(Ph. Eur. monograph 1584)
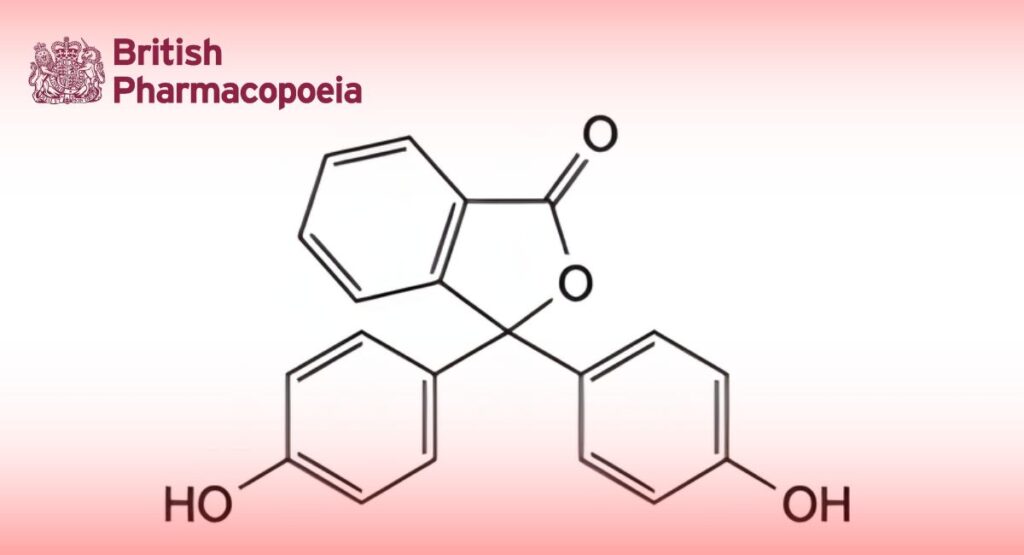
C20H14O4 318.3 77-09-8
Action and use
Stimulant laxative.
DEFINITION
Phenolphthalein contains not less than 98.0 per cent and not more than the equivalent of 101.0 per cent of 3,3-bis(4-hydroxyphenyl)isobenzofuran-1(3H)-one, calculated with reference to the dried substance.
CHARACTERS
A white or almost white powder, practically insoluble in water, soluble in ethanol (96 per cent).
It melts at about 260 °C.
IDENTIFICATION
A. Dissolve 25.0 mg in alcohol R and dilute to 100.0 mL with the same solvent (solution A). To 2.0 mL of solution A add 5.0 mL of 1 M hydrochloric acidand dilute to 50.0 mL with alcohol R (solution A1). To 10.0 mL of solution A add 5.0 mL of 1 M hydrochloric acid and dilute to 50.0 mL with alcohol R (solution A2). To 2.0 mL of solution A add 5.0 mL of 1 M sodium hydroxide and dilute to 50.0 mL with alcohol R (solution B). Examined between 220 nm and 250 nm (2.2.25), solution A1 shows an absorption maximum at 229 nm. The specific absorbance at the maximum at 229 nm is 922 to 1018. Examined between 250 nm and 300 nm, solution A2 shows an absorption maximum at 276 nm. The specific absorbance at the maximum at 276 nm is 142 to 158. Examined between 230 nm and 270 nm, solution B shows an absorption maximum at 249 nm. The specific absorbance at the maximum at 249 nm is 744 to 822.
B. Dissolve about 10 mg in alcohol R. Add 1 mL of dilute sodium hydroxide solution R. The solution is red. Add 5 mL of dilute sulfuric acid R. The colour disappears.
TESTS
Solution S
To 2.0 g add 40 mL of distilled water R and heat to boiling. Cool and filter.
Appearance of solution
Dissolve 0.20 g in 5 mL of alcohol R. The solution is clear (2.2.1) and not more intensely coloured than reference solution Y7 (2.2.2, Method II).
Acidity or alkalinity
To 10 mL of solution S add 0.15 mL of bromothymol blue solution R1. Add 0.05 mL of 0.01 M hydrochloric acid, the solution is yellow. Add 0.10 mL of 0.01 M sodium hydroxide, the solution is blue.
Related substances
Examine by thin-layer chromatography (2.2.27), using a TLC silica gel F254 plate R.
Test solution: Dissolve 0.5 g of the substance to be examined in alcohol R and dilute to 10 mL with the same solvent.
Reference solution (a): Dilute 1 mL of the test solution to 10 mL with alcohol R. Dilute 5 mL of this solution to 100 mL with alcohol R.
Reference solution (b): Dissolve 25 mg of fluorene R in alcohol R, add 0.5 mL of the test solution and dilute to 10 mL with alcohol R.
Apply to the plate 5 μL of the test solution and 5 μL of each of the reference solutions. Develop over a path corresponding to two-thirds of the plate height using a mixture of 50 volumes of acetone R and 50 volumes of methylene chloride R. Allow the plate to dry in air. Examine in ultraviolet light at 254 nm and re-examine after exposure to ammonia vapour. Any spot in the chromatogram obtained with the test solution, apart from the principal spot, is not more intense than the spot in the chromatogram obtained with reference solution (a) (0.5 per cent). The test is not valid unless the chromatogram obtained with reference solution (b) shows 2 clearly separated spots.
Chlorides (2.4.4)
Dilute 10 mL of solution S to 15 mL with water R. The solution complies with the limit test for chlorides (100 ppm).Sulfates (2.4.13) 15 mL of solution S complies with the limit test for sulfates (200 ppm).
Loss on drying (2.2.32)
Not more than 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Sulfated ash (2.4.14)
Not more than 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.100 g in 5 mL of dimethylformamide R. Add 5 mL of sodium carbonate solution R, 10 mL of sodium hydrogen carbonate solution R, 35 mL of water R and 50.0 mL of 0.05 M iodine. Add 10 mL of methylene chloride R and 20 mL of dilute sulfuric acid R. Titrate the excess of iodine with 0.1 M sodium thiosulfate, using 0.3 mL of starch solution R added towards the end of the titration, as indicator. Carry out a blank titration.
1 mL of 0.05 M iodine is equivalent to 3.979 mg of C20H14O4.
STORAGE
Store protected from light.