(Ph. Eur. monograph 2604)
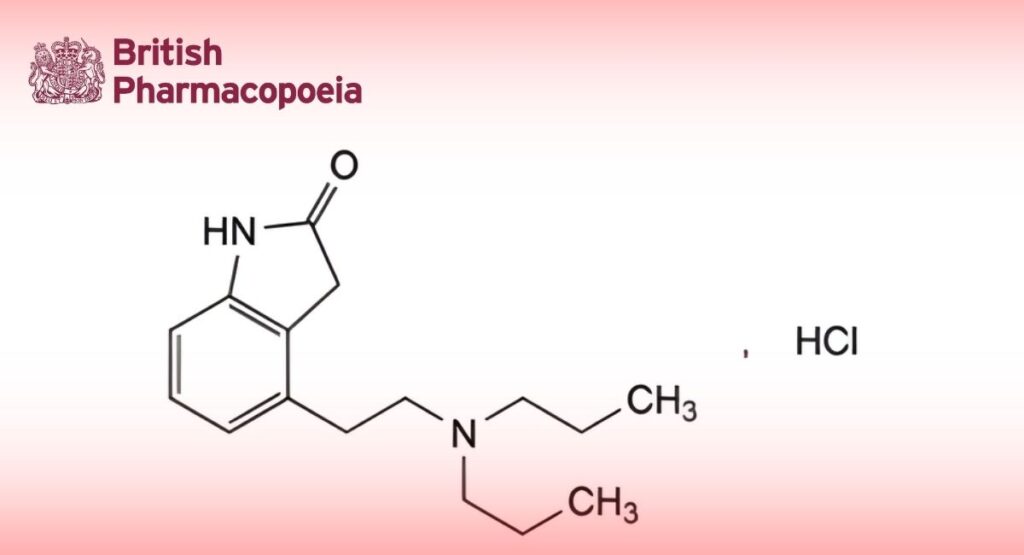
C16H25ClN2O 296.8 91374-20-8
Action and use
Dopamine receptor agonist.
DEFINITION
4-[2-(Dipropylamino)ethyl]-1,3-dihydro-2H-indol-2-one hydrochloride.
Content
98.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or yellowish, crystalline powder.
Solubility
Freely soluble in water and in methanol, slightly soluble in ethanol (96 per cent), practically insoluble in heptane.
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Comparison: ropinirole hydrochloride CRS.
B. It gives reaction (a) of chlorides (2.3.1).
TESTS
Related substances
Liquid chromatography (2.2.29).
Test solution (a): Dissolve, using sonication, 50.0 mg of the substance to be examined in 4 mL of mobile phase B and dilute to 25.0 mL with mobile phase A.
Test solution (b): Dilute 1.0 mL of test solution (a) to 20.0 mL with mobile phase A.
Reference solution (a): Dissolve, using sonication, 7.5 mg of ropinirole impurity A CRS and 5.0 mg of the substance to be examined in 16 mL of mobile phase B and dilute to 100.0 mL with mobile phase A.
Reference solution (b): To 1.0 mL of reference solution (a) add 4 mL of mobile phase B and dilute to 25.0 mL with mobile Dphase A.
Reference solution (c): Dissolve, using sonication, 50.0 mg of ropinirole hydrochloride CRS in 4 mL of mobile phase B and dilute to 25.0 mL with mobile phase A. Dilute 1.0 mL of the solution to 20.0 mL with mobile phase A.
Reference solution (d): Dissolve 5.0 mg of ropinirole for peak identification 1 CRS (containing impurity D) in mobile phase A and dilute to 5.0 mL with mobile phase A.
Reference solution (e): Dissolve 5.0 mg of ropinirole for peak identification 2 CRS (containing impurities G and H) in mobile phase A and dilute to 5.0 mL with mobile phase A.
Reference solution (f): Dissolve 5.0 mg of ropinirole for peak identification 3 CRS (containing impurity E) in mobile phase A and dilute to 5.0 mL with mobile phase A.
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: end-capped octylsilyl silica gel for chromatography R (5 μm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: dissolve 3.85 g of ammonium acetate R in 950 mL of water for chromatography R, adjust to pH 2.5 with dilute phosphoric acid R and dilute to 1000 mL with water for chromatography R;
— mobile phase B: methanol R, acetonitrile R1 (30:70 V/V);
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 15 | 92 | 8 |
| 15 – 30 | 92 → 80 | 8 → 20 |
| 30 – 50 | 80 → 60 | 20 → 40 |
| 50 – 75 | 60 | 40 |
Flow rate: 1.0 mL/min.
Detection: Spectrophotometer at 250 nm.
Injection: 10 μL of test solution (a) and reference solutions (a), (b), (d), (e) and (f).
Identification of impurities: Use the chromatogram obtained with reference solution (a) to identify the peak due to impurity A; use the chromatogram supplied with ropinirole for peak identification 1 CRS and the chromatogram obtained with reference solution (d) to identify the peak due to impurity D; use the chromatogram supplied with ropinirole for peak identification 2 CRS and the chromatogram obtained with reference solution (e) to identify the peaks due to impurities G and H; use the chromatogram supplied with ropinirole for peak identification 3 CRS and the chromatogram obtained with reference solution (f) to identify the peak due to impurity E.
Relative retention: With reference to ropinirole (retention time = about 30 min): impurity D = about 0.5; impurity A = about 0.9; impurity E = about 1.1; impurity H = about 1.2; impurity G = about 1.4.
System suitability:
— resolution: minimum 3.0 between the peaks due to impurity A and ropinirole in the chromatogram obtained with reference solution (a);
— signal-to-noise ratio: minimum 60 for the peak due to ropinirole in the chromatogram obtained with reference solution (b).
Calculation of percentage contents:
— correction factors: multiply the peak areas of the following impurities by the corresponding correction factor: impurity G = 2.7; impurity H = 0.5;
— for impurity A, use the concentration of impurity A in reference solution (b);
— for impurities other than A, use the concentration of ropinirole hydrochloride in reference solution (b).
Limits:
— impurities A, D, E, G, H: for each impurity, maximum 0.15 per cent;
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 0.5 per cent;
— reporting threshold: 0.05 per cent.
Water (2.5.12)
Maximum 0.5 per cent, determined on 1.000 g.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Mobile phase: Mobile phase B, mobile phase A (19:81 V/V).
Injection: Test solution (b) and reference solution (c).
Calculate the percentage content of C16H25ClN2O taking into account the assigned content of ropinirole hydrochloride CRS.
IMPURITIES
Specified impurities A, D, E, G, H.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) B, C, F.
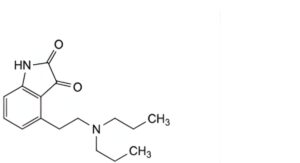
A. 4-[2-(dipropylamino)ethyl]-1H-indole-2,3-dione,
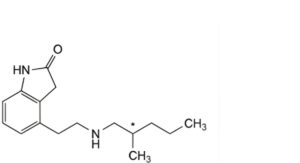
B. 4-[2-[(2-methylpentyl)amino]ethyl]-1,3-dihydro-2H-indol-2-one,
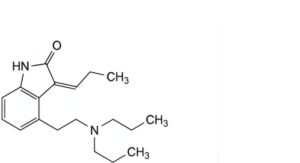
C. (3Z)-4-[2-(dipropylamino)ethyl]-3-propylidene-1,3-dihydro-2H-indol-2-one,
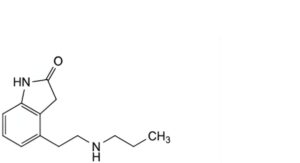
D. 4-[2-(propylamino)ethyl]-1,3-dihydro-2H-indol-2-one,
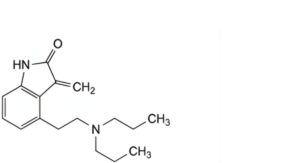
E. 4-[2-(dipropylamino)ethyl]-3-methylidene-1,3-dihydro-2H-indol-2-one,
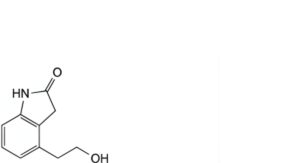
F. 4-(2-hydroxyethyl)-1,3-dihydro-2H-indol-2-one,
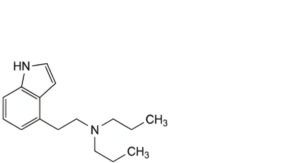
G. 4-[2-(dipropylamino)ethyl]-1H-indole,
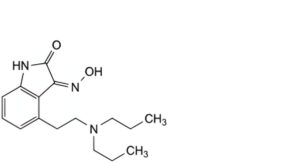
H. (3Z)-4-[2-(dipropylamino)ethyl]-3-(hydroxyimino)-1,3-dihydro-2H-indol-2-one.