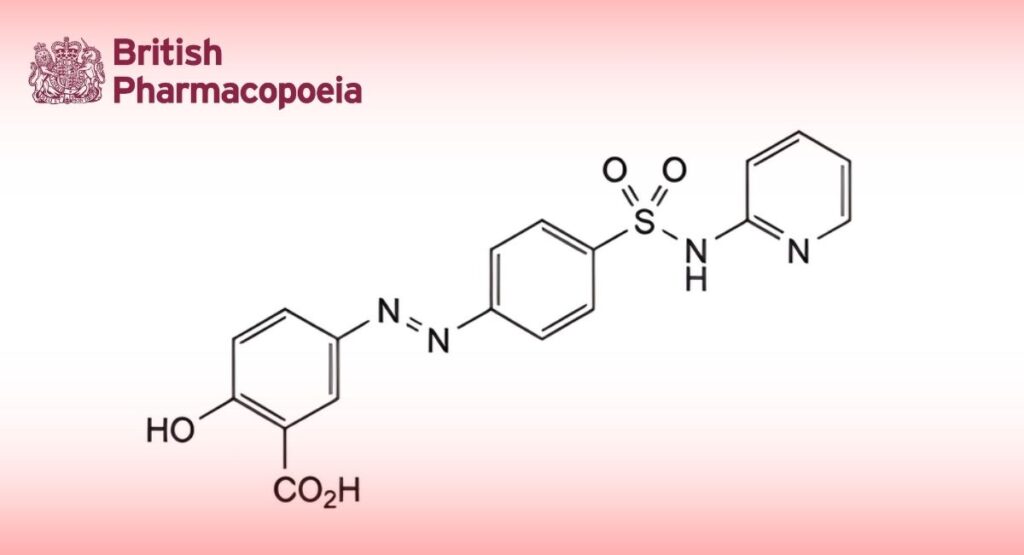
(Ph. Eur. monograph 0863)
C18H14N4O5S 398.4 599-79-1
Action and use
Sulfonamide aminosalicylate; treatment of ulcerative colitis.
Preparations
Sulfasalazine Gastro-resistant Tablets
Sulfasalazine Tablets
DEFINITION
2-Hydroxy-5-[2-[4-(pyridin-2-ylsulfamoyl)phenyl]diazenyl]benzoic acid.
Content
97.0 per cent to 101.5 per cent (dried substance).
CHARACTERS
Appearance
Bright yellow or brownish-yellow, fine powder.
Solubility
Practically insoluble in water, very slightly soluble in ethanol (96 per cent), practically insoluble in methylene chloride. It dissolves in dilute solutions of alkali hydroxides.
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Preparation: Discs.
Comparison: sulfasalazine CRS.
TESTS
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 25.0 mg of the substance to be examined in dilute ammonia R3 and dilute to 25.0 mL with the same solvent.
Reference solution (a): Dilute 1.0 mL of the test solution to 100.0 mL with dilute ammonia R3.
Reference solution (b): Dissolve 1.0 mg of sulfasalazine derivative for resolution CRS in 10.0 mL of reference solution (a).
Dilute 1.0 mL of this solution to 10.0 mL with reference solution (a).
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: octadecylsilyl silica gel for chromatography R (5 μm).
Mobile phase:
— mobile phase A: in a 1000 mL volumetric flask dissolve 1.13 g of sodium dihydrogen phosphate R and 2.5 g of sodium acetate R in 900 mL of water R; adjust to pH 4.8 with glacial acetic acid R and dilute to 1000 mL with water R;
— mobile phase B: mobile phase A, methanol R (10:40 V/V);
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 15 | 60 → 45 | 40 → 55 |
| 15 – 25 | 45 | 55 |
| 25 – 60 | 45 → 0 | 55 → 100 |
| 60 – 65 | 0 | 100 |
Flow rate: 1 mL/min.
Detection: Spectrophotometer at 320 nm.
Injection: 20 μL.
Relative retention: With reference to sulfasalazine: impurity H = about 0.16; impurity I = about 0.28; impurity C = about 0.80; impurity F = about 0.85; impurity G = about 1.39; impurity E = about 1.63; impurity B = about 1.85; impurity D = about 1.90; impurity A = about 2.00.
System suitability: Reference solution (b):
— resolution: minimum 3.0 between the peaks due to sulfasalazine and sulfasalazine derivative for resolution.
Limits:
— impurities A, B, C, D, E, F, G, I: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (1 per cent);
— total: not more than 4 times the area of the principal peak in the chromatogram obtained with reference solution (a) (4 per cent);
— disregard limit: 0.05 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent); disregard any peak with a retention time less than 6 min (due to impurities H and J).
Impurities H and J
Liquid chrom: Dissolve 25.0 mg of the substance to be examined in dilute ammonia R3 and dilute to 25.0 mL with the same solvent.
Reference solution (a): Dissolve 5.0 mg of salicylic acid R (impurity H) and 5.0 mg of sulfapyridine CRS (impurity J) in dilute ammonia R3 and dilute to 10.0 mL with the same solvent.
Reference solution (b): Dilute 2.0 mL of reference solution (a) to 100.0 mL with dilute ammonia R3.
Column:
— size: l = 0.25, Ø = 4.6 mm;
— stationary phase: octadecylsilyl silica gel for chromatography R (5 μm).
Mobile phase: Mobile phase B (described in the test for related substances), mobile phase A (described in the test for related substances) (30:70 V/V).
Flow rate: 1 mL/min.
Detection: Spectrophotometer at 300 nm.
Injection: 20 μL of the test solution and reference solution (b).
Run time: 10 min.
Retention time: Impurity H = about 6 min; impurity J = about 7 min.
System suitability: Reference solution (b):
— resolution: minimum 2 between the peaks due to impurities H and J.
Limits:
— impurities H, J: for each impurity, not more than 0.5 times the area of the corresponding peak in the chromatogram obtained with reference solution (b) (0.5 per cent);
— disregard limit: 0.05 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Chlorides (2.4.4)
Maximum 140 ppm.
To 1.25 g add 50 mL of distilled water R. Heat at about 70 °C for 5 min. Cool and filter. To 20 mL of the filtrate add 1 mL of nitric acid R, allow to stand for 5 min and filter to obtain a clear solution.
Sulfates (2.4.13)
Maximum 400 ppm.
To 20 mL of the filtrate prepared for the test for chlorides add 1 mL of dilute hydrochloric acid R, allow to stand for 5 min and filter.
Loss on drying (2.2.32)
Maximum 1.0 per cent, determined on 1.000 g by drying in an oven at 105 °C for 2 h.
Sulfated ash (2.4.14)
Maximum 0.5 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.150 g in 0.1 M sodium hydroxide and dilute to 100.0 mL with the same solvent. Transfer 5.0 mL of this solution to a 1000 mL volumetric flask containing about 750 mL of water R. Add 20.0 mL of a 6 g/L solution of glacial acetic acid R and dilute to 1000.0 mL with water R. Prepare a standard solution at the same time and in the same manner using 0.150 g of sulfasalazine CRS. Measure the absorbance (2.2.25) of the 2 solutions at the absorption maximum at 359 nm.
Calculate the content of C18H14N4O5S from the absorbances measured and the concentration of the solutions.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D, E, F, G, H, I, J.
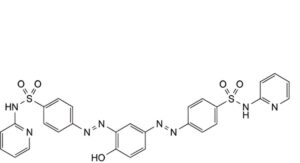
A. 4,4′-[(4-hydroxy-1,3-phenylene)bis(diazenediyl)]bis[N-(pyridin-2-yl)benzenesulfonamide],
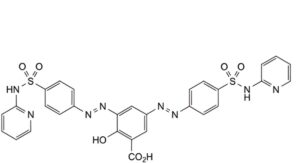
B. 2-hydroxy-3,5-bis[2-[4-(pyridin-2-ylsulfamoyl)phenyl]diazenyl]benzoic acid,
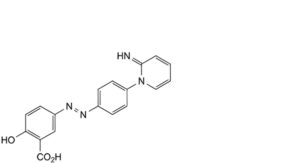
C. 2-hydroxy-5-[2-[4-(2-iminopyridin-1(2H)-yl)phenyl]diazenyl]benzoic acid,
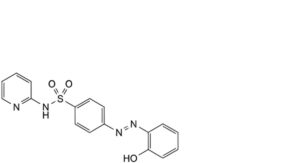
D. 4-[2-(2-hydroxyphenyl)diazenyl]-N-(pyridin-2-yl)benzenesulfonamide,
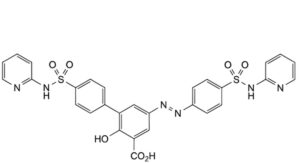
E. 2-hydroxy-4′-(pyridin-2-ylsulfamoyl)-5-[2-[4-(pyridin-2-ylsulfamoyl)phenyl]diazenyl]biphenyl-3-carboxylic acid,
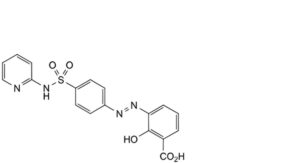
F. 2-hydroxy-3-[2-[4-(pyridin-2-ylsulfamoyl)phenyl]diazenyl]benzoic acid,
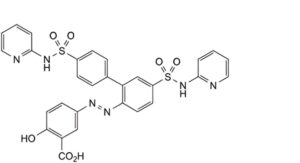
G. 5-[2-[4′,5-bis(pyridin-2-ylsulfamoyl)biphenyl-2-yl]diazenyl]-2-hydroxybenzoic acid,

H. 2-hydroxybenzenecarboxylic acid (salicylic acid),
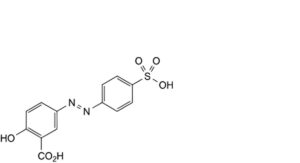
I. 2-hydroxy-5-[2-(4-sulfophenyl)diazenyl]benzoic acid,
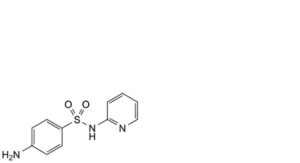
J. 4-amino-N-(pyridin-2-yl)benzenesulfonamide (sulfapyridine).