(Ph. Eur. monograph 1961)
DEFINITION
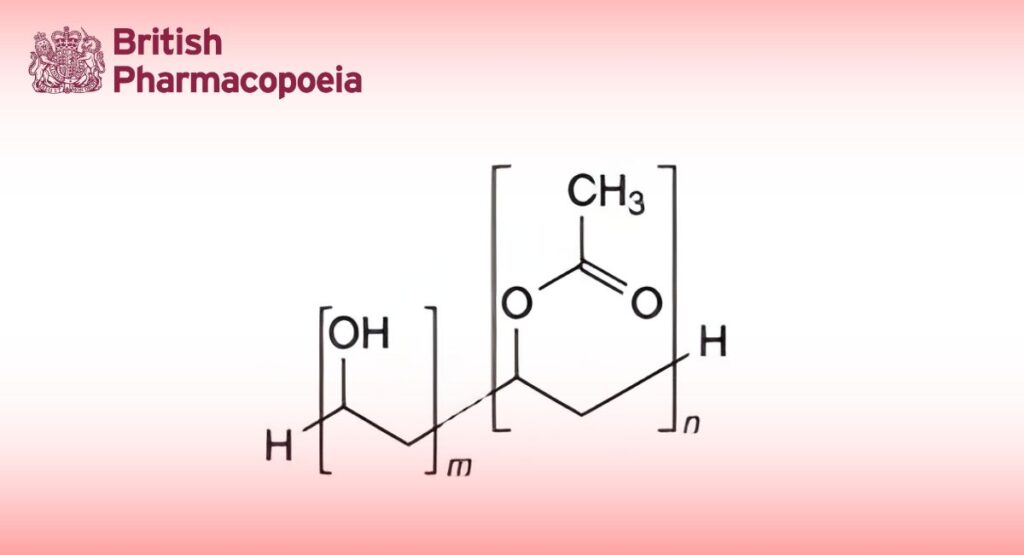
Poly(vinyl alcohol) is obtained by polymerisation of vinyl acetate, followed by partial or almost complete hydrolysis of poly(vinyl acetate) in the presence of catalytic amounts of alkali or mineral acids.
Poly(vinyl alcohol) polymers comply with the following indices:
0 ≤ n/m ≤ 0.35
The mean relative molecular mass lies between 20 000 and 150 000. The viscosity is 3 mPa·s to 70 mPa·s. The ester value, which characterises the degree of hydrolysis, is not greater than 280.
CHARACTERS
Appearance
Yellowish-white powder or translucent granules.
Solubility
Soluble in water, slightly soluble in anhydrous ethanol, practically insoluble in acetone.
Various grades of poly(vinyl alcohol) are available. They differ in their degree of polymerisation and their degree of hydrolysis which determine the physical properties of the different grades. They are characterised by the viscosity and the
ester value of the substance.
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Comparison: poly(vinyl alcohol) CRS.
The intensities of the absorption bands at about 1720 cm and 1260 cm are inversely proportional to the degree of hydrolysis.
B. Viscosity (see Tests).
TESTS
Solution S
Heat on a water-bath 250 mL of water R in a borosilicate round-bottomed flask attached to a reflux condenser with stirrer, add 10.0 g of the substance to be examined (correcting for the loss on drying) and continue heating for 30 min with continuous stirring. Remove the flask from the water-bath and continue stirring until room temperature is reached.
Appearance of solution
Solution S is clear (2.2.1) and not more intensely coloured than reference solution Y7 (2.2.2, Method II).
pH (2.2.3)
4.5 to 6.5 for solution S.
Viscosity (2.2.49, Method A)
85 per cent to 115 per cent of the value stated on the label.
Determine the viscosity using a falling ball viscometer immediately after preparation of solution S at 20 ± 0.1 °C.
Acid value
Maximum 3.0.
Add 1 mL of phenolphthalein solution R to 50 mL of solution S and titrate with 0.05 M potassium hydroxide until the pink colour persists for 15 s. Calculate the acid value using the following expression:
2.805V / 2
V = volume of 0.05 M potassium hydroxide used, in millilitres.
Ester value (2.5.2)
90 per cent to 110 per cent of the value stated on the label.
Saponify (2.5.6) 1.00 g in a mixture of 25.0 mL of 0.5 M alcoholic potassium hydroxide and 25.0 mL of water R.
Loss on drying (2.2.32)
Maximum 5.0 per cent, determined on 1.000 g by drying in an oven at 105 °C for 3 h.
Sulfated ash (2.4.14)
Maximum 1.0 per cent, determined on 1.0 g.
LABELLING
The label states:
— the viscosity for a 40 g/L solution;
— the ester value.
FUNCTIONALITY-RELATED CHARACTERISTICS
This section provides information on characteristics that are recognised as being relevant control parameters for one or more functions of the substance when used as an excipient (see chapter 5.15). Some of the characteristics described in the Functionality-related characteristics section may also be present in the mandatory part of the monograph since they also represent mandatory quality criteria. In such cases, a cross-reference to the tests described in the mandatory part is included in the Functionality-related characteristics section. Control of the characteristics can contribute to the quality of a medicinal product by improving the consistency of the manufacturing process and the performance of the medicinal product during use. Where control methods are cited, they are recognised as being suitable for the purpose, but other methods can also be used. Wherever results for a particular characteristic are reported, the control method must be indicated.
The following characteristics may be relevant for poly(vinyl alcohol) used as viscosity-increasing agent, binder or film former.
Viscosity
(see Tests).
Ester value
(see Tests).
Infrared absorption spectrophotometry
(see Identification A).