Edition: BP 2025 (Ph. Eur. 11.6 update)
(Ph. Eur. monograph 1050)
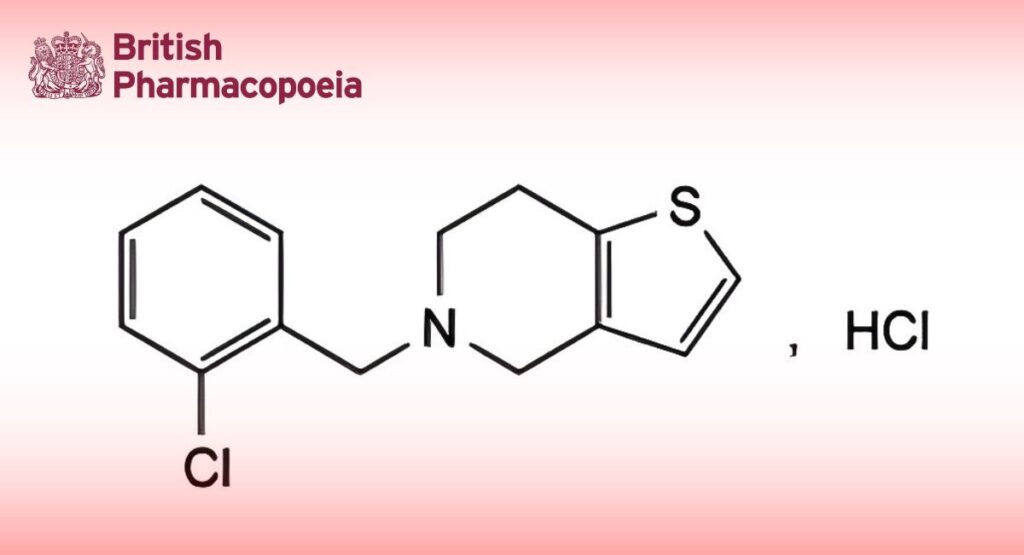
C14H15Cl2NS 300.2 53885-35-1
Action and use
Inhibition of adenosine diphosphate (ADP)-mediated platelet aggregation; antiplatelet drug.
Ph Eur
DEFINITION
5-(2-Chlorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine hydrochloride.
Content
99.0 per cent to 101.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Sparingly soluble in water and in anhydrous ethanol, very slightly soluble in ethyl acetate.
IDENTIFICATION
First identification: B, D.
Second identification: A, C, D.
A. Ultraviolet and visible absorption spectrophotometry (2.2.25).
Test solution (a) Dissolve 40 mg in water R and dilute to 100.0 mL with the same solvent.
Test solution (b) Dilute 5.0 mL of test solution (a) to 100.0 mL with water R.
Spectral range 250-350 nm for test solution (a); 200-350 nm for test solution (b).
Absorption maxima At 268 nm and 275 nm for test solution (a); at 214 nm and 232 nm for test solution (b).
Absorption ratio A268/A275 = 1.1 to 1.2.
B. Infrared absorption spectrophotometry (2.2.24).
Comparison ticlopidine hydrochloride CRS.
C. Mix about 6 mg of citric acid monohydrate R and 0.3 mL of acetic anhydride R. Add about 5 mg of the substance to be examined and heat in a water-bath at 80 °C. A red colour develops.
D. About 20 mg gives reaction (a) of chlorides (2.3.1).
TESTS
Appearance of solution
The solution is clear (2.2.1) and colourless (2.2.2, Method II).
Dissolve 0.5 g in a 1 per cent V/V solution of hydrochloric acid R and dilute to 20 mL with the same solution.
pH (2.2.3)
3.5 to 4.0.
Dissolve 0.5 g in carbon dioxide-free water R and dilute to 20 mL with the same solvent.
Related substances
Liquid chromatography (2.2.29).
Solvent mixture Mobile phase B, mobile phase A (20:80 V/V).
Test solution Dissolve 0.250 g of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution Dissolve 5.0 mg of ticlopidine impurity F CRS in the solvent mixture. Add 1.00 mL of the test solution and dilute to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Column:
— size: l = 0.15 m, Ø = 4.6 mm;
— stationary phase: octadecylsilyl silica gel for chromatography R (5 μm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: 0.95 g/L solution of sodium pentanesulfonate monohydrate R, adjusted to pH 3.4 with a 50 per cent V/V solution of phosphoric acid R;
— mobile phase B: methanol R1;
| Time (min) | Mobile phase A (per cent V/V) | Mobile phase B (per cent V/V) |
| 0 – 45 | 80 → 20 | 20 → 80 |
| 45 – 50 | 20 | 80 |
Flow rate 1.3 mL/min.
Detection Spectrophotometer at 220 nm.
Injection 10 μL; inject the solvent mixture as a blank.
Identification of impurities Use the chromatogram obtained with the reference solution to identify the peak due to impurity F.
Retention time Ticlopidine = about 15 min.
System suitability Reference solution:
— resolution: minimum 2.0 between the peaks due to ticlopidine and impurity F; if necessary, adjust the pH of mobile phase A;
— signal-to-noise ratio: minimum 50 for the peak due to ticlopidine.
Limits:
— impurity F: not more than 0.5 times the area of the corresponding peak in the chromatogram obtained with the reference solution (0.05 per cent);
— unspecified impurities: for each impurity, not more than the area of the peak due to ticlopidine in the chromatogram obtained with the reference solution (0.10 per cent);
— total: not more than the area of the peak due to ticlopidine in the chromatogram obtained with the reference solution (0.10 per cent);
— disregard limit: 0.5 times the area of the peak due to ticlopidine in the chromatogram obtained with the reference solution (0.05 per cent). Do not disregard the peak due to impurity F.
Formaldehyde
Maximum 20 ppm.
Dissolve 0.200 g in 4.0 mL of water R. Add 0.4 mL of dilute sodium hydroxide solution R. Centrifuge, filter the supernatant through cotton previously impregnated with water R and dilute to 5.0 mL with water R. Transfer to a test-tube. Add 5.0 mL of acetylacetone reagent R1. Place the test-tube in a water-bath at 40 °C for 40 min. The test solution is not more intensely coloured than a standard prepared at the same time and in the same manner using 5.0 mL of a 0.8 ppm solution of formaldehyde (CH2O), obtained by dilution of formaldehyde standard solution (5 ppm CH2O) R with water R. Examine the tubes down their vertical axis.
Water (2.5.12)
Maximum 0.5 per cent, determined on 0.500 g.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.150 g in 15 mL of anhydrous acetic acid R. Add 35 mL of acetic anhydride R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 30.02 mg of C14H15Cl2NS .
IMPURITIES
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities
for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, B, C, D, E, G, H, I, J, K, L.
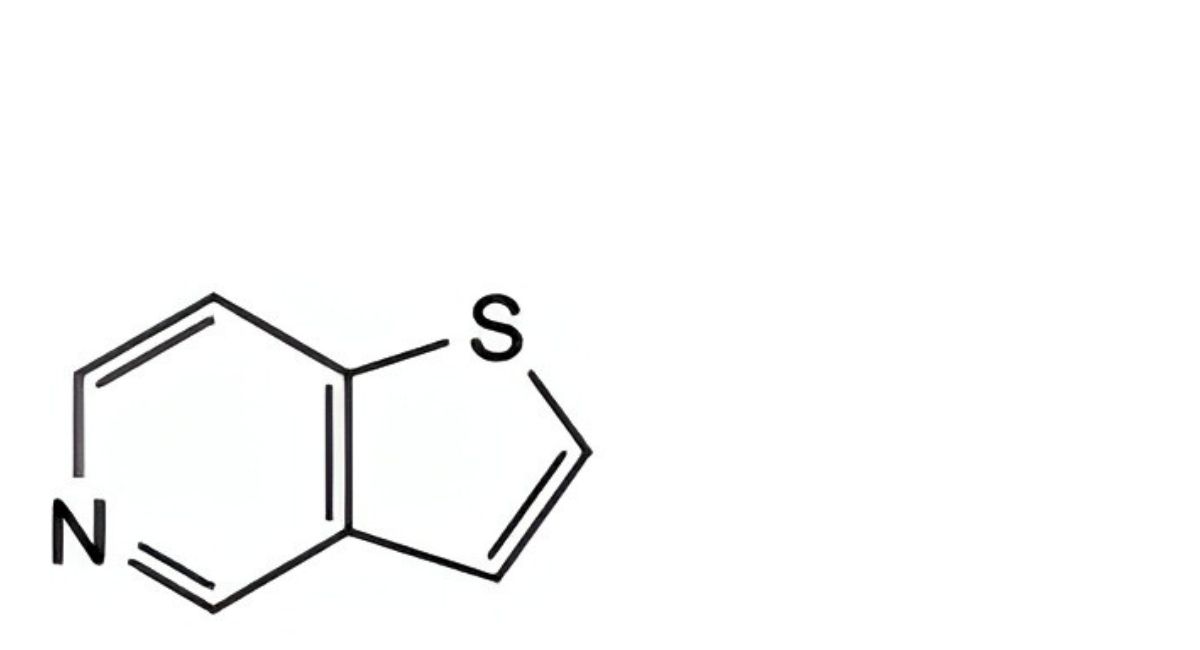
A. thieno[3,2-c]pyridine,
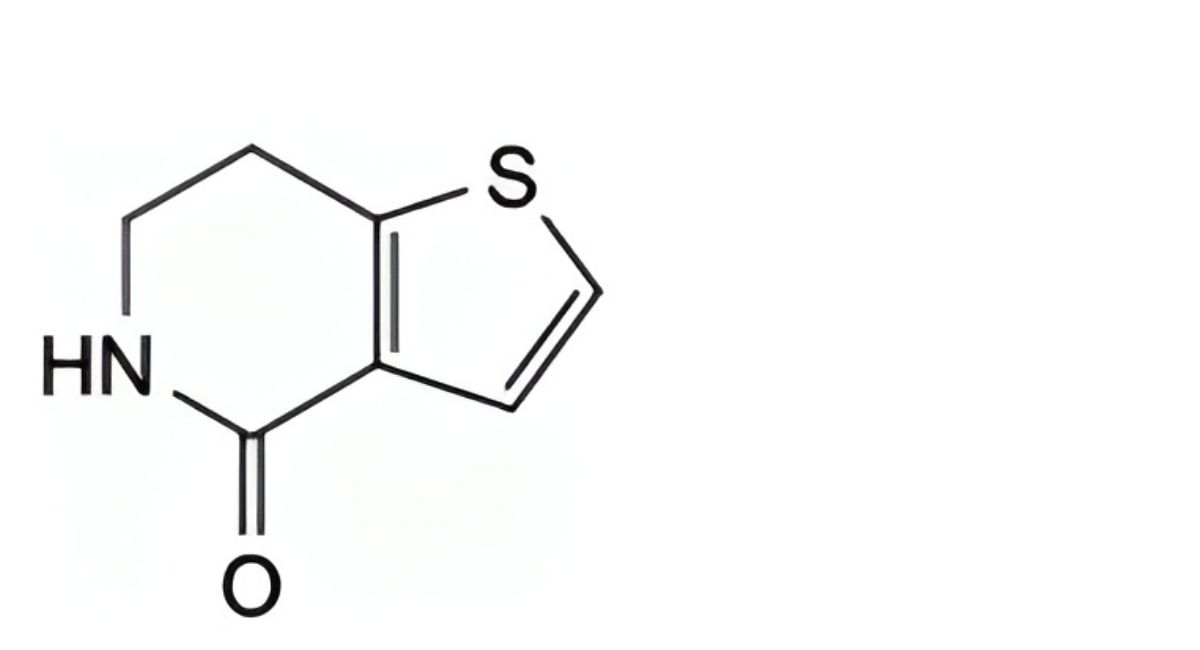
B. 6,7-dihydrothieno[3,2-c]pyridin-4(5H)-one,
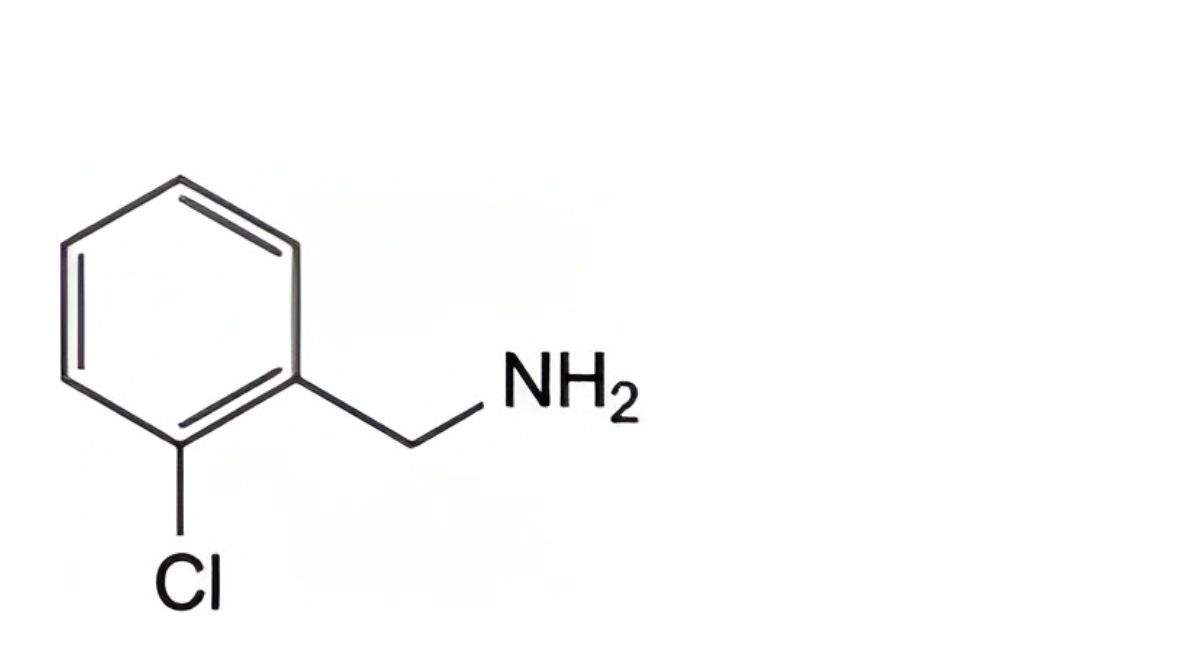
C. (2-chlorophenyl)methanamine,
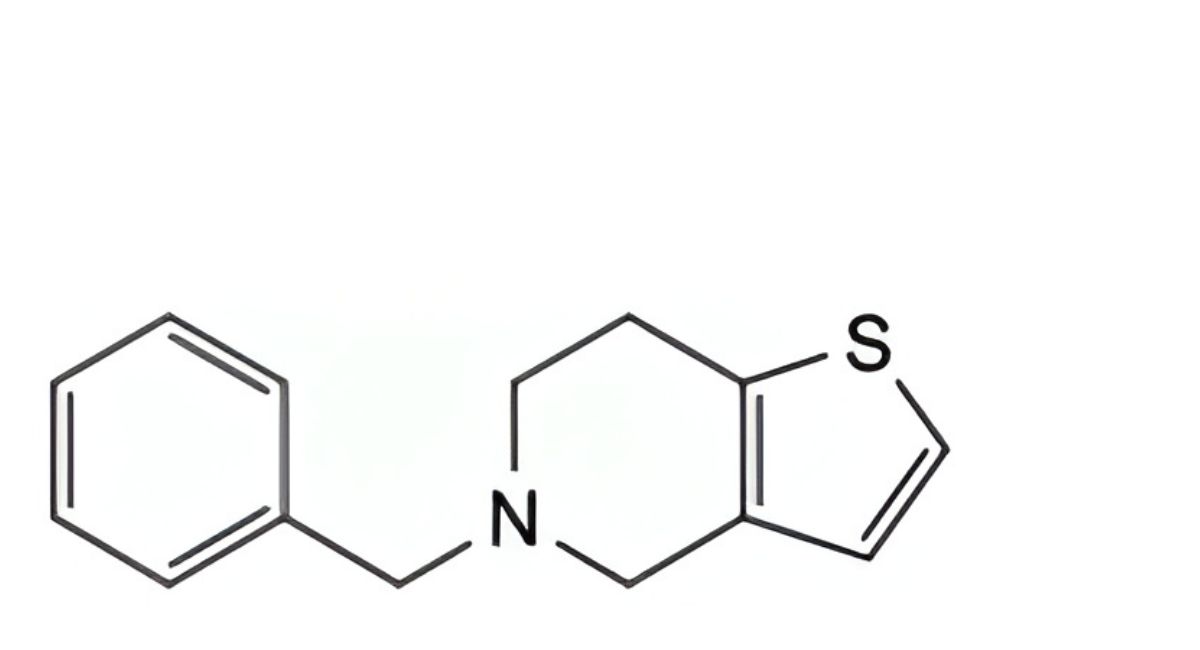
D. 5-benzyl-4,5,6,7-tetrahydrothieno[3,2-c]pyridine,
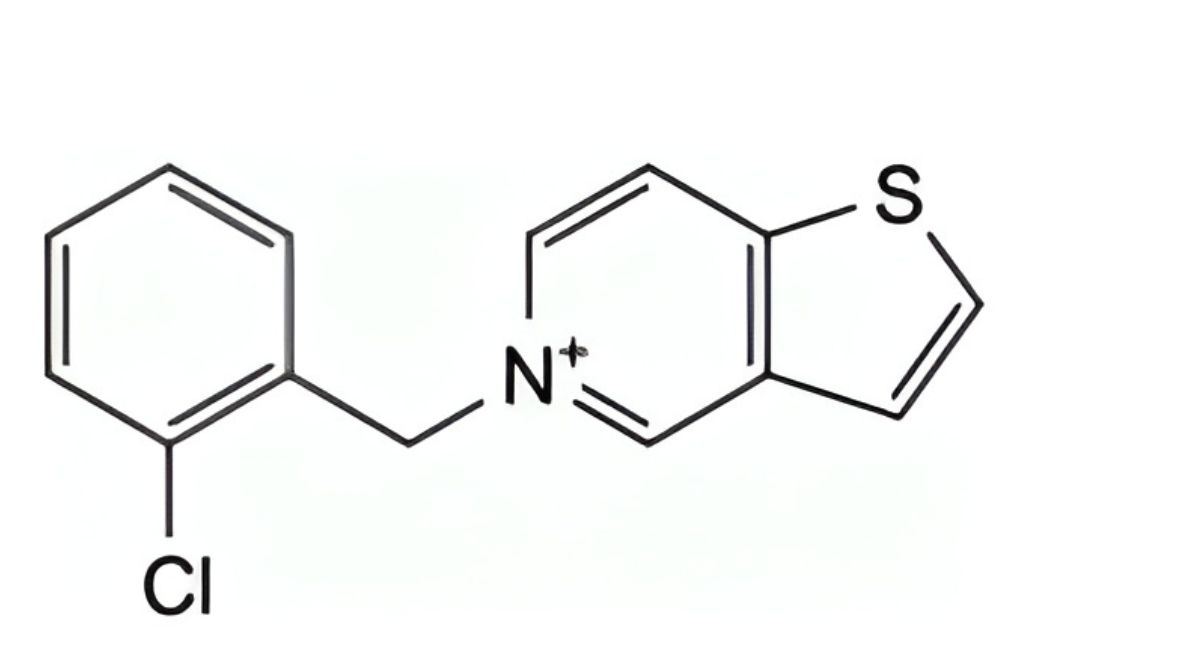
E. 5-(2-chlorobenzyl)thieno[3,2-c]pyridinium,
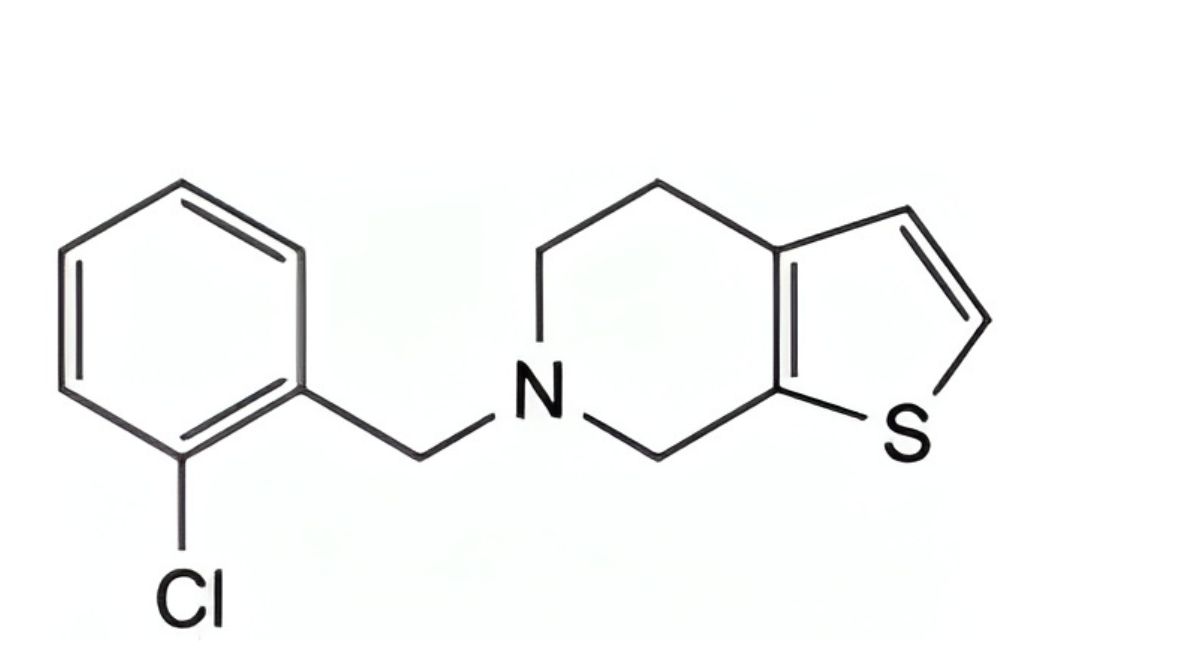
F. 6-(2-chlorobenzyl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine,
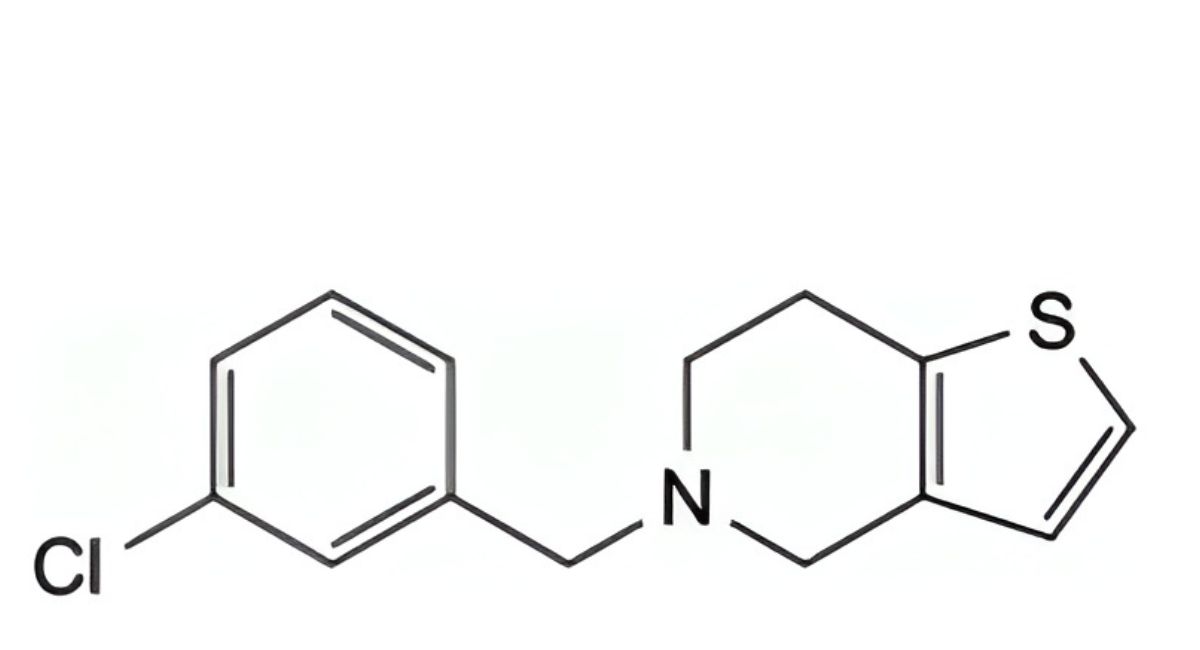
G. 5-(3-chlorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine,
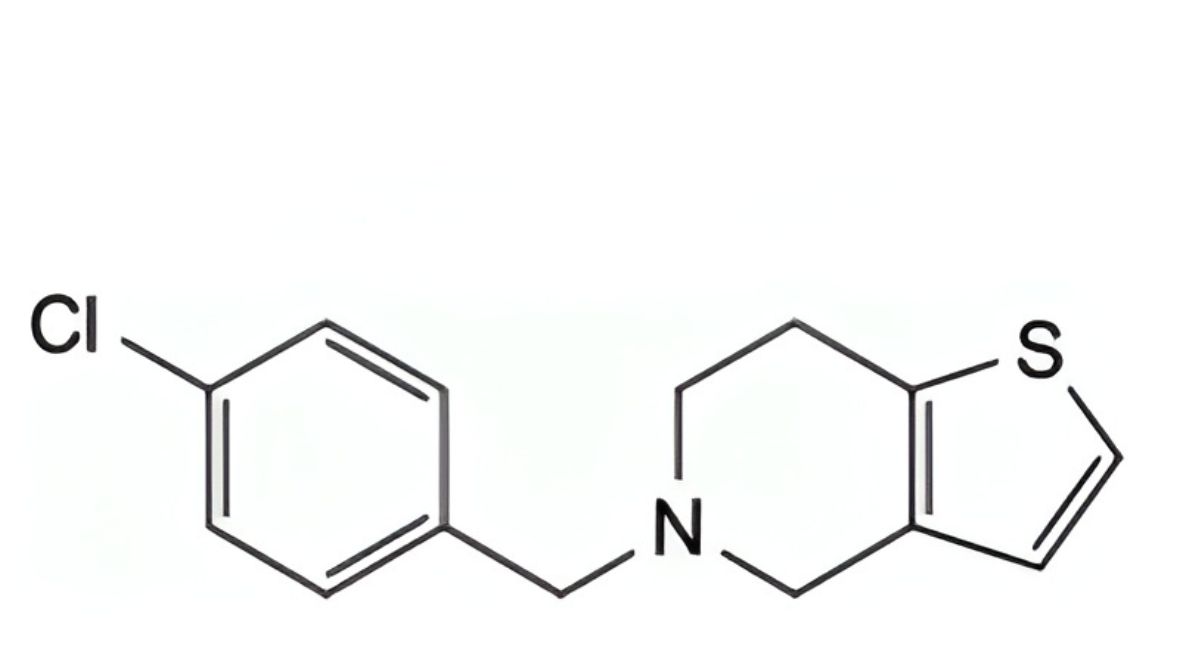
H. 5-(4-chlorobenzyl)-4,5,6,7-tetrahydrothieno[3,2-c]pyridine,
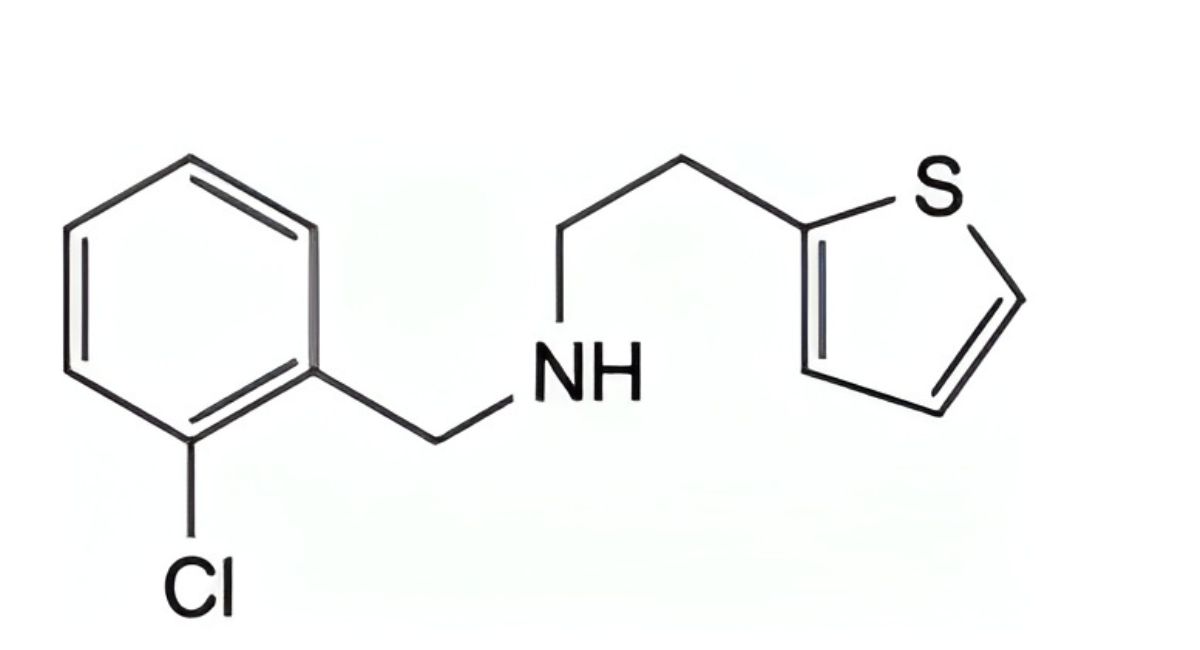
I. N-(2-chlorobenzyl)-2-(thiophen-2-yl)ethanamine,
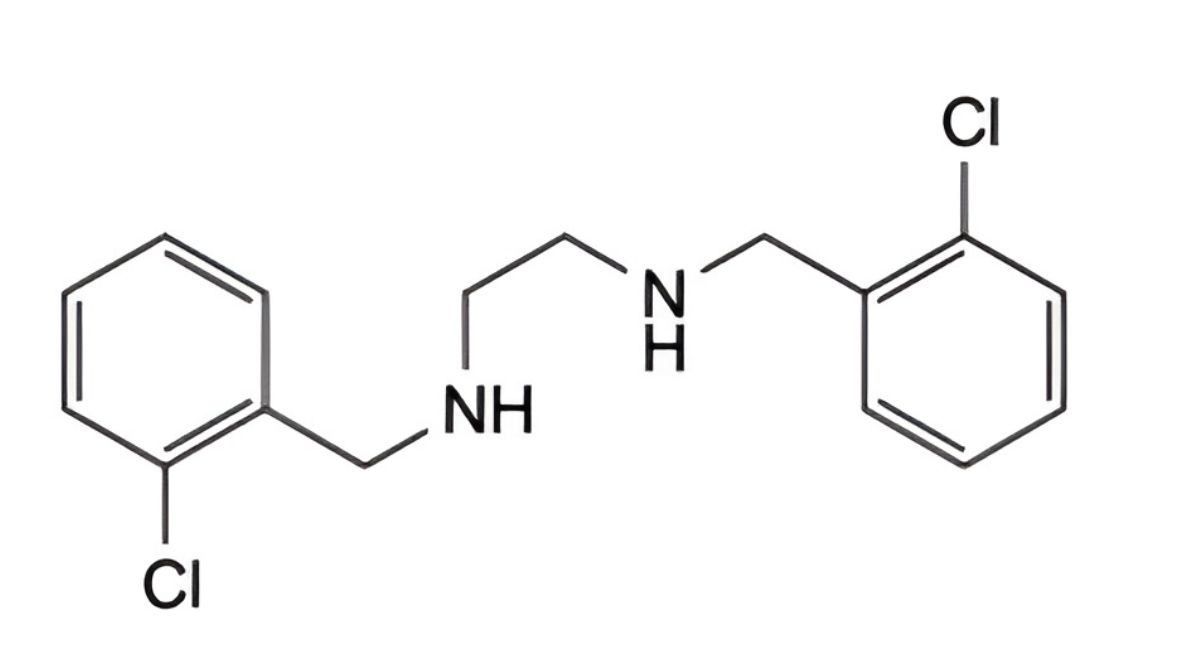
J. N,N′-bis(2-chlorobenzyl)ethane-1,2-diamine,
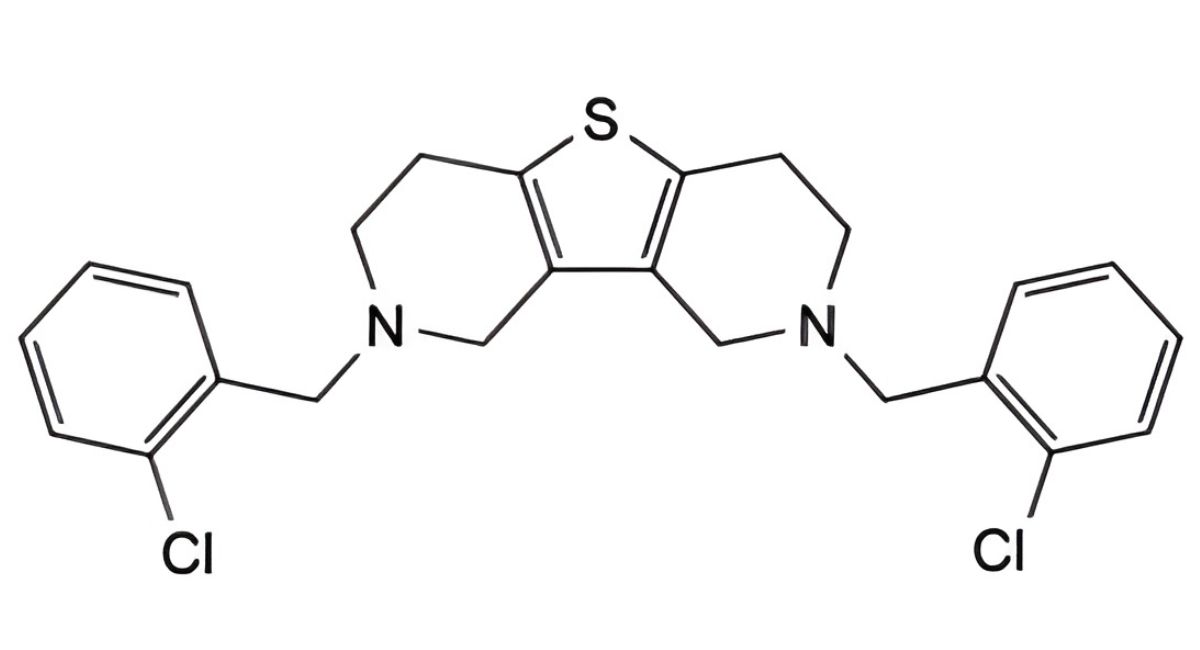
K. 2,8-bis(2-chlorobenzyl)-1,2,3,4,6,7,8,9-octahydrothieno[3,2-c:4,5-c′]dipyridine (bis-ticlopidine),
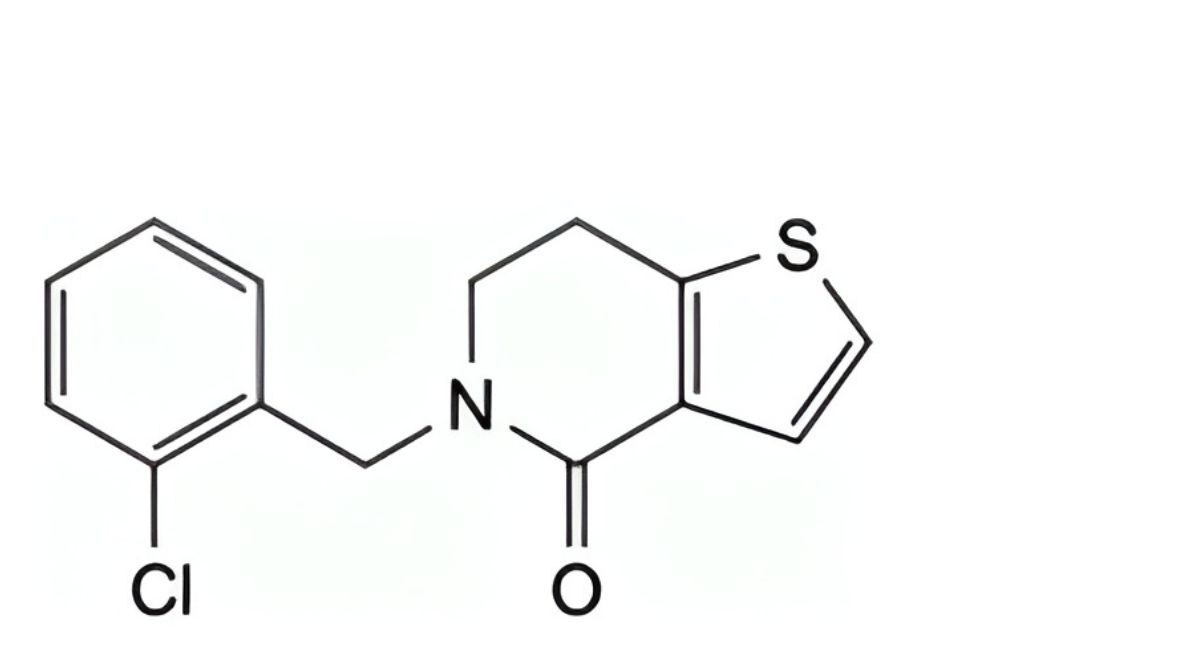
L. 5-(2-chlorobenzyl)-6,7-dihydrothieno[3,2-c]pyridin-4(5H)-one.
Ph Eur