(Ph. Eur. monograph 2804)
C42H70-nO35(C4H8SO3Na)n 2163 when n = 6.5 182410-00-0
DEFINITION
Sulfobutylbetadex sodium (β-cyclodextrin, sodium 4-sulfonatobutyl ether) is the sodium salt of a partially substituted poly(sulfobutyl) ether of betadex.
Content
— sulfobutylbetadex sodium: 95.0 per cent to 105.0 per cent (anhydrous substance);
— average number of sulfobutyl groups per cyclodextrin ring, expressed as average degree of substitution (DS): 5.9 to 6.6.
CHARACTERS
Appearance
White or almost white, hygroscopic powder.
Solubility
Freely soluble in water, practically insoluble in anhydrous ethanol and in methylene chloride.
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Comparison: sulfobutylbetadex sodium CRS.
Results: The spectrum obtained with the substance to be examined shows the same absorption bands as the spectrum obtained with sulfobutylbetadex sodium CRS. Due to differences in the degree of substitution of the substance, the relative intensity of some absorption bands may vary.
B. It gives reaction (a) of sodium (2.3.1).
TESTS
Solution S
Dissolve 7.50 g in carbon dioxide-free water R and dilute to 50.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and colourless (2.2.2, Method II).
pH (2.2.3)
5.0 to 7.5 for solution S.
Use intensive nitrogen purging to avoid carbon dioxide absorption during measurement.
Reducing sugars
Maximum 0.05 per cent.
Reagent solution: Dissolve 1.0 g of triphenyltetrazolium chloride R in 100 mL of aldehyde-free methanol R.
Test solution: Mix 0.45 g of the substance to be examined with 2.0 mL of dimethyl sulfoxide R in a test tube and add 0.5 mL of a 40 g/L solution of sodium hydroxide R and 7.5 mL of the reagent solution. Mix and store at room temperature.
Reference solution: Prepare at the same time and in the same manner as for the test solution, using 0.10 mL of a 2.25 g/L solution of glucose R instead of the substance to be examined.
After 1 h, filter if necessary and measure the absorbance (2.2.25) of the solutions at 482 nm. The absorbance of the test solution is not greater than that of the reference solution.
Impurities A, C and D
Liquid chromatography (2.2.29).
Solvent mixture acetonitrile R, water R (50:50 V/V).
Test solution: Dissolve 0.300 g of the substance to be examined in the solvent mixture and dilute to 5.0 mL with the solvent mixture.
Reference solution (a): Taking the assigned content of betadex CRS into account, dissolve a quantity of betadex CRS corresponding to 12.0 mg of betadex (impurity A) and 12.0 mg of sulfobutylbetadex impurity C CRS in the solvent mixture and dilute to 100.0 mL with the solvent mixture.
Reference solutions (b), (c), (d), (e): Dilute reference solution (a) with the solvent mixture to obtain 4 reference solutions containing respectively 0.09 mg/mL, 0.06 mg/mL (reference solution (c)), 0.03 mg/mL and 0.012 mg/mL of impurity A and impurity C.
Reference solution (f): Taking the assigned content of sulfobutylbetadex impurity D CRS into account, dissolve a quantity of sulfobutylbetadex impurity D CRS corresponding to 12.0 mg of impurity D in the solvent mixture and dilute to 100.0 mL with the solvent mixture.
Reference solutions (g), (h), (i), (j), (k): Dilute reference solution (f) with the solvent mixture to obtain 5 reference solutions containing respectively 0.06 mg/mL (reference solution (g)), 0.045 mg/mL, 0.03 mg/mL, 0.015 mg/mL and 0.006 mg/mL of impurity D.
Reference solution (l): Dissolve 0.300 g of sulfobutylbetadex sodium CRS in 1.0 mL of reference solution (f) and dilute to 5.0 mL with the solvent mixture.
Column:
— size: l = 0.15 m, Ø = 3.0 mm;
— stationary phase: solid core zwitterion-bonded silica gel for chromatography R (2.7 μm);
— temperature: 20 °C.
Mobile phase:
— mobile phase A: 5.0 g/L solution of ammonium formate R1 adjusted to pH 4.0 with formic acid R;
— mobile phase B: mobile phase A, acetonitrile for chromatography R (10:90 V/V);
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
Flow rate (mL/min) |
| 0 – 4.5 | 23 | 77 | 0.5 |
| 4.5 – 11 | 23 → 90 | 77 → 10 | 0.7 |
Detection: Evaporative light-scattering detector; the following settings have been found to be suitable; if the detector has different setting parameters, adjust the detector settings so as to comply with the system suitability criteria. The use of a 2- position/6-port valve is advisable for ‘heart-cutting’ sulfobutylbetadex peaks and the sodium peak, if needed, to protect the detector from the significant amount of injected substance.
— carrier gas: nitrogen R;
— flow rate: 1.5 L/min;
— evaporator temperature: 50 °C.
Equilibration: At initial isocratic conditions for 11 min.
Injection 5 μL of the test solution and reference solutions (a), (b), (c), (d), (e), (g), (h), (i), (j), (k) and (l).
Identification of impurities: Use the chromatogram obtained with reference solution (a) to identify the peaks due to impurities A and C; use the chromatogram obtained with reference solution (g) to identify the peak due to impurity D.
Relative retention: With reference to impurity A (retention time = about 6.0 min): impurity C = about 0.4; impurity D = about 0.5; sulfobutylbetadex elutes as several peaks after impurity A unless heart-cutting is applied.
Establish the calibration curves representing the logarithm of the concentration of impurities A and C in reference solutions (a), (b), (c), (d) and (e) and impurity D in reference solutions (g), (h), (i), (j) and (k) as the abscissa and the logarithm of the corresponding peak areas as ordinates.
System suitability:
— repeatability: maximum relative standard deviation of 3.0 per cent for the area of the peak due to impurity A determined on 5 injections of reference solution (c);
— number of theoretical plates: minimum 20000, calculated for the peak due to impurity A in the chromatogram obtained with reference solution (c);
— resolution: baseline separation between the peaks due to impurity D and sodium in the chromatogram obtained with reference solution (l);
— the coefficient of determination (R ) calculated for each calibration curve is not less than 0.950. 2
Calculate the percentage content of impurities A, C and D with reference to the dried substance using the calibration curves.
Limits:
— impurities A, C: for each impurity, maximum 0.10 per cent;
— impurity D: maximum 0.05 per cent;
— reporting threshold: 0.02 per cent; disregard any peaks other than those due to impurities A, C and D and sulfobutylbetadex.
Impurity B
Gas chromatography (2.2.28). Prepare the solutions immediately before use.
Internal standard solution: Dissolve 25.0 mg of diethyl sulfone R in 10.0 mL of dimethylformamide R and dilute to 1000.0 mL with water R. Dilute 1.0 mL of the solution to 100.0 mL with water R.
Test solutions (a), (b), (c) and (d): In 4 test tubes with screw caps, prepare 4 solutions as indicated in Table 2804.-1. Mix each test tube on a vortex mixer for 30 s and allow to stand for at least 5 min or until complete separation of the phases.
Decant the organic phase into a vial and seal.
Reference solution (a): Dissolve 50.0 mg of sulfobutylbetadex impurity B CRS in 5.0 mL of dimethylformamide R and dilute with water R to obtain a concentration of 2.0 μg/mL.
Reference solution (b): Dilute 1.0 mL of reference solution (a) to 2.0 mL with water R.
Reference solution (c): Dilute 1.0 mL of reference solution (a) to 4.0 mL with water R.
Blank solution: Prepare as described for test solution (d), but omitting the substance to be examined.
Table 2804.-1.
| Substance to be examined (g) | Internal standard solution (mL) | Solvent/solution (mL) | Methylene chloride R (mL) | Final content of sulfobutylbetadex impurity B CRS (μg) | |
| Blank solution | 0 | 4.0 | Water R, 1.0 | 1.0 | 0 |
| Test solution (a) | 1.000 | 4.0 | Reference solution (a), 1.0 | 1.0 | 2.0 |
| Test solution (b) | 1.000 | 4.0 | Reference solution (b), 1.0 | 1.0 | 1.0 |
| Test solution (c) | 1.000 | 4.0 | Reference solution (c), 1.0 | 1.0 | 0.5 |
| Test solution (d) | 1.000 | 4.0 | Water R, 1.0 | 1.0 | 0 |
Column:
— material: fused silica;
— size: l = 30 m, Ø = 0.32 mm;
— stationary phase: cyanopropyl(7)phenyl(7)methyl(86)polysiloxane R (film thickness 0.5 μm).
Carrier gas helium for chromatography R.
Flow rate: 2.4 mL/min.
Temperature:
| Time (min) |
Temperature (°C) |
|
| Column | 0 – 10 | 100 → 200 |
| 10 – 11.5 | 200 → 250 | |
| 11.5 – 16.5 | 250 | |
| Injection port | 250 | |
| Detector | 270 |
Detection: Flame ionisation.
Injection: 1 μL.
Relative retention: With reference to the internal standard (retention time = about 6.5 min): impurity B = about 1.4.
System suitability:
— repeatability: maximum relative standard deviation of 10.0 per cent for the corrected ratios (as described under Calculation of content) determined on 5 injections of test solution (b).
Calculation of content: Calculate the ratio of the area of the peak due to impurity B to the area of the peak due to the internal standard in the chromatograms obtained with test solutions (a), (b), (c) and (d) respectively.
Subtract from these values the ratio of the area of the peak due to impurity B to the area of the peak due to the internal standard in the chromatogram obtained with the blank solution (diethyl sulfone may contain trace amounts of impurity B). Plot the added quantity of impurity B (in micrograms) as the abscissa and the corrected ratios as the ordinates. Extrapolate the line joining the points on the graph until it meets the x-axis. The distance between this point and the origin corresponds to the content of impurity B in micrograms per gram (ppm) of the substance to be examined.
Limit:
— impurity B: maximum 0.5 ppm.
Chlorides (2.4.4)
Maximum 0.12 per cent (corresponding to 0.20 per cent expressed as sodium chloride).
Dissolve a quantity of the substance to be examined corresponding to 42 mg of anhydrous substance in 15.0 mL of water R.
Water (2.5.12)
Maximum 10.0 per cent, determined on 0.200 g.
Microbial contamination
If intended for use in the manufacture of parenteral preparations:
— TAMC: acceptance criterion 102 CFU/g (2.6.12).
If not intended for use in the manufacture of parenteral preparations:
— TAMC: acceptance criterion 103 CFU/g (2.6.12);
— TYMC: acceptance criterion 102 CFU/g (2.6.12);
— absence of Escherichia coli (2.6.13);
— absence of Salmonella (2.6.13).
ASSAY
Size-exclusion chromatography (2.2.30).
Test solution: Dissolve 0.100 g of the substance to be examined in the mobile phase and dilute to 10.0 mL with the mobile phase.
Reference solution: Dissolve 0.100 g of sulfobutylbetadex sodium CRS in the mobile phase and dilute to 10.0 mL with the mobile phase.
Column:
— size: l = 0.30 m, Ø = 7.8 mm;
— stationary phase: polymethacrylate gel R.
Mobile phase: Dissolve 10.11 g of potassium nitrate R in a mixture of 1 volume of acetonitrile for chromatography R and 4 volumes of water for chromatography R and dilute to 1000 mL with the same mixture of solvents.
Flow rate: 1.0 mL/min.
Detection: Differential refractometer maintained at 35 ± 2 °C.
Injection: 20 μL.
Between-run rinsing: Rinse the column with a mixture of 1 volume of acetonitrile for chromatography R and 9 volumes of water for chromatography R.
System suitability: Reference solution:
— repeatability: maximum relative standard deviation of 2.0 per cent for the area of the peak due to sulfobutylbetadex, determined on 5 injections.
Calculate the percentage content of sulfobutylbetadex sodium taking into account the assigned content of sulfobutylbetadex sodium CRS.
Average degree of substitution
Nuclear magnetic resonance spectrometry (2.2.33).
The average degree of substitution (DS) is calculated from the ratio between the signals of the inner-chain protons of the sulfobutyl ether groups (-O-CH2-CH2-CH2-CH2-SO3H) and the signals of the glycosidic protons attached to the C1 carbon of the anhydroglucose units.
Test solution: Introduce not less than 10.0 mg of the substance to be examined, previously dried for at least 1 day in a desiccator, into a 5 mm NMR tube. Add about 0.70 mL of deuterium oxide R1. Cap the tube and mix thoroughly.
Apparatus FT-NMR spectrometer operating at minimum 250 MHz and suitable for recording at 25 °C.
Acquisition of H NMR spectra The following parameters may be used:
— sweep width: 10 ppm (0 to + 10.0 ppm);
— irradiation frequency offset: none;
— time domain: 32 K;
— pulse width: 90°;
— pulse delay: 15 s;
— dummy scans: 2;
— number of scans: 16.
Call the integration sub-routine after phase corrections and baseline correction between -1 ppm and + 9 ppm.
The signal of exchangeable protons (solvent) at + 4.8 ppm is used as reference.
Make a zero filling at least twice in size relative to the acquisition data file and transform the FID to the spectrum without any correction of the Gaussian broadening factor (GB = 0) and with a line broadening factor not greater than 0.3 Hz (LB ≤ 0.3). Adjust integral level and tilting.
Measure the peak areas of the signals between 1.43 ppm and 2.12 ppm (A1, -O-CH2-CH2-CH2-CH2-SO3H), and the signals of the glycosidic protons between 4.95 ppm and 5.41 ppm (A2, -O-CH-O-).
Calculate the average degree of substitution (DS) using the following expression:
(7 x A1)/ (4 x A2)
A1 = area of the peaks due to the inner methylene protons (indicated in bold) of the sulfobutyl ether functional group;
A2 = area of the peaks due to the glycosidic protons (indicated in bold).
STORAGE
In an airtight container.
LABELLING
The label states:
— the average degree of substitution (DS);
— where applicable, that the substance is suitable for use in the manufacture of parenteral preparations.
IMPURITIES
Specified impurities A, B, C, D.
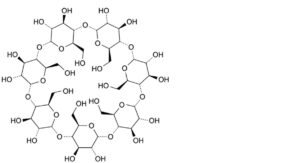
A. cycloheptakis-(1→4)-(α-D-glucopyranosyl) (betadex or cyclomaltoheptaose or β-cyclodextrin)

B. 1,2λ -oxathiane-2,2-dione,
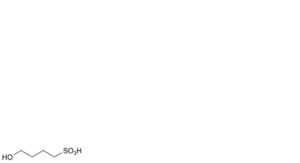
C. 4-hydroxybutane-1-sulfonic acid,
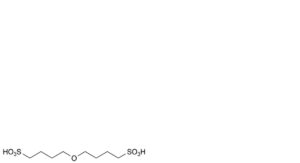
D. 4,4′-oxydi(butane-1-sulfonic acid).