Edition: BP 2025 (Ph. Eur. 11.6 update)
General Notices
(Ph. Eur monograph 3087)
C23H28F2N6O4S 522.6 274693-27-5
Action and use
Platelet aggregation inhibitor.
DEFINITION
(1 S,2S,3R,5S)-3-[7-[[(1 R,2S)-2-(3,4-Difluorophenyl)cyclopropyl]amino]-5-(propylsulfanyl)-3R-[1,2,3]triazolo[4,5-d]pyrimidin- 3-yl]-5-(2-hydroxyethoxy)cyclopentane-1,2-diol.
Content
97.5 per cent to 102.0 per cent (anhydrous substance).
PRODUCTION
It is produced by highly stereoselective methods of manufacture; consideration must be given to the formation of potential stereoisomeric impurities during the manufacturing process, and procedures must be implemented for the appropriate control of these impurities.
CHARACTERS
Appearance
White or almost white to pale pink powder.
Solubility
Practically insoluble in water, freely soluble in methanol, soluble in anhydrous ethanol, practically insoluble in heptane.
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Comparison ticagrelor CRS.
TESTS
Impurity E
Liquid chromatography (2.2.29).
Buffer solution 156 g/L solution of sodium dihydrogen phosphate R adjusted to pH 3.0 with phosphoric acid R.
Test solution Dissolve 30.0 mg of the substance to be examined in 2 mL of a mixture of equal volumes of acetonitrile R and water R and dilute to 10.0 mL with water R. Centrifuge for 15 min at about 1200 g or until a clear solution is obtained. Use the clear supernatant.
Reference solution Dissolve 4.5 mg of ticagrelor impurity E CRS ((R)-mandelate salt) in acetonitrile R and dilute to 100.0 mL with the same solvent. Dilute 1.0 mL of the solution to 100.0 mL with acetonitrile R. Dilute 1.0 mL of this solution to 10.0 mL with water R (solution equivalent to a 24 ng/mL solution of impurity E).
Column:
— size: l = 0.15 m, 0 = 4.6 mm;
— stationary phase: octadecylsilyl silica gel for chromatography R (1.8 pm);
— temperature: 55 °C.
Mobile phase:
— mobile phase A: mix 1 volume of the buffer solution and 89 volumes of water for chromatography R and add 10 volumes of acetonitrile for chromatography R;
— mobile phase B: mix 1 volume of the buffer solution and 29 volumes of water for chromatography R and add
70 volumes of acetonitrile for chromatography R;
| Time (min) | Mobile phase A (per cent V/V) | Mobile phase B (per cent V/V) |
| 0 – 8 | 94 | 6 |
| 8 – 9 | 94 -> 0 | 6 -> 100 |
| 9 – 20 | 0 | 100 |
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 222 nm.
Injection: 100 pL.
Relative retention With reference to ticagrelor (retention time = about 11.5 min): impurity E = about 0.6.
System suitability Reference solution:
— repeatability: maximum relative standard deviation of 10.0 per cent determined on 6 injections.
Limit:
— impurity E: not more than the area of the principal peak in the chromatogram obtained with the reference solution (8 ppm).
Related substances
Liquid chromatography (2.2.29). Carry out the test protected from light.
Solvent mixture acetonitrile R, water R (35:65 V/V).
Buffer solution 156 g/L solution of sodium dihydrogen phosphate R adjusted to pH 3.0 with phosphoric acid R.
Test solution Dissolve 50.0 mg of the substance to be examined in the solvent mixture and dilute to 100.0 mL with the solvent mixture.
Reference solution (a) Dissolve 50.0 mg of ticagrelor CRS in the solvent mixture and dilute to 100.0 mL with the solvent mixture.
Reference solution (b) Dilute 1.0 mL of the test solution to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Reference solution (c) Dissolve 5 mg of ticagrelor for system suitability CRS (containing impurities A, B, C and D) in the solvent mixture and dilute to 10 mL with the solvent mixture.
Column:
— size: l = 0.15 m, 0 = 3.0 mm;
— stationary phase: base-deactivated end-capped phenylsilyl silica gel for chromatography R (3 pm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: mix 1 volume of the buffer solution and 89 volumes of water for chromatography R and add 10 volumes of acetonitrile for chromatography R;
— mobile phase B: mix 1 volume of the buffer solution and 29 volumes of water for chromatography R and add 70 volumes of acetonitrile for chromatography R;
| Time (min) | Mobile phase A (per cent V/V) | Mobile phase B (per cent V/V) |
| 0 – 2 | 80 | 20 |
| 2 – 42 | 80 -> 25 | 20-> 75 |
| 42 – 47 | 25 | 75 |
Flow rate 0.65 mL/min.
Detection Spectrophotometer at 242 nm.
Injection 5 pL of the test solution and reference solutions (b) and (c).
Identification of impurities Use the chromatogram supplied with ticagrelor for system suitability CRS and the chromatogram obtained with reference solution (c) to identify impurities A, B, C and D.
Relative retention With reference to ticagrelor (retention time = about 23 min): impurity A = about 0.15; impurity B = about 1.06; impurity C = about 1.23; impurity D = about 1.5.
System suitability Reference solution (c):
— resolution: minimum 4.0 between the peaks due to ticagrelor and impurity B.
Calculation of percentage contents:
— correction factor: multiply the peak area of impurity A by 0.5;
— for each impurity, use the concentration of ticagrelor in reference solution (b).
Limits:
— impurity D: maximum 0.3 percent;
— impurities A, B: for each impurity, maximum 0.2 per cent;
— impurity C: maximum 0.1 per cent;
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 1.0 per cent;
— reporting threshold: 0.05 per cent.
Water (2.5.32)
Maximum 0.5 per cent determined on 0.500 g using the evaporation technique at 130 °C.
Sulfated ash (2.4.14)
Maximum 0.6 per cent, determined on 1.0 g in a platinum crucible.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection Test solution and reference solutions (a) and (c).
Mobile phase Mix 1 volume of the buffer solution and 56 volumes of water for chromatography R and add 43 volumes of acetonitrile for chromatography R.
Run time 4.5 times the retention time of ticagrelor.
Relative retention With reference to ticagrelor (retention time = about 6 min): impurity B = about 1.15.
System suitability Reference solution (c):
— resolution: minimum 1.5 between the peaks due to ticagrelor and impurity B.
Calculate the percentage content of C23H28F2N6O4S using the chromatogram obtained with reference solution (a) and taking into account the assigned content of ticagrelor CRS.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D, E.
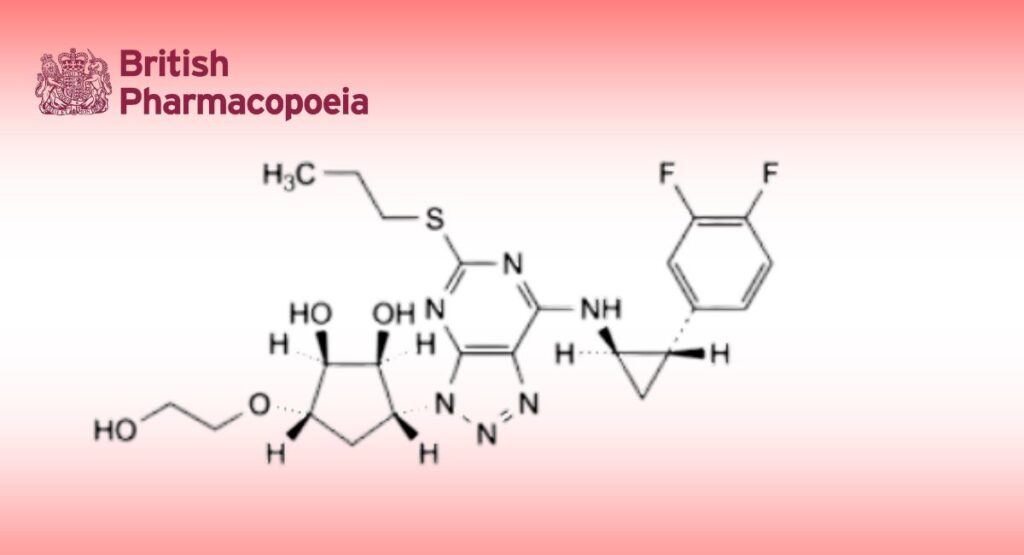
A. (1 S,2S,3R,5S)-3-[7-amino-5-(propylsulfanyl)-3H-[1,2,3]triazolo[4,5-d]pyrimidin-3-yl]-5-(2-hydroxyethoxy)cyclopentane-1,2-diol,
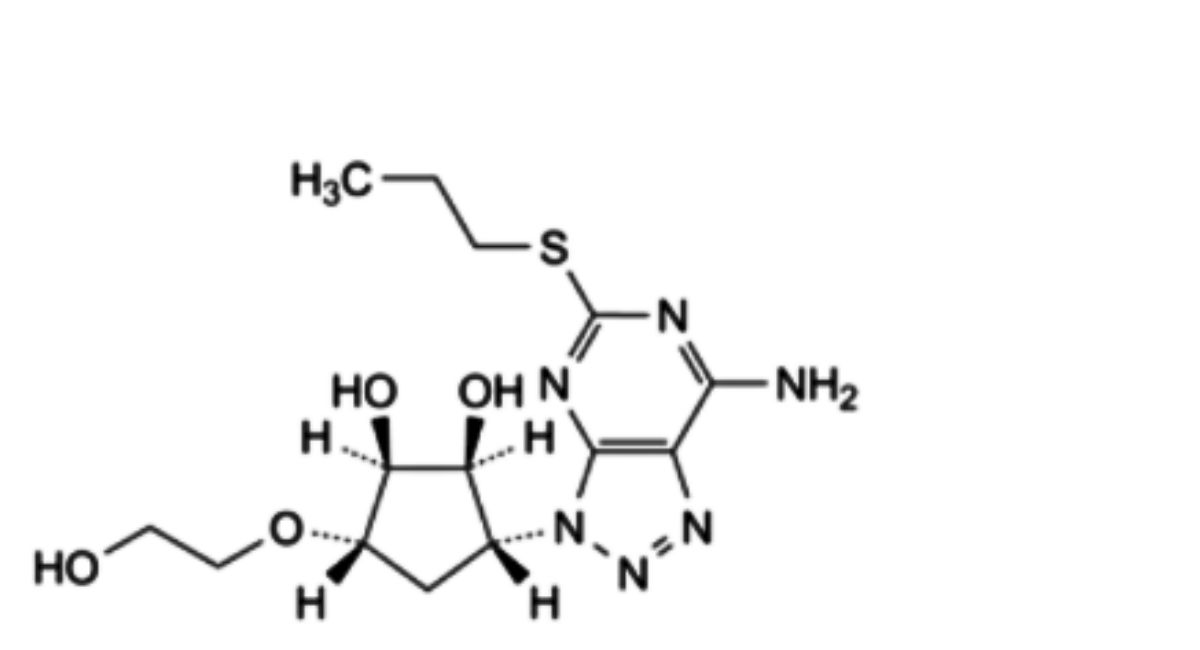
B. (1 S,2S,3R,5S)-3-[[3-[(1 R,2S)-2-(3,4-difluorophenyl)cyclopropyl]-5-(propylsulfanyl)-3R-[1,2,3]triazolo[4,5-d]pyrimidin-7- yl]amino]-5-(2-hydroxyethoxy)cyclopentane-1,2-diol,
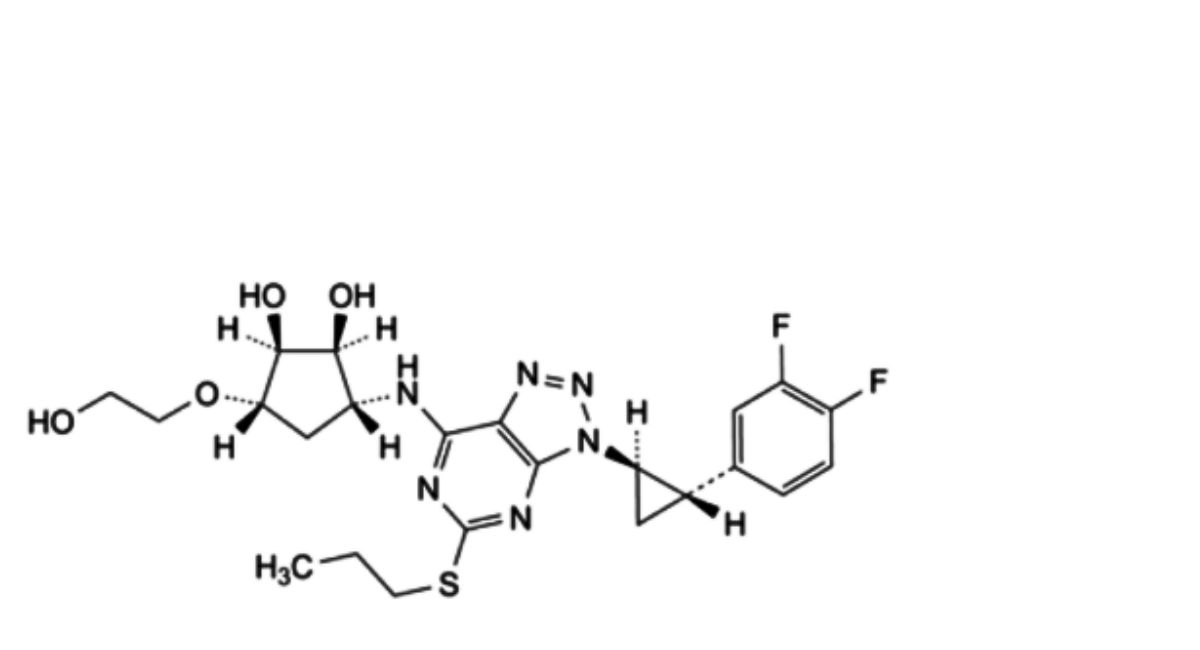
C. 2-[[(1 S,2S,3S,4R)-4-[7-[[(1 R,2S)-2-(3,4-difluorophenyl)cyclopropyl]amino]-5-(propylsulfanyl)-3R-[1,2,3]triazolo[4,5- d]pyrimidin-3-yl]-2,3-dihydroxycyclopentyl]oxy]ethyl acetate,
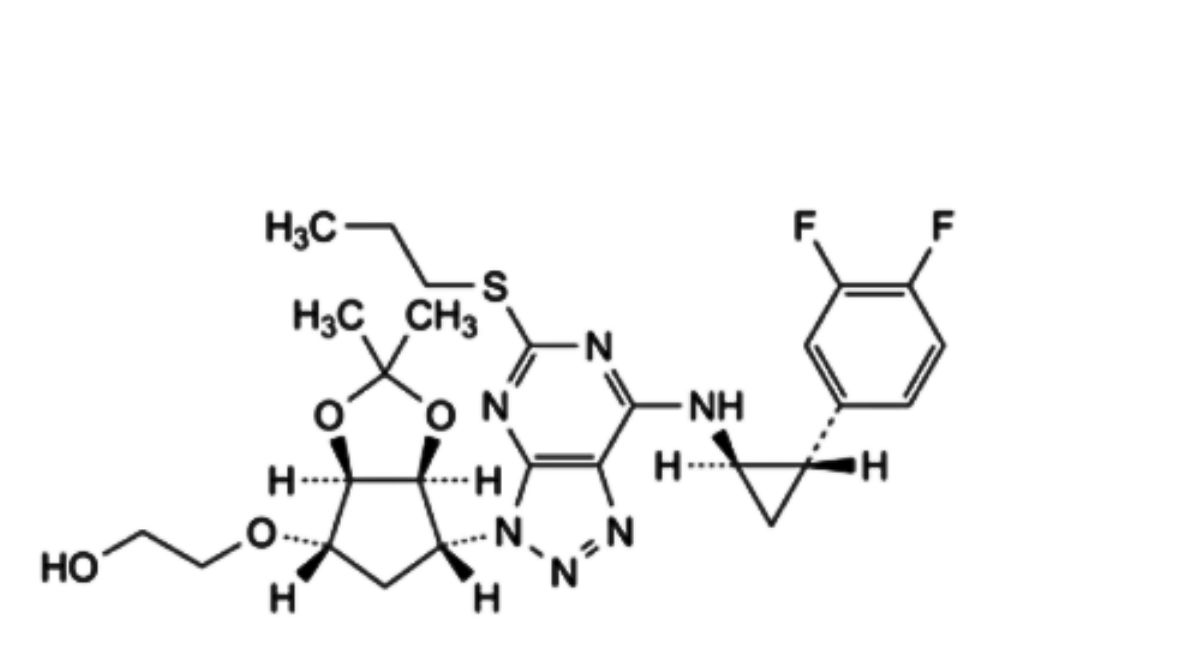
D. 2-[[(3aR,4S,6R,6aS)-6-[7-[[(1R,2S)-2-(3,4-difluorophenyl)cyclopropyl]amino]-5-(propylsulfanyl)-3R-[1,2,3]triazolo[4,5-d]pyrimidin-3-yl]-2,2-dimethyltetrahydro-2R,3aR-cyclopenta[d][1,3]dioxol-4-yl]oxy]ethan-1-ol,
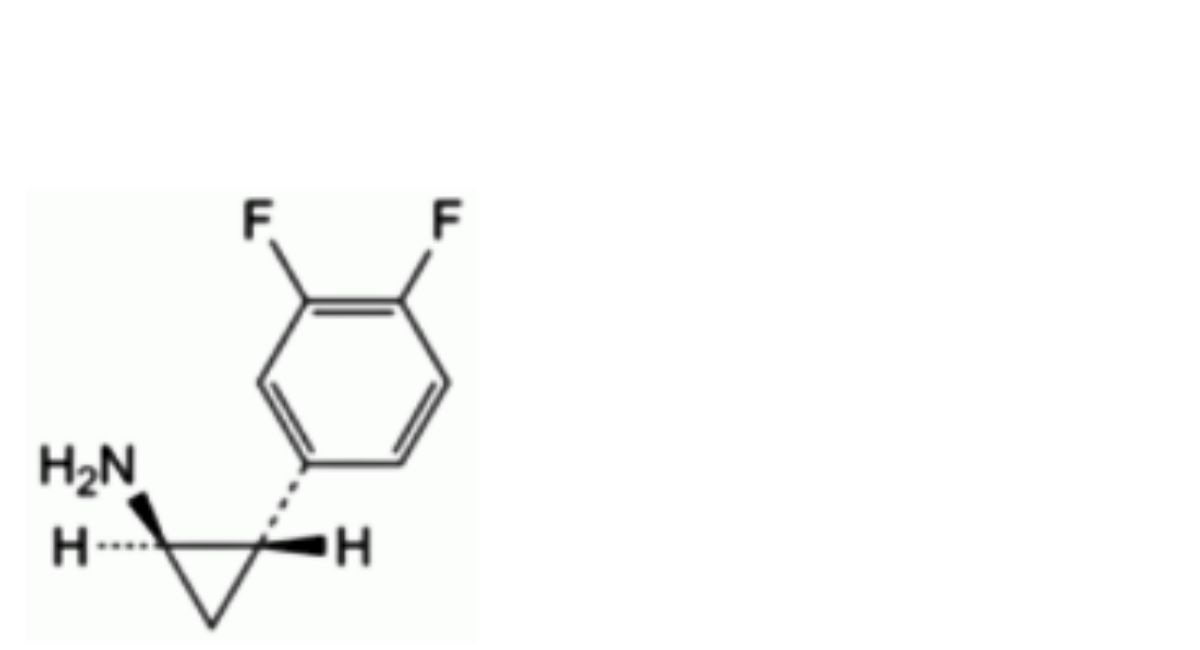
E. (1 R,2S)-2-(3,4-difluorophenyl)cyclopropan-1-amine.