(Ph. Eur. monograph 0632)
C9H14ClNO2 203.7 61-76-7
Action and use
Alpha-adrenoceptor agonist.
Preparations
Phenylephrine Eye Drops
Phenylephrine Injection
Phenylephrine Intracameral Injection
DEFINITION
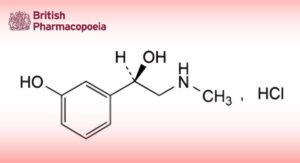
(1R)-1-(3-Hydroxyphenyl)-2-(methylamino)ethanol hydrochloride.
Content
98.5 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Freely soluble in water and in ethanol (96 per cent).
mp
About 143 °C.
IDENTIFICATION
First identification: A, C, E.
Second identification: A, B, D, E.
A. Specific optical rotation (see Tests).
B. Melting point (2.2.14): 171 °C to 176 °C.
Dissolve 0.3 g in 3 mL of water R, add 1 mL of dilute ammonia R1 and initiate crystallisation by scratching the wall of the tube with a glass rod. Wash the crystals with iced water R and dry at 105 °C for 2 h.
C. Infrared absorption spectrophotometry (2.2.24).
Preparation: Discs.
Comparison: phenylephrine hydrochloride CRS.
D. Dissolve about 10 mg in 1 mL of water R and add 0.05 mL of a 125 g/L solution of copper sulfate pentahydrate R and 1 mL of a 200 g/L solution of sodium hydroxide R. A violet colour is produced. Add 1 mL of ether R and shake; the upper layer remains colourless.
E. It gives reaction (a) of chlorides (2.3.1).
TESTS
Solution S
Dissolve 2.00 g in carbon dioxide-free water R prepared from distilled water R and dilute to 100.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and colourless (2.2.2, Method II).
Acidity or alkalinity
To 10 mL of solution S add 0.1 mL of methyl red solution R and 0.2 mL of 0.01 M sodium hydroxide. The solution is yellow.
Not more than 0.4 mL of 0.01 M hydrochloric acid is required to change the colour of the indicator to red.
Specific optical rotation (2.2.7)
-43 to -47 (dried substance), determined on solution S.
Related substances
Liquid chromatography (2.2.29).
Solvent mixture Mobile phase B, mobile phase A (20:80 V/V).
Buffer solution: pH 2.8 Dissolve 3.25 g of sodium octanesulfonate monohydrate R in 1000 mL of water for chromatography R by stirring for 30 min and adjust to pH 2.8 with dilute phosphoric acid R.
Test solution: Dissolve 50.0 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a): Dilute 5.0 mL of the test solution to 100.0 mL with the solvent mixture. Dilute 2.0 mL of this solution to 100.0 mL with the solvent mixture.
Reference solution (b): Dissolve the contents of a vial of phenylephrine hydrochloride for peak identification CRS (containing impurities C and E) in 2 mL of the solvent mixture.
Column:
— size: l = 0.055 m, Ø = 4.0 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R1 (3 μm);
— temperature: 45 °C.
Mobile phase:
— mobile phase A: acetonitrile R1, buffer solution pH 2.8 (10:90 V/V);
— mobile phase B: buffer solution pH 2.8, acetonitrile R1 (10:90 V/V);
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 3 | 93 | 7 |
| 3 – 13 | 93 → 70 | 7 → 30 |
| 13 – 14 | 70 → 93 | 30 → 7 |
Flow rate: 1.5 mL/min.
Detection: Spectrophotometer at 215 nm.
Injection: 10 μL.
Identification of impurities: Identify the peaks due to impurities C and E using the chromatogram obtained with reference solution (b).
Relative retention: With reference to phenylephrine (retention time = about 2.8 min): impurity C = about 1.3; impurity E = about 3.6.
System suitability:
— symmetry factor: maximum 1.9 for the principal peak in the chromatogram obtained with the test solution;
— peak-to-valley ratio: minimum 5.0, where Hp = height above the baseline of the peak due to impurity C and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to
phenylephrine in the chromatogram obtained with reference solution (b).
Limits:
— correction factors: for the calculation of content, multiply the peak areas of the following impurities by the corresponding correction factor: impurity C = 0.5; impurity E = 0.5;
— impurities C, E: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
— total: not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Sulfates (2.4.13)
Maximum 500 ppm, determined on solution S.
Loss on drying (2.2.32)
Maximum 1.0 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.150 g in a mixture of 0.5 mL of 0.1 M hydrochloric acid and 80 mL of ethanol (96 per cent) R. Carry out a potentiometric titration (2.2.20) using 0.1 M ethanolic sodium hydroxide. Read the volume added between the 2 points of inflexion.
1 mL of 0.1 M ethanolic sodium hydroxide is equivalent to 20.37 mg of C9H14ClNO2.
IMPURITIES
Specified impurities C, E.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, D.
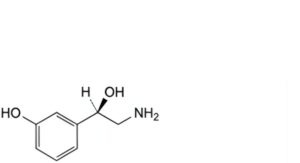
A. (1R)-2-amino-1-(3-hydroxyphenyl)ethanol (norphenylephrine),
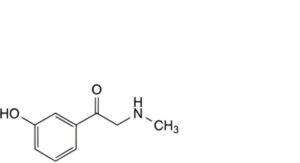
C. 1-(3-hydroxyphenyl)-2-(methylamino)ethanone (phenylephrone),
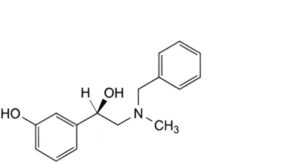
D. (1R)-2-(benzylmethylamino)-1-(3-hydroxyphenyl)ethanol (benzylphenylephrine),
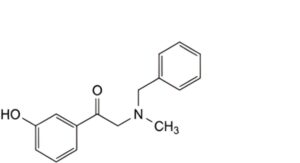
E. 2-(benzylmethylamino)-1-(3-hydroxyphenyl)ethanone (benzylphenylephrone).





