 mL of solution A.
Column
— size: l = 0.125 m, Ø = 4.0 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R (5 μm);
— temperature: 30 °C.
Mobile phase
Methanol R, solution A (40:60 V/V).
Flow rate. 0.8 mL/min.
Detection. Spectrophotometer at 288 nm.
Injection. 20 μL.
Run time. 2.5 times the retention time of torasemide.
Identification of impurities Use the chromatogram supplied with torasemide for system suitability CRS and the chromatogram obtained with reference solution (a) to identify the peaks due to impurities A, B, C and D; use the chromatogram obtained with reference solution (c) to identify the peak due to impurity E.
Relative retention. With reference to torasemide (retention time = about 10 min): impurity A = about 0.3; impurity B = about 0.4; impurity C = about 0.5; impurity E = about 0.7; impurity D = about 2.3.
System suitability
— resolution: minimum 3.0 between the peaks due to impurities B and C in the chromatogram obtained with reference solution (a);
— signal-to-noise ratio: minimum 100 for the principal peak in the chromatogram obtained with reference solution (b).
Limits
— correction factors: for the calculation of content, multiply the peak areas of the following impurities by the corresponding correction factor: impurity A = 5.1; impurity B = 0.76;
— impurity B: not more than 5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.5 per cent);
— impurities A, C, D, E: for each impurity, not more than 1.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.15 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.10 per cent);
— total: not more than 6 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.6 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C for 3 h.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.300 g in 50 mL of anhydrous acetic acid R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 34.84 mg of. C16H20N4O3S
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D, E.
![A. 4-(3-methylphenyl)-2H-pyrido[4,3-e]-1,2,4-thiadiazin-3(4H)-one 1,1-dioxide,](https://nhathuocngocanh.com/bp/wp-content/uploads/2025/11/A.-4-3-methylphenyl-2H-pyrido43-e-124-thiadiazin-34H-one-11-dioxide.jpg)
A. 4-(3-methylphenyl)-2H-pyrido[4,3-e]-1,2,4-thiadiazin-3(4H)-one 1,1-dioxide,
![B. 4-[(3-methylphenyl)amino]pyridine-3-sulfonamide,](https://nhathuocngocanh.com/bp/wp-content/uploads/2025/11/B.-4-3-methylphenylaminopyridine-3-sulfonamide.jpg)
B. 4-[(3-methylphenyl)amino]pyridine-3-sulfonamide,
![C. 1-ethyl-3-[[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]urea,](https://nhathuocngocanh.com/bp/wp-content/uploads/2025/11/C.-1-ethyl-3-4-3-methylphenylaminopyridin-3-ylsulfonylurea.jpg)
C. 1-ethyl-3-[[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]urea,
![D. 1-butyl-3-[[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]urea,](https://nhathuocngocanh.com/bp/wp-content/uploads/2025/11/D.-1-butyl-3-4-3-methylphenylaminopyridin-3-ylsulfonylurea.jpg)
D. 1-butyl-3-[[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]urea,
![E. ethyl [[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]carbamate](https://nhathuocngocanh.com/bp/wp-content/uploads/2025/11/E.-ethyl-4-3-methylphenylaminopyridin-3-ylsulfonylcarbamate.jpg)
E. ethyl [[4-[(3-methylphenyl)amino]pyridin-3-yl]sulfonyl]carbamate.