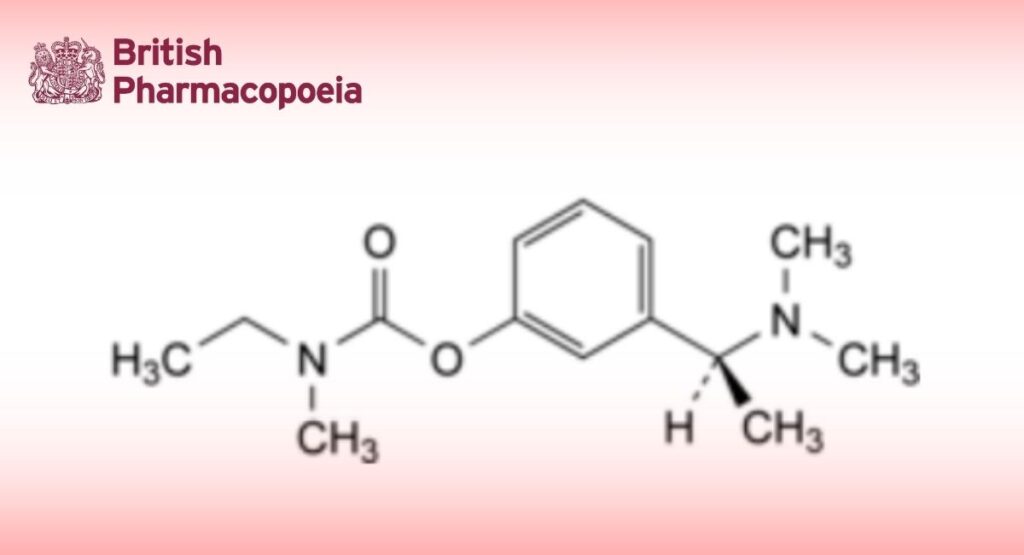
(Ph. Eur. monograph 2629)
C14H22N2O2 250.3 123441-03-2
Action and use
Cholinesterase inhibitor; treatment of dementia in Alzheimer’s disease and Parkinson’s disease.
Preparation
Rivastigmine Transdermal Patches
DEFINITION
3-[(1S)-1-(Dimethylamino)ethyl]phenyl ethyl(methyl)carbamate.
Content
98.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
Viscous, clear, colourless or yellow or very slightly brown, hygroscopic liquid.
Solubility
Sparingly soluble in water, very soluble in anhydrous ethanol and in heptane.
IDENTIFICATION
Carry out either tests A, B or tests B, C.
A. Specific optical rotation (2.2.7): -44.0 to -38.0 (anhydrous substance). Prepare the solution immediately before use.
Dissolve 0.300 g in ethyl acetate R and dilute to 50.0 mL with the same solvent.
B. Infrared absorption spectrophotometry (2.2.24).
Preparation: Film.
Comparison rivastigmine hydrogen tartrate CRS, treated as follows: dissolve 0.100 g in 30 mL of buffer solution pH 11 R, then add 30 mL of 1,1-dimethylethyl methyl ether R and shake vigorously for 2 min. Allow the layers to separate. Filter the upper organic layer through anhydrous sodium sulfate R. Evaporate the filtrate under reduced pressure at a temperature not exceeding 60 °C to obtain a residue. Record the reference spectrum using this residue.
C. Enantiomeric purity (see Tests).
TESTS
Enantiomeric purity
Liquid chromatography (2.2.29).
Solution A: Solution containing 1.78 g/L of disodium hydrogen phosphate dihydrate R and 1.38 g/L of sodium dihydrogen phosphate monohydrate R. Adjust to pH 6.0 with phosphoric acid R.
Test solution: Dissolve 25.0 mg of the substance to be examined in the mobile phase and dilute to 100.0 mL with the mobile phase. Dilute 5.0 mL of the solution to 20.0 mL with the mobile phase.
Reference solution (a): Dissolve 2.0 mg of rivastigmine impurity D CRS in the mobile phase and dilute to 200.0 mL with the mobile phase. Dilute 1.0 mL of the solution to 100.0 mL with the mobile phase.
Reference solution (b): Dissolve 1 mg of rivastigmine hydrogen tartrate CRS in reference solution (a) and dilute to 10 mL with reference solution (a).
Column:
— size: l = 0.10 m, Ø = 4.0 mm;
— stationary phase: α1-acid-glycoprotein silica gel for chiral separation R (5 μm).
Mobile phase: Mix 205 μL of N,N-dimethyloctylamine R and 20.0 mL of acetonitrile R1 and dilute to 1000 mL with solution A.
Flow rate: 0.5 mL/min.
Detection: Spectrophotometer at 200 nm.
Injection: 20 μL.
Run time: Twice the retention time of rivastigmine.
Relative retention: With reference to rivastigmine (retention time = about 9 min): impurity D = about 0.8.
System suitability Reference solution (b):
— peak-to-valley ratio: minimum 2.5, where Hp = height above the baseline of the peak due to impurity D and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to rivastigmine.
Calculation of percentage content:
— use the concentration of impurity D in reference solution (a).
Limit:
— impurity D: maximum 0.3 per cent.
Related substances
Liquid chromatography (2.2.29). Carry out the test protected from light.
Test solution: Dissolve 62.5 mg of the substance to be examined in the mobile phase and dilute to 100.0 mL with the mobile phase.
Reference solution (a): Dilute 1.0 mL of the test solution to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (b): Dissolve the contents of a vial of rivastigmine for system suitability CRS (containing impurities A, B and C) in 1 mL of the mobile phase.
Reference solution (c): Dissolve 50.0 mg of rivastigmine hydrogen tartrate CRS in the mobile phase and dilute to 50.0 mL with the mobile phase.
Column:
— size: l = 0.25 m, Ø = 4.0 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R1 (5 μm);
— temperature: 40 °C.
Mobile phase: Mix 42 volumes of an 8.9 g/L solution of disodium hydrogen phosphate dihydrate R previously adjusted to pH 7.0 with phosphoric acid R and 58 volumes of methanol R2.
Flow rate: 1.0 mL/min.
Detection: Spectrophotometer at 214 nm.
Injection: 20 μL of the test solution and reference solutions (a) and (b).
Run time: Twice the retention time of rivastigmine.
Identification of impurities: Use the chromatogram supplied with rivastigmine for system suitability CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities A, B and C.
Relative retention: With reference to rivastigmine (retention time = about 10 min): impurity A = about 0.4; impurity C = about 0.6; impurity B = about 0.7.
System suitability: Reference solution (b):
— resolution: minimum 2.0 between the peaks due to impurities C and B.
Calculation of percentage contents:
— for each impurity, use the concentration of rivastigmine in reference solution (a).
Limits:
— impurity A: maximum 0.3 per cent;
— impurity B: maximum 0.15 per cent;
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 0.5 per cent;
— reporting threshold: 0.05 per cent.
Water (2.5.12)
Maximum 0.5 per cent, determined on 1.000 g.
Change the solvent after standardisation of the titrant and after every 3nd sample.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection Test solution and reference solution (c).
System suitability Reference solution (c):
— symmetry factor: maximum 2.5 for the peak due to rivastigmine.
Calculate the percentage content of C14H22N2O2 taking into account the assigned content of rivastigmine hydrogen tartrate CRS and a conversion factor of 0.625.
STORAGE
Under an inert gas, in an airtight container, protected from light, at a temperature of 2 °C to 8 °C.
IMPURITIES
Specified impurities A, B, D.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) C.
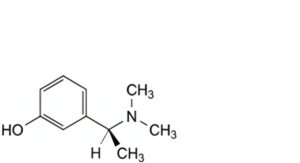
A. 3-[(1S)-1-(dimethylamino)ethyl]phenol (dimetol),
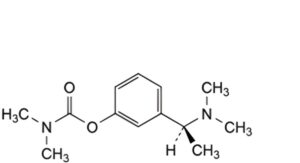
B. 3-[(1S)-1-(dimethylamino)ethyl]phenyl dimethylcarbamate,
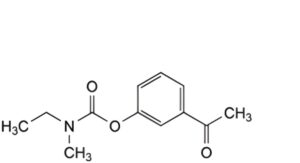
C. 3-acetylphenyl ethyl(methyl)carbamate,
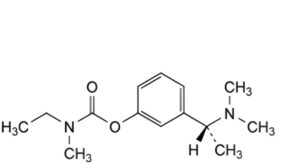
D. 3-[(1R)-1-(dimethylamino)ethyl]phenyl ethyl(methyl)carbamate ((R)-enantiomer).