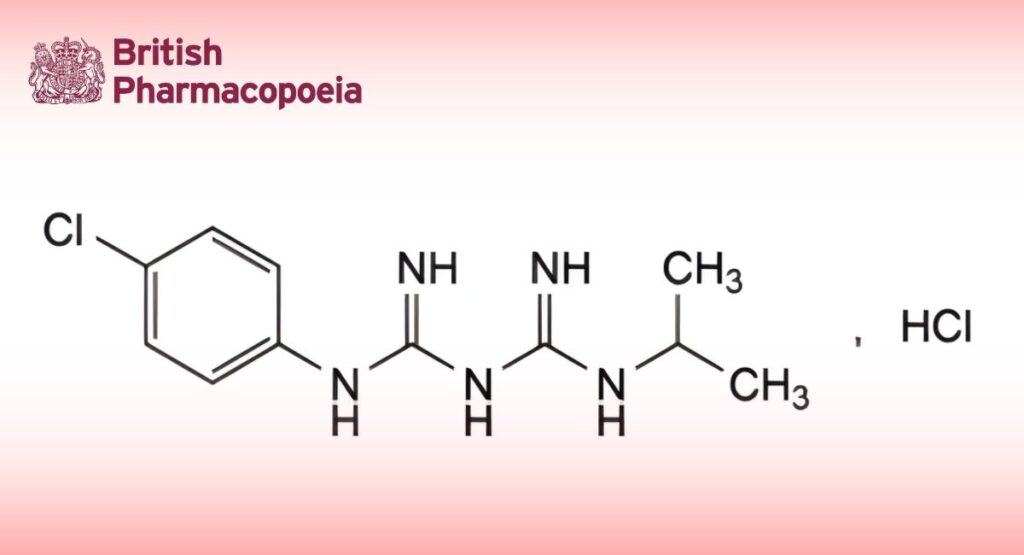
(Ph. Eur. monograph 2002)
C11H17Cl2N5 290.2 637-32-1
Action and use
Antiprotozoal (malaria).
Preparation
Proguanil Tablets
DEFINITION
N1 -(4-Chlorophenyl)-N3 -(propan-2-yl)imidodicarbonimidic diamide hydrochloride.
Content
98.5 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Slightly soluble in water, sparingly soluble in anhydrous ethanol, practically insoluble in methylene chloride.
IDENTIFICATION
First identification: A, C.
Second identification: B, C.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison: proguanil hydrochloride CRS.
B. Dissolve 0.4 g in 50 mL of water R (solution A). To 15 mL of solution A add 2 mL of dilute sodium hydroxide solution R. Extract with 20 mL of ethyl acetate R. Wash the organic layer with water R, evaporate to dryness and dry at 105 °C. The melting point (2.2.14) of the residue is 130 °C to 133 °C.
C. It gives reaction (a) of chlorides (2.3.1).
TESTS
Acidity or alkalinity
To 35 mL of water R maintained at 60-65 °C, add 0.2 mL of methyl red mixed solution R. Neutralise to a grey colour with either 0.01 M sodium hydroxide or 0.01 M hydrochloric acid. Add 0.4 g of the substance to be examined and stir until completely dissolved. The solution is grey or green. Not more than 0.2 mL of 0.01 M hydrochloric acid is required to change the colour of the solution to reddish-violet.
Related substances
Liquid chromatography (2.2.29).
Solvent mixture acetonitrile R, methanol R, water R (20:20:60 V/V/V).
Test solution (a): Dissolve 20 mg of the substance to be examined in the solvent mixture and dilute to 100.0 mL with the solvent mixture.
Test solution (b): Dissolve 20 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a): Dissolve 5 mg of proguanil for system suitability CRS (containing impurity G) in the solvent mixture and dilute to 25.0 mL with the solvent mixture.
Reference solution (b): Dilute 1.0 mL of test solution (a) to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Reference solution (c): Dissolve 4.0 mg of proguanil impurity B CRS in the solvent mixture and dilute to 100.0 mL with the solvent mixture. Dilute 1.0 mL of the solution to 50.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Column:
— size: l = 0.075 m, Ø = 4.6 mm;
— stationary phase: octylsilyl silica gel for chromatography R (3.5 μm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: dissolve 0.65 g of sodium pentanesulfonate R and 7.0 g of sodium perchlorate R in water for chromatography R and dilute to 1000 mL with the same solvent; adjust to pH 3.0 with a 1 per cent V/V solution of trifluoroacetic acid R;
— mobile phase B: dissolve 0.65 g of sodium pentanesulfonate R and 7.0 g of sodium perchlorate R in a mixture of 20 volumes of water for chromatography R, 40 volumes of acetonitrile for chromatography R and 40 volumes of methanol R1 and dilute to 1000 mL with the same solvent mixture; add 4 mL of a 1 per cent V/V solution of trifluoroacetic acid R;
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 3 | 75 | 25 |
| 3 – 23 | 75 → 40 | 25 → 60 |
Flow rate: 1.5 mL/min.
Detection: Spectrophotometer at 235 nm.
Injection: 20 μL of test solution (a) and reference solutions (a) and (b).
Identification of impurities: Use the chromatogram supplied with proguanil for system suitability CRS and the chromatogram obtained with reference solution (a) to identify the peak due to impurity G.
Relative retention: With reference to proguanil (retention time = about 16 min): impurity G = about 1.05.
System suitability Reference solution (a):
— peak-to-valley ratio: minimum 5.0, where Hp = height above the baseline of the peak due to impurity G and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to proguanil.
Calculation of percentage contents:
— for each impurity, use the concentration of proguanil hydrochloride in reference solution (b).
Limits:
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 0.2 per cent;
— reporting threshold: 0.05 per cent, except for impurity B.
Impurity B
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection: Test solution (b) and reference solution (c).
Identification of impurities Use the chromatogram obtained with reference solution (c) to identify the peak due to impurity B.
Relative retention: With reference to proguanil (retention time = about 16 min): impurity B = about 0.2.
System suitability: Reference solution (c):
— signal-to-noise ratio: minimum 10 for the peak due to impurity B.
Limit:
— impurity B: not more than the area of the corresponding peak in the chromatogram obtained with reference solution (c) (200 ppm).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Suspend 0.100 g in 20 mL of anhydrous acetic acid R, shake and heat at 50 °C for 5 min. Cool to room temperature and add 40 mL of acetic anhydride R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 14.51 mg of C11H17Cl2N5.
STORAGE
Protected from light.
IMPURITIES
Specified impurities B.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, C, D, E, F, G.
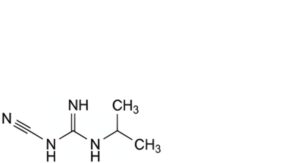
A. N-cyano-N′-(propan-2-yl)guanidine,
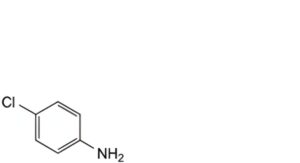
B. 4-chloroaniline,
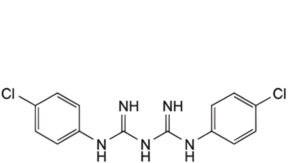
C. N1 ,N3 -bis(4-chlorophenyl)imidodicarbonimidic diamide,
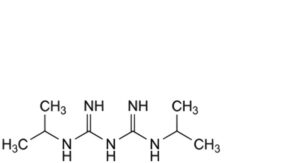
D. N1 ,N3 -bis(propan-2-yl)imidodicarbonimidic diamide,
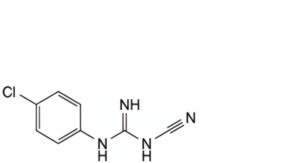
E. N-(4-chlorophenyl)-N′-cyanoguanidine,
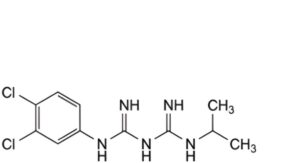
F. N1 -(3,4-dichlorophenyl)-N3 -(propan-2-yl)imidodicarbonimidic diamide,
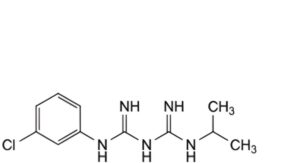
G. N1 -(3-chlorophenyl)-N3 -(propan-2-yl)imidodicarbonimidic diamide.