Edition: BP 2025 (Ph. Eur. 11.6 update)
Action and use
Penicillin antibacterial.
Ph Eur
DEFINITION
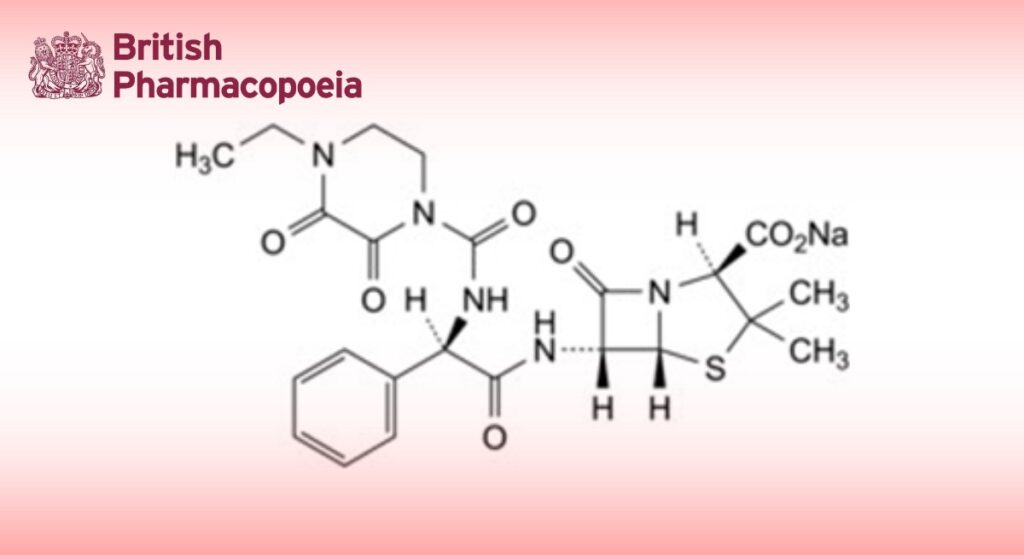
Sodium (2S,5R,6R)-6-[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia- 1-azabicyclo[3.2.0]heptane-2-carboxylate.
Semi-synthetic product derived from a fermentation product.
Content
95.5 per cent to 102.0 per cent (anhydrous substance).
PRODUCTION
The manufacturing process is evaluated to determine the potential presence of N,N-dimethylaniline. Where necessary, the manufacturing process is validated to demonstrate that the piperacillin sodium complies with the following test:
N,N-Dimethylaniline (2.4.26, Method A) Maximum 20 ppm.
CHARACTERS
Appearance
White or almost white, hygroscopic powder.
Solubility
Freely soluble in water and in methanol, practically insoluble in ethyl acetate.
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Preparation Dissolve 0.250 g in water R, add 0.5 mL of dilute hydrochloric acid R and 5 mL of ethyl acetate R; stir and allow to stand for 10 min in iced water. Filter the crystals through a small sintered-glass filter (40), applying suction. Wash with 5 mL of water R and 5 mL of ethyl acetate R, then dry in an oven at 60 °C for 60 min.
Comparison piperacillin CRS.
B. It gives reaction (a) of sodium (2.3.1).
TESTS
Solution S
Dissolve 2.50 g in carbon dioxide-free water R and dilute to 25 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and its absorbance (2.2.25) at 430 nm is not greater than 0.10.
pH (2.2.3)
5.0 to 7.0 for solution S.
Related substances
Liquid chromatography (2.2.29).
Test solution Dissolve 0.120 g of the substance to be examined in water R and dilute to 20.0 mL with the same solvent.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with mobile phase B.
Reference solution (b) Dissolve 6 mg of piperacillin impurity I CRS in mobile phase B and dilute to 20 mL with mobile phase B.
Reference solution (c) Dissolve 6 mg of anhydrous ampicillin CRS (impurity A) in mobile phase B and dilute to 20 mL with mobile phase B.
Reference solution (d) To 2 mL of reference solution (b), add 1 mL of reference solution (c) and dilute to 10 mL with mobile phase B.
Reference solution (e) Dissolve 6 mg of piperacillin for peak identification CRS (containing impurities A, B, C, D, E, F, G, I, J, K, L, M, O, P, Q, R, S and T) in mobile phase B and dilute to 1 mL with mobile phase B.
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: end-capped octadecylsilyl amorphous organosilica polymer for chromatography R (5 µm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: mix 3 mL of a 320 g/L solution of tetrabutylammonium hydroxide R, 100 mL of a 27.6 g/L solution of sodium dihydrogen phosphate R, 275 mL of methanol R1 and 622 mL of water for chromatography R; adjust the apparent pH to 5.5 with phosphoric acid R;
— mobile phase B: mix 3 mL of a 320 g/L solution of tetrabutylammonium hydroxide R, 100 mL of a 27.6 g/L solution of sodium dihydrogen phosphate R, 282 mL of water for chromatography R and 615 mL of methanol R1; adjust the apparent pH to 5.5 with phosphoric acid R;
| Time (min) | Mobile phase A (per cent V/V) | Mobile phase B (per cent V/V) |
| 0 – 6 | 100 | 0 |
| 6 – 55 | 100 → 71 | 0 → 29 |
| 55 – 73 | 71 → 10 | 29 → 90 |
| 73 – 85 | 10 | 90 |
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 220 nm.
Autosampler Set at 4 °C.
Injection 10 µL of the test solution and reference solutions (a), (d) and (e).
Identification of impurities Use the chromatogram supplied with piperacillin for peak identification CRS and the chromatogram obtained with reference solution (e) to identify the peaks due to impurities A, B, C, D, E, F, G, I, J, K, L, M, O, P, Q, R, S and T.
Relative retention With reference to piperacillin (retention time = about 54 min): impurity E = about 0.05; impurity I = about 0.12; impurity A = about 0.14; impurity G = about 0.30; impurity J = about 0.36; impurity F = about 0.57; impurity K = about 0.60; impurity L = about 0.65; impurity B (isomer 1) = about 0.71; impurity M = about 0.75; impurity B (isomer 2) = about 0.83; impurity C (isomer 1) = about 0.87; impurity C (isomer 2) = about 0.92; impurity O = about 1.23; impurity P = about 1.26; impurity Q = about 1.31; impurity R = about 1.36; impurity S = about 1.38; impurity T = about 1.41; impurity D = about 1.54.
System suitability Reference solution (d):
— resolution: minimum 1.5 between the peaks due to impurities I and A.
Calculation of percentage contents:
— correction factors: multiply the peak areas of the following impurities by the corresponding correction factor: impurity A = 1.3; impurity E = 0.4; impurity I = 3.2;
— for each impurity, use the concentration of piperacillin sodium in reference solution (a).
Limits:
— impurity G: maximum 1.5 per cent;
— impurities B (sum of isomers), D: for each impurity, maximum 1.0 per cent;
— impurity F: maximum 0.8 per cent;
— impurity C (sum of isomers): maximum 0.7 per cent;
— impurity S: maximum 0.5 per cent;
— impurities L, T: for each impurity, maximum 0.3 per cent;
— impurities A, E, I, J, K, M, O, P, Q, R: for each impurity, maximum 0.2 per cent;
— any other impurity: for each impurity, maximum 0.15 per cent;
— total: maximum 2.5 per cent;
— reporting threshold: 0.05 per cent.
Water (2.5.12)
Maximum 2.0 per cent, determined on 0.500 g.
ASSAY
Liquid chromatography (2.2.29).
Solvent mixture acetonitrile R, 31.2 g/L solution of sodium dihydrogen phosphate R (25:75 V/V).
Test solution Dissolve 0.100 g of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture. Dilute 5.0 mL of the solution to 50.0 mL with the solvent mixture.
Reference solution (a) Dissolve 50.0 mg of piperacillin CRS in the solvent mixture and dilute to 50.0 mL with the solvent mixture. Dilute 10.0 mL of the solution to 50.0 mL with the solvent mixture.
Reference solution (b) Dissolve 0.1 g of the substance to be examined in the solvent mixture and dilute to 50 mL with the solvent mixture.
Reference solution (c) Dissolve 5 mg of piperacillin impurity N CRS in the solvent mixture and dilute to 25 mL with the solvent mixture.
Reference solution (d) To 5 mL of reference solution (b), add 0.1 mL of reference solution (c) and dilute to 50 mL with the solvent mixture.
Column:
— size: l = 0.15 m, Ø = 4.6 mm;
— stationary phase: end-capped octadecylsilyl amorphous organosilica polymer for chromatography R (3.5 µm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: mix 24 mL of an 80 g/L solution of tetrabutylammonium hydroxide R, 200 mL of acetonitrile for chromatography R, 200 mL of a 31.2 g/L solution of sodium dihydrogen phosphate R and 576 mL of water for chromatography R; adjust the apparent pH to 5.5 with dilute phosphoric acid R or dilute sodium hydroxide solution R;
— mobile phase B: mix 24 mL of an 80 g/L solution of tetrabutylammonium hydroxide R, 126 mL of water for chromatography R, 200 mL of a 31.2 g/L solution of sodium dihydrogen phosphate R and 650 mL of acetonitrile for chromatography R; adjust the apparent pH to 5.5 with dilute phosphoric acid R or dilute sodium hydroxide solution R;
| Time (min) | Mobile phase A (per cent V/V) | Mobile phase B (per cent V/V) |
| 0 – 3.5 | 100 | 0 |
| 3.5 – 4 | 100 → 92 | 0 → 8 |
| 4 – 14 | 92 → 86 | 8 → 14 |
| 14 – 15 | 86 → 0 | 14 → 100 |
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 220 nm.
Injection 10 µL of the test solution and reference solutions (a) and (d).
Relative retention With reference to piperacillin (retention time = about 13 min): impurity N = about 0.96.
System suitability Reference solution (d):
— resolution: minimum 1.5 between the peaks due to impurity N and piperacillin.
Calculate the percentage content of C23H26N5NaO7S using the chromatogram obtained with reference solution (a) and taking into account the assigned content of piperacillin CRS and a conversion factor of 1.042.
STORAGE
In an airtight container. If the substance is sterile, the container is also sterile and tamper-evident.
LABELLING
The label states, where applicable, that the substance is suitable for use in the manufacture of parenteral preparations.
IMPURITIES
Specified impurities A, B, C, D, E, F, G, I, J, K, L, M, O, P, Q, R, S, T.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities. It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) H, N.
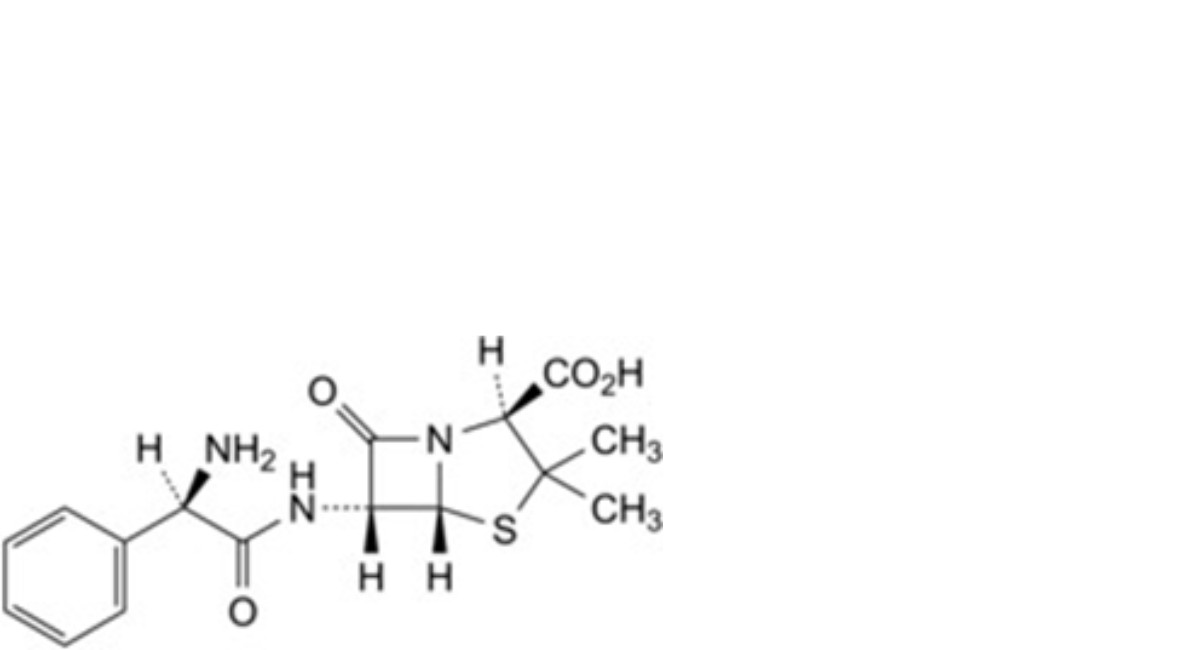
A. (2S,5R,6R)-6-[(2R)-2-amino-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid (ampicillin),
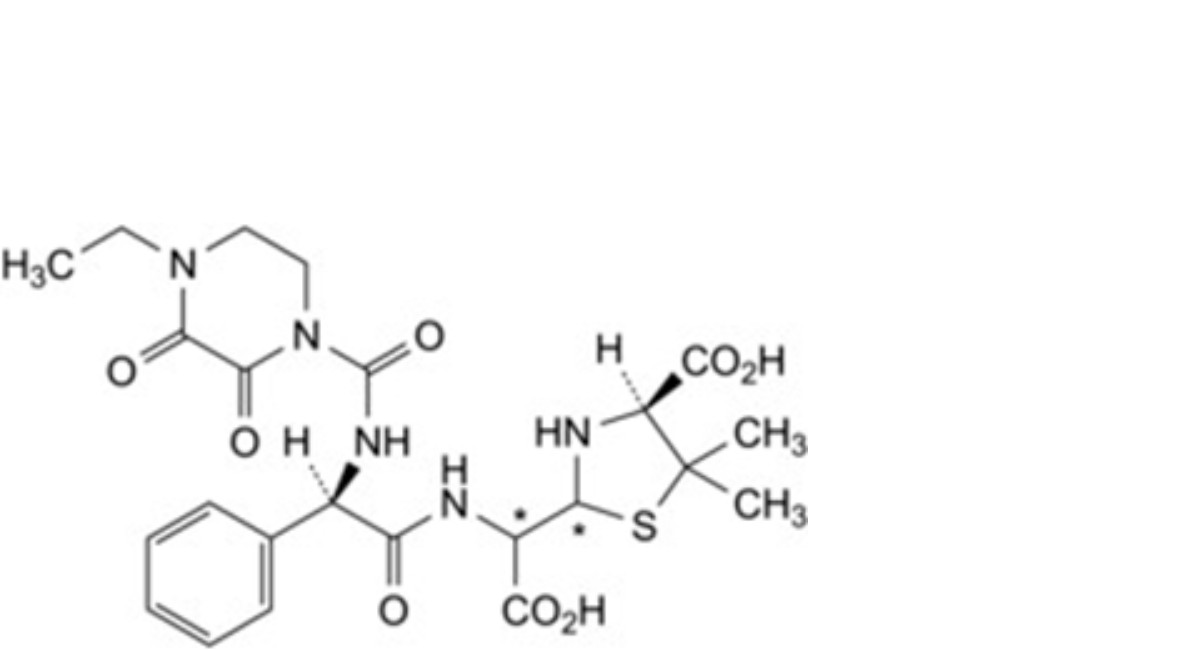
B. (2Ξ,4S)-2-[(Ξ)-carboxy[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]methyl]-5,5-dimethyl- 1,3-thiazolidine-4-carboxylic acid (penicilloic acids of piperacillin),
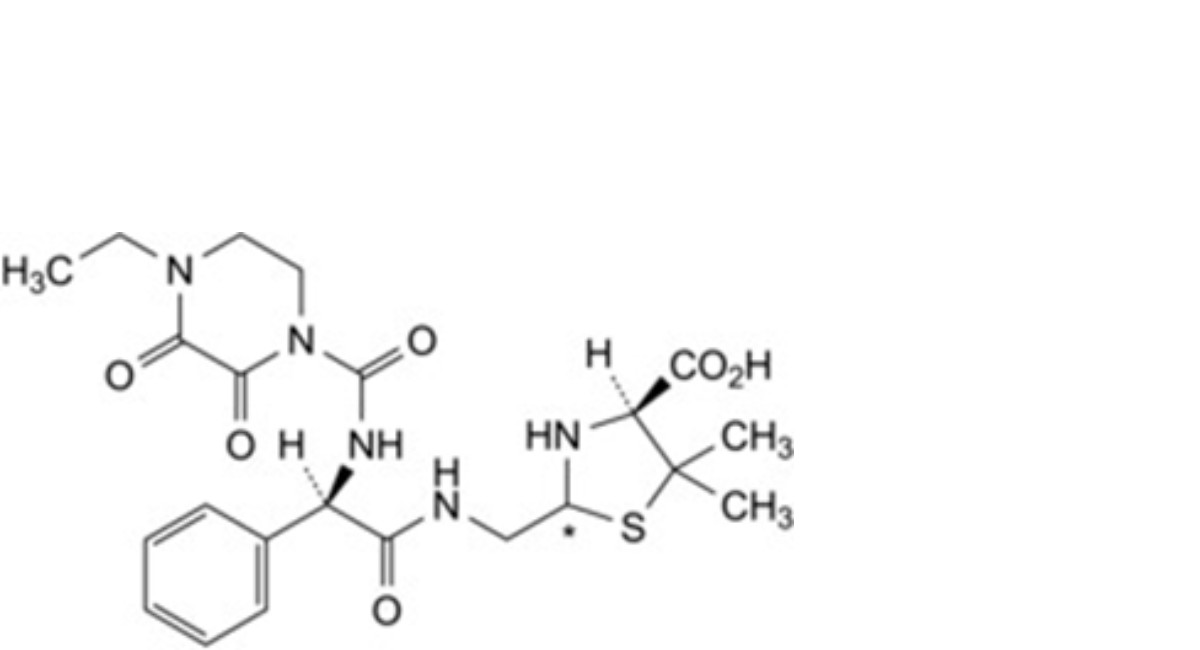
C. (2Ξ,4S)-2-[[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]methyl]-5,5-dimethyl-1,3- thiazolidine-4-carboxylic acid (penilloic acids of piperacillin),
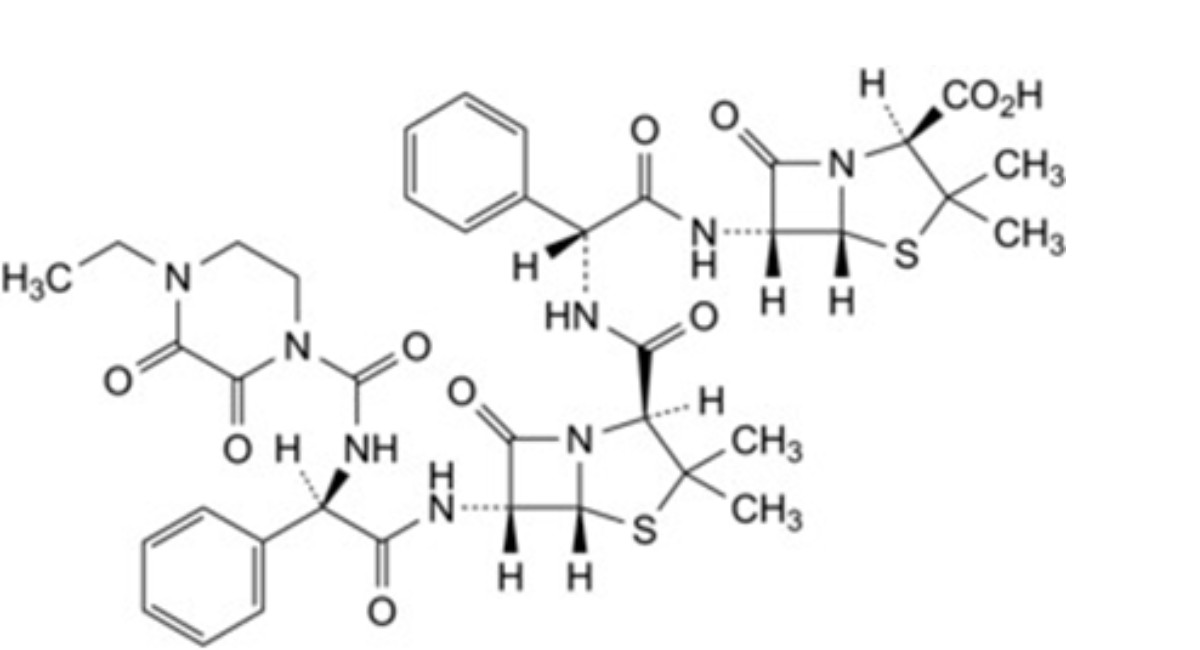
D. (2S,5R,6R)-6-[(2R)-2-[(2S,5R,6R)-6-[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]-3,3- dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxamido]-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1- azabicyclo[3.2.0]heptane-2-carboxylic acid (piperacillinylampicillin),
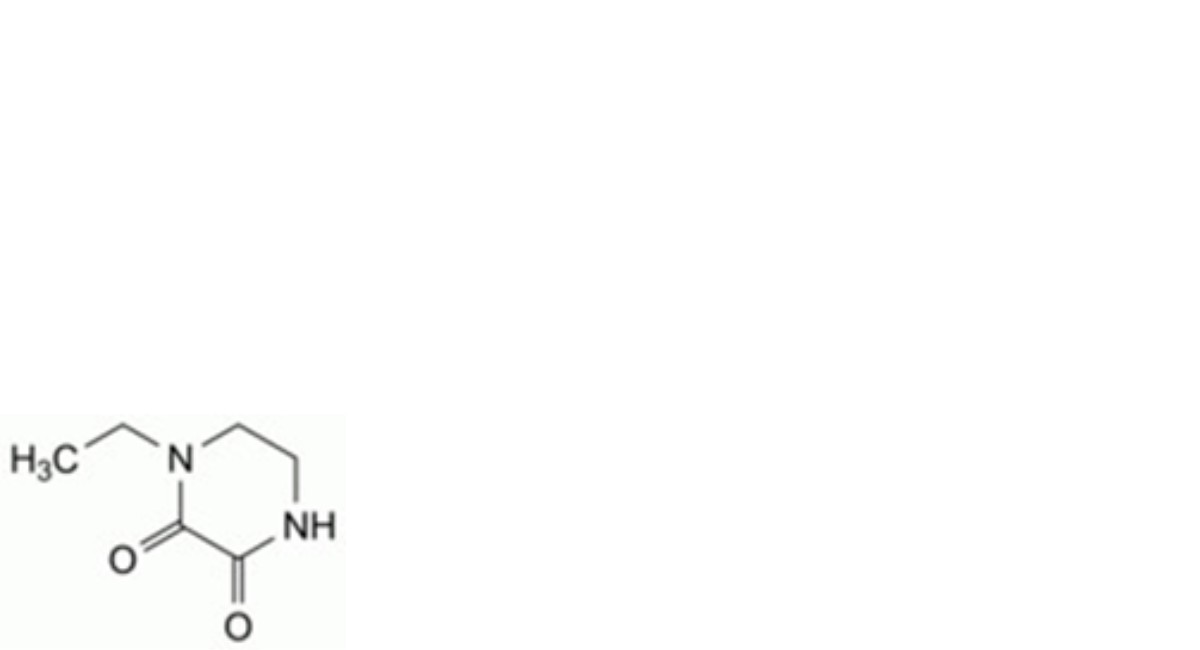
E. 1-ethylpiperazine-2,3-dione,
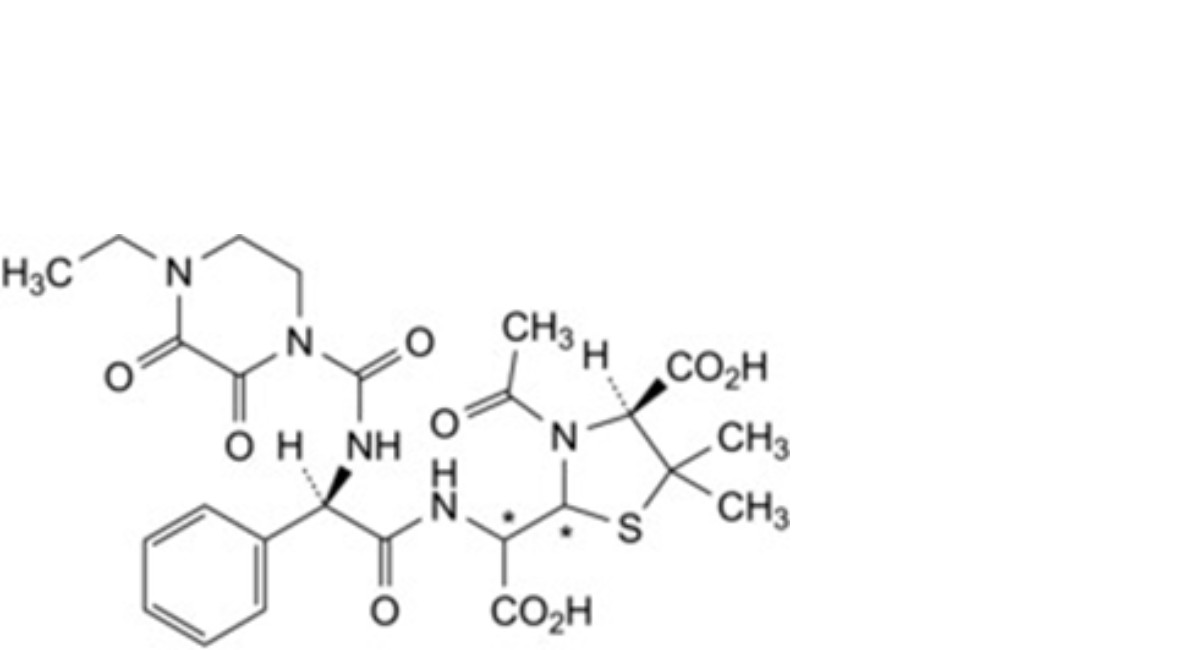
F. (2Ξ,4S)-3-acetyl-2-[(Ξ)-carboxy[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]methyl]-5,5- dimethyl-1,3-thiazolidine-4-carboxylic acid (acetylated penicilloic acids of piperacillin),
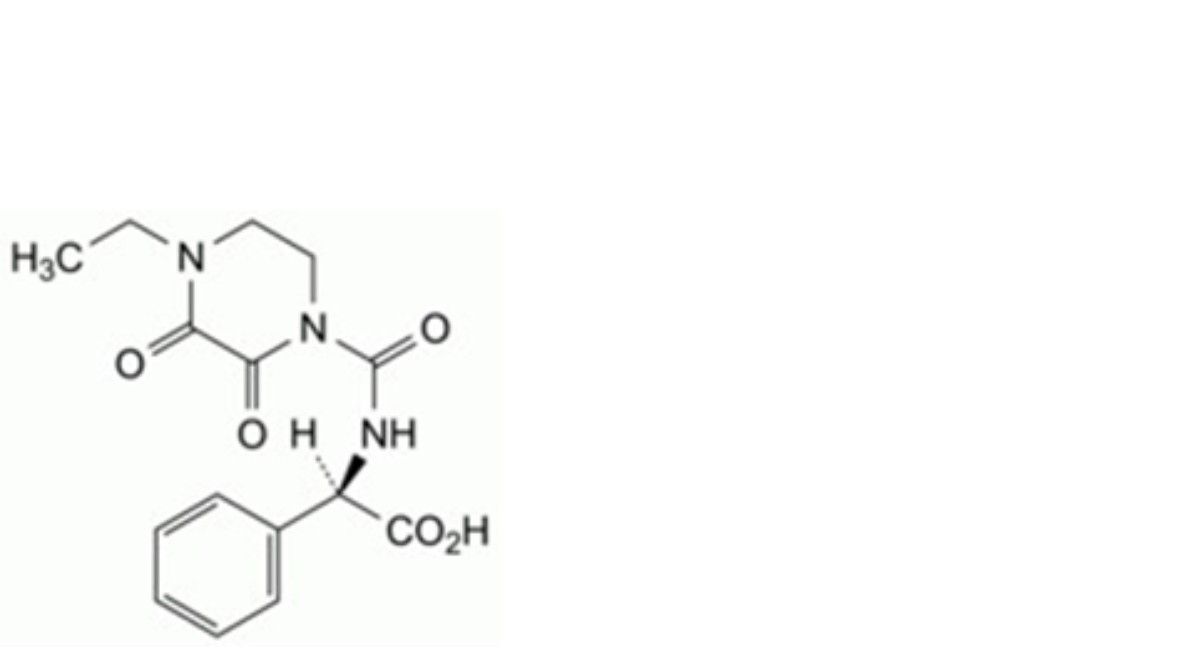
G. (R)-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)phenylacetic acid,
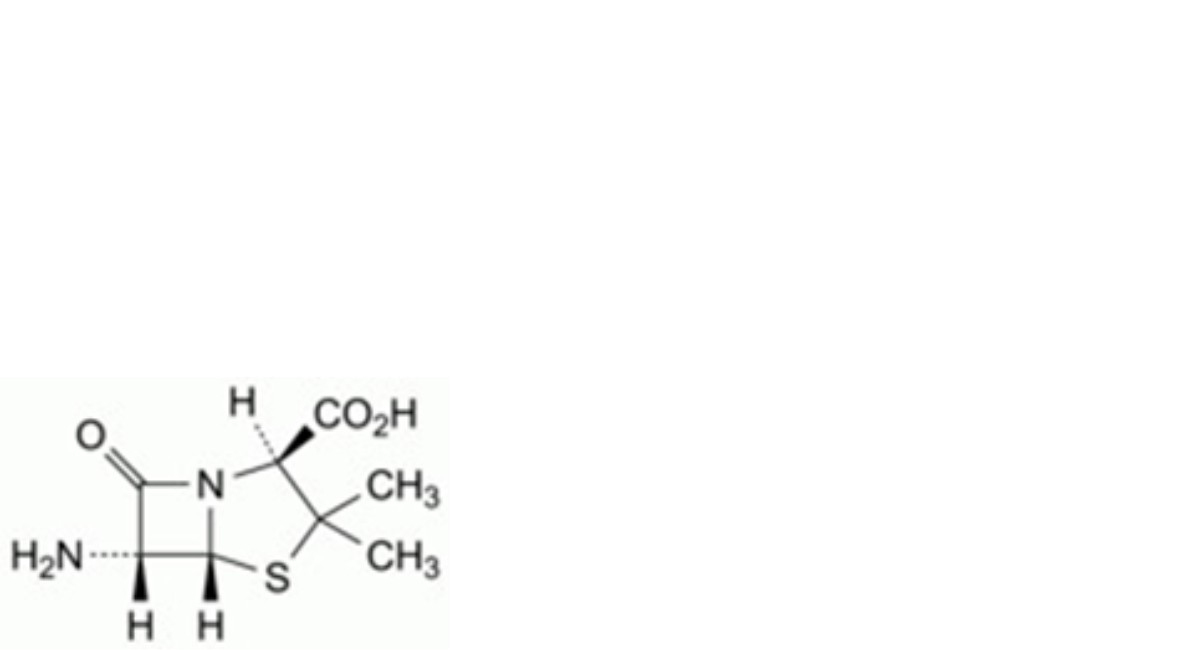
H. (2S,5R,6R)-6-amino-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid (6-aminopenicillanic acid),
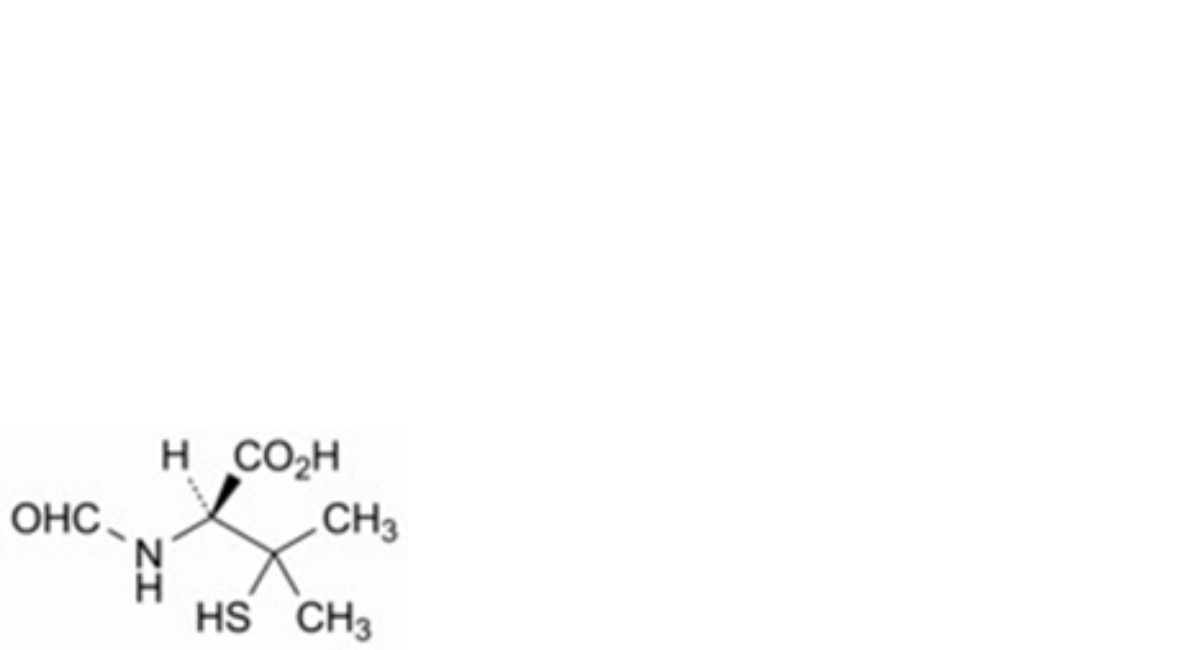
I. (2S)-2-formamido-3-methyl-3-sulfanylbutanoic acid (N-formylpenicillamine),
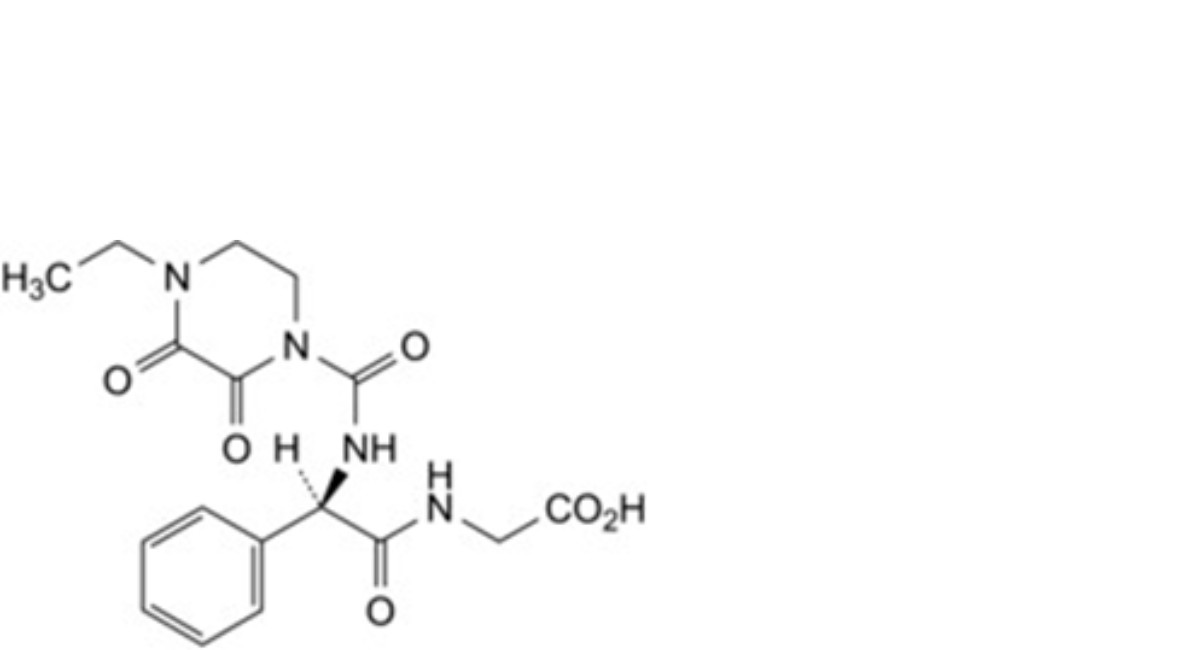
J. [(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]acetic acid,
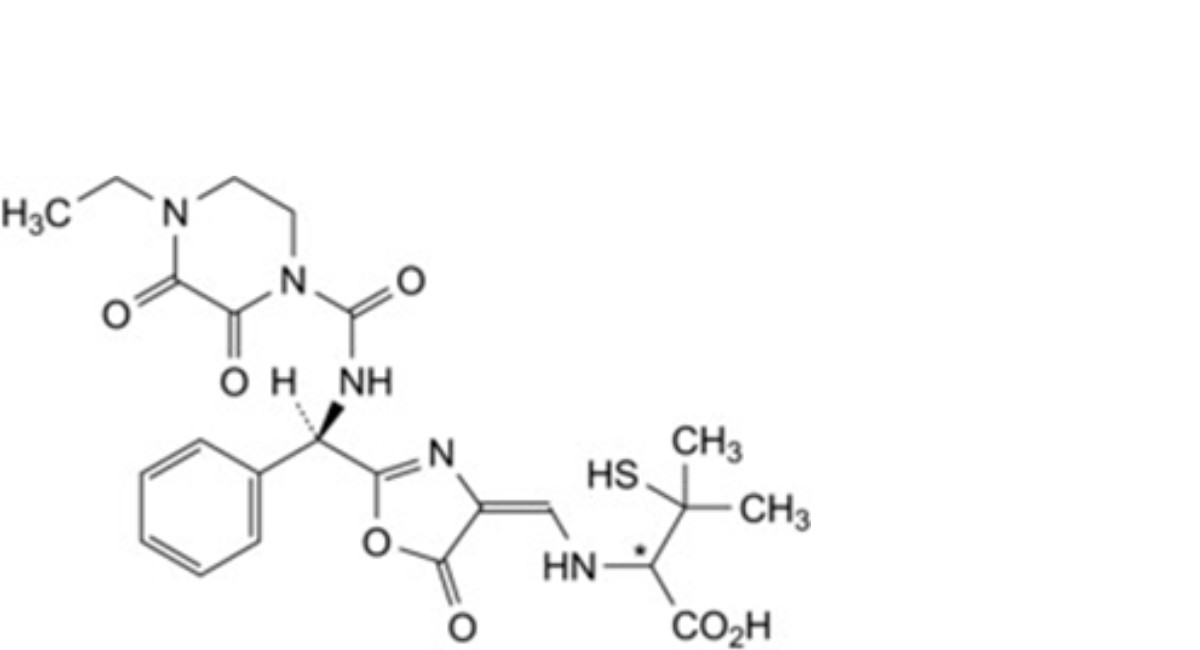
K. (2Ξ)-2-[[(E)-[2-[(R)-[(4-ethyl-2,3-dioxopiperazine-1-carboxamido)phenylmethyl]-5-oxo-1,3-oxazol-4(5H)- ylidene]methyl]amino]-3-methyl-3-sulfanylbutanoic acid (penicillenic acid),
L. unknown structure,
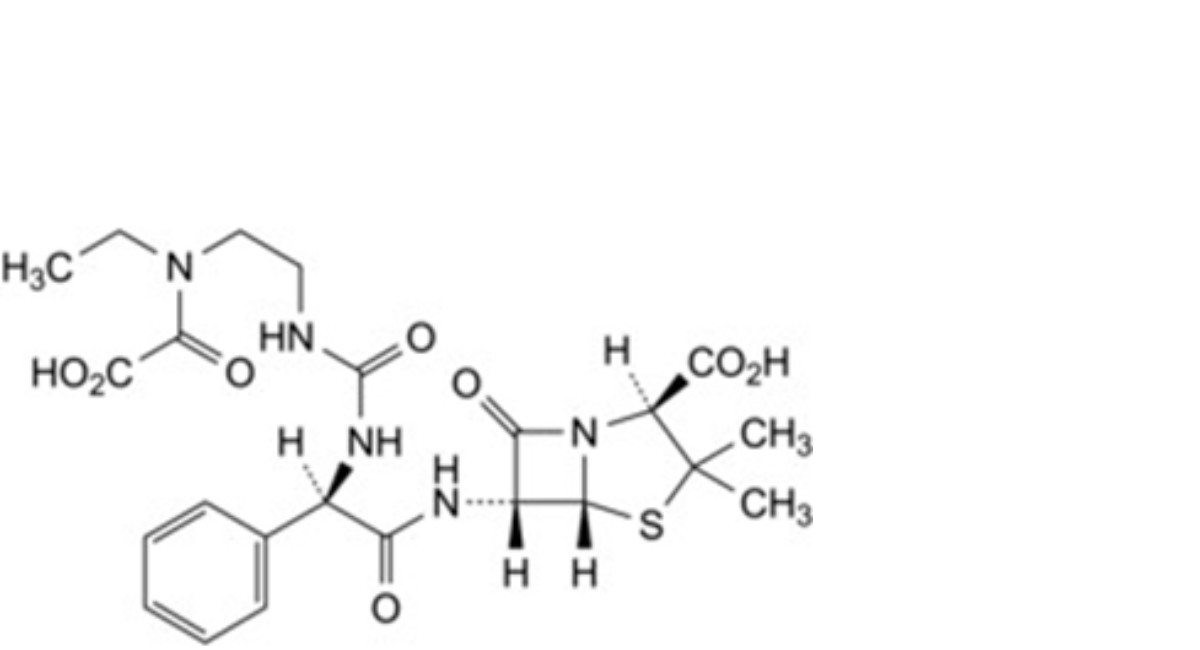
M. (2S,5R,6R)-6-[(2R)-2-[[[2-[ethyl(oxalo)amino]ethyl]carbamoyl]amino]-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1- azabicyclo[3.2.0]heptane-2-carboxylic acid,
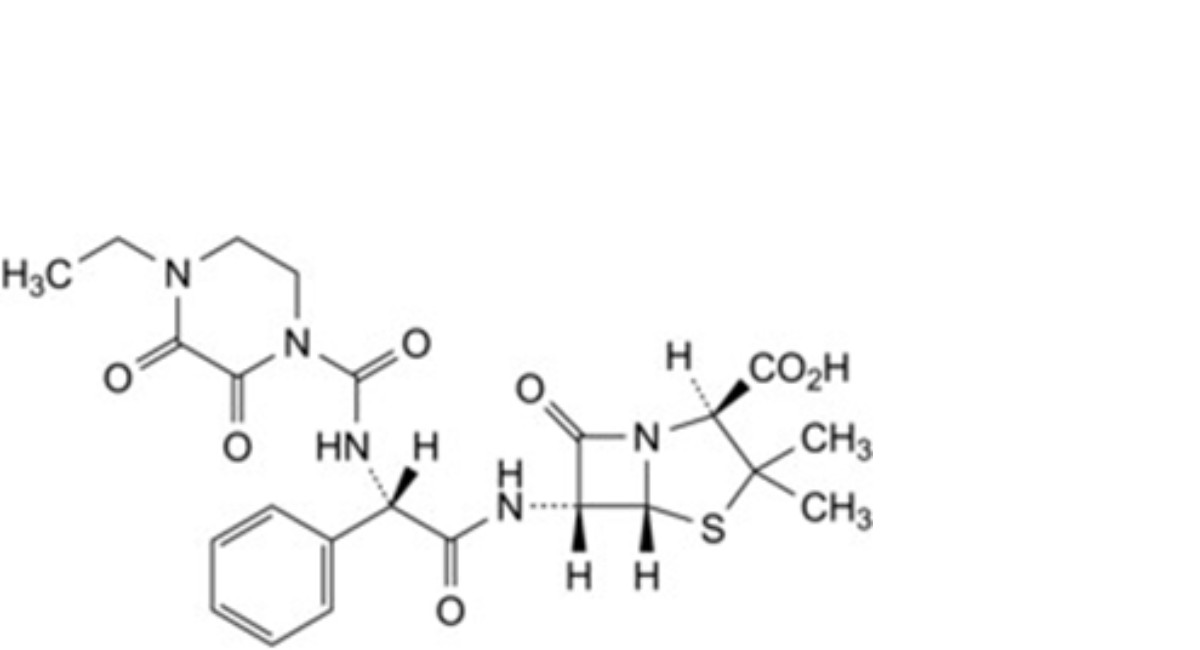
N. (2S,5R,6R)-6-[(2S)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1- azabicyclo[3.2.0]heptane-2-carboxylic acid (L-piperacillin),
O. unknown structure,
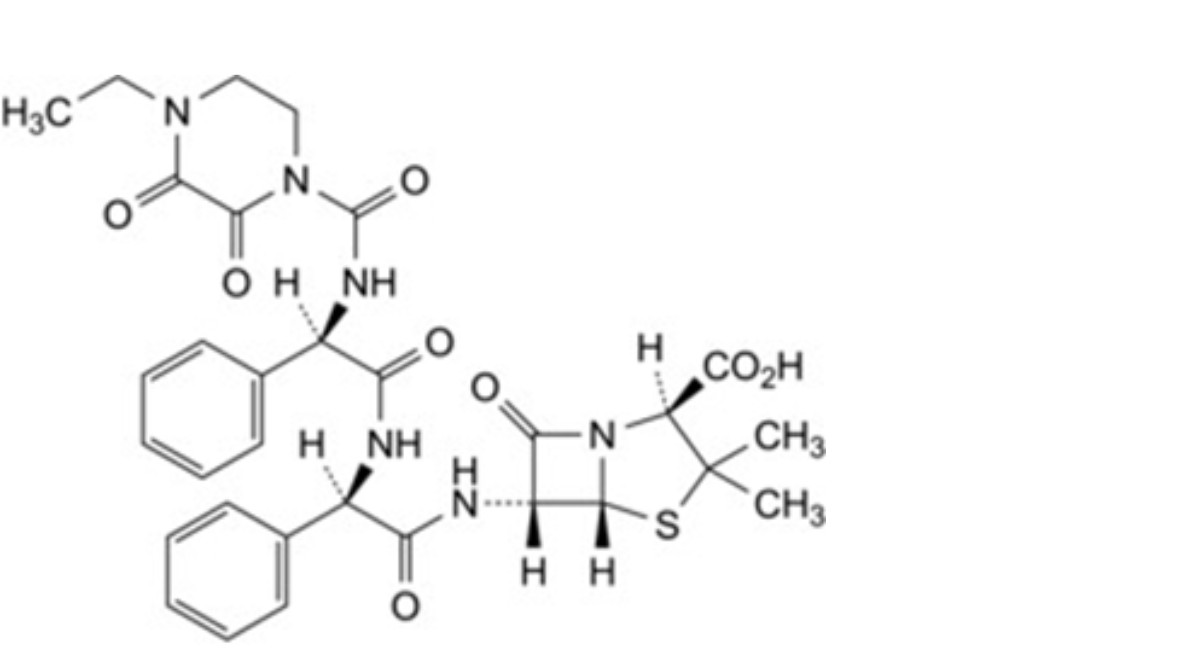
P. (2S,5R,6R)-6-[(2R)-2-[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]-2- phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid,
Q. unknown structure,
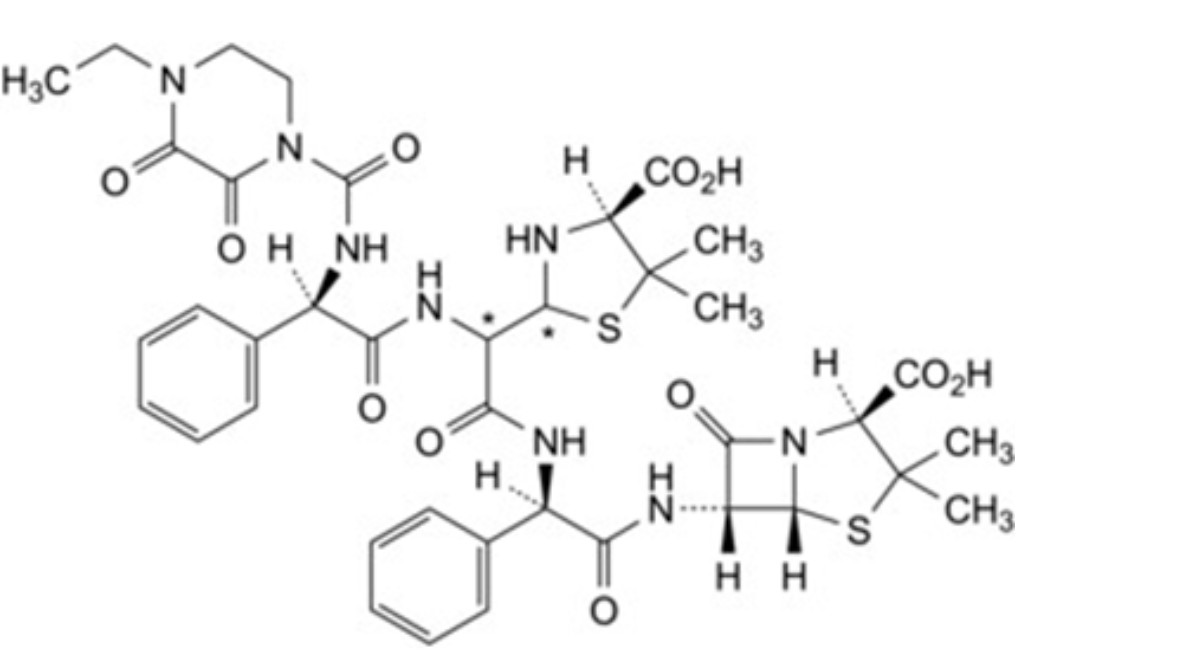
R. (2S,5R,6R)-6-[(2R)-2-[(2Ξ)-2-[(2Ξ,4S)-4-carboxy-5,5-dimethyl-1,3-thiazolidin-2-yl]-2-[(2R)-2-(4-ethyl-2,3- dioxopiperazine-1-carboxamido)-2-phenylacetamido]acetamido]-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1- azabicyclo[3.2.0]heptane-2-carboxylic acid,
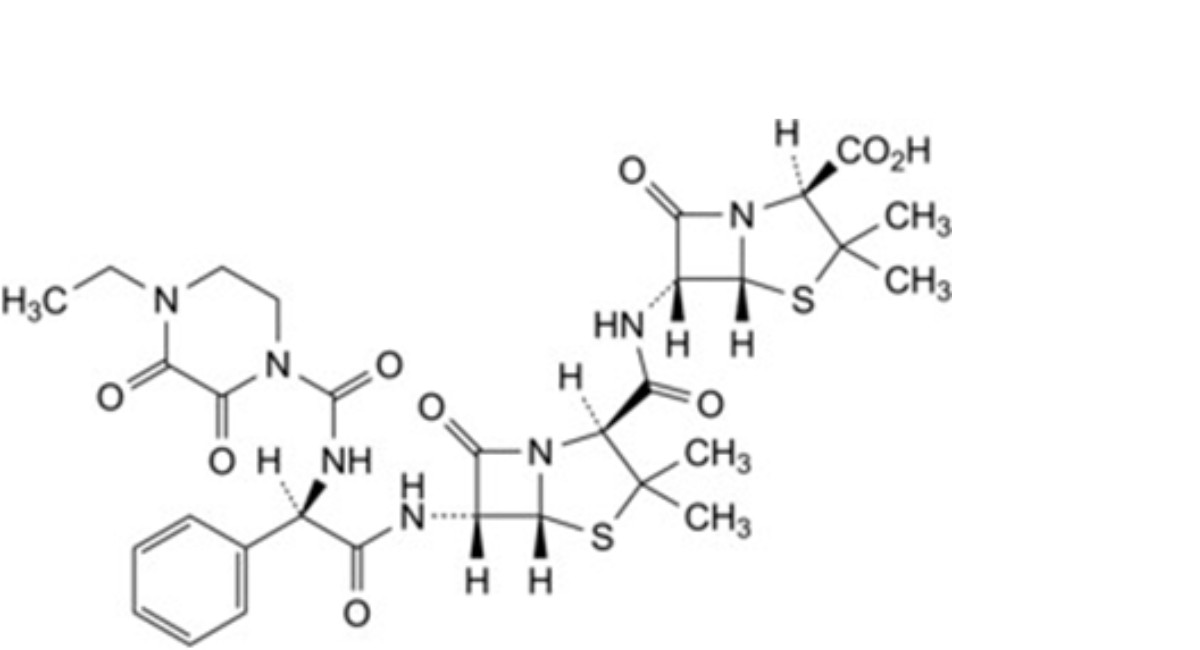
S. (2S,5R,6R)-6-[(2S,5R,6R)-6-[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7- oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid,
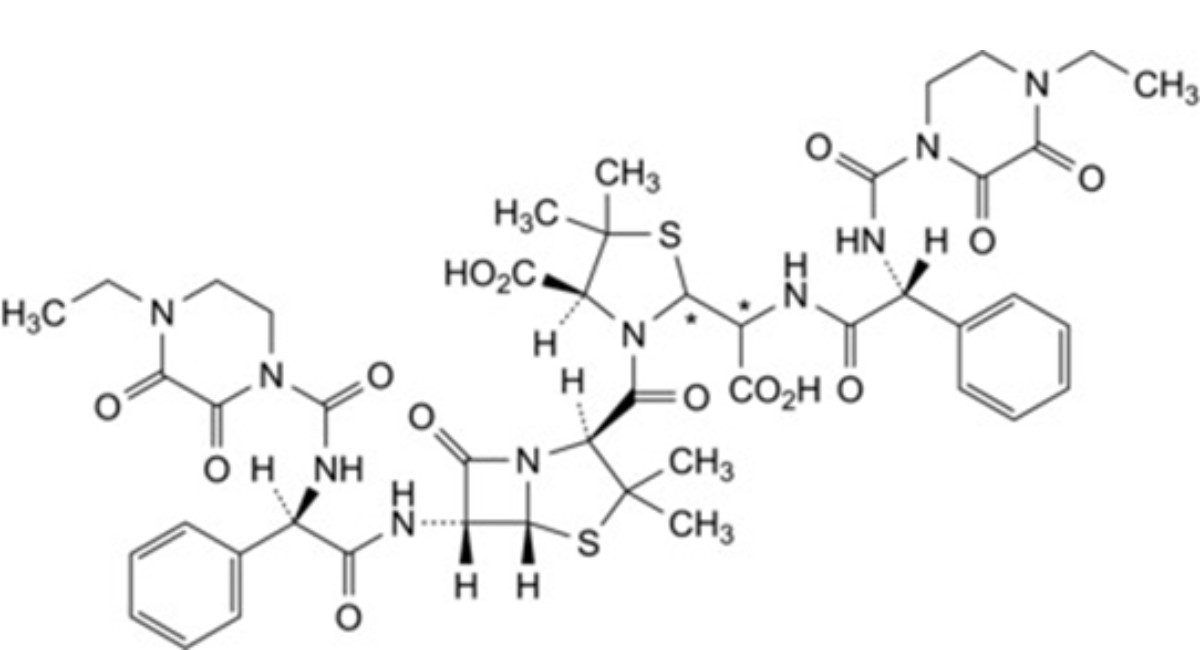
T. (2Ξ,4S)-2-[(Ξ)-carboxy[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]methyl]-3- [(2S,5R,6R)-6-[(2R)-2-(4-ethyl-2,3-dioxopiperazine-1-carboxamido)-2-phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carbonyl]-5,5-dimethyl-1,3-thiazolidine-4-carboxylic acid.
Ph Eur