(Ph. Eur. monograph 0517)
C47H75NO17 926
Action and use
Antifungal.
Preparations
Nystatin Ointment
Nystatin Oral Suspension
Nystatin Vaginal Tablets
Nystatin Tablets
DEFINITION
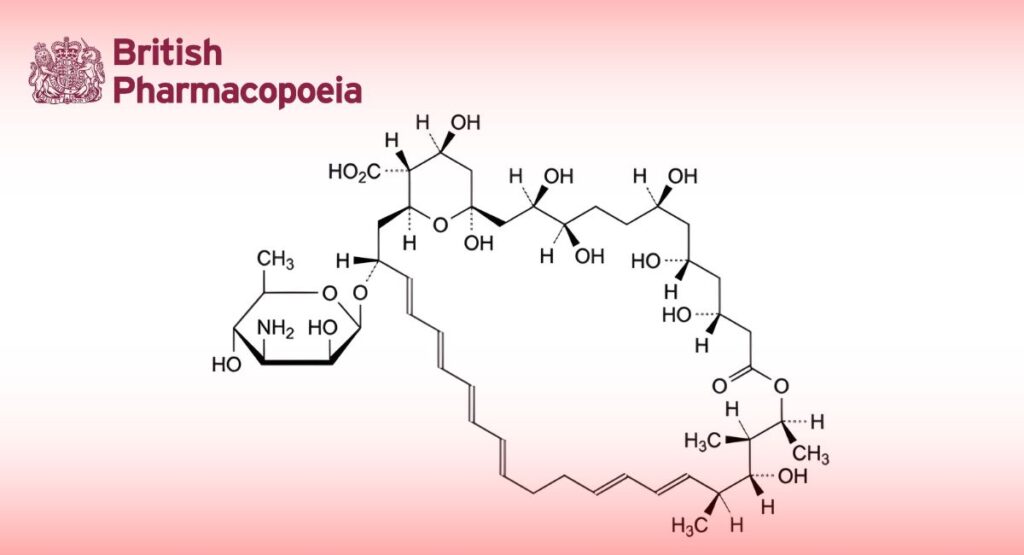
Antifungal substance obtained by fermentation using certain strains of Streptomyces noursei as the production micro-organism. It contains mainly tetraenes, the principal component being (1S, 3R, 4R, 7R, 9R, 11R, 15S, 16R, 17R, 18S, 19E, 21E, 25E, 27E, 29E, 31E, 33R, 35S, 36R, 37S)-33-[(3-amino-3,6-Nystatin dideoxy-β-D-mannopyranosyl)oxy]-1,3,4,7,9,11,17,37-octahydroxy-15,16,18-trimethyl-13-oxo-14,39-
dioxabicyclo[33.3.1]nonatriaconta-19,21,25,27,29,31-hexaene-36-carboxylic acid (nystatin A1).
Content
Minimum 4400 IU/mg (dried substance) and minimum 5000 IU/mg (dried substance) if intended for oral administration.
CHARACTERS
Appearance
Yellow or slightly brownish powder, hygroscopic.
Solubility
Practically insoluble in water, freely soluble in dimethylformamide and in dimethyl sulfoxide, slightly soluble in methanol, practically insoluble in ethanol (96 per cent).
IDENTIFICATION
First identification: B, E.
Second identification: A, C, D.
A. Examine the solution prepared in the test for absorbance between 220 nm and 350 nm (2.2.25). The solution shows 4 absorption maxima at 230 nm, 291 nm, 305 nm and 319 nm, and a shoulder at 280 nm.
The ratios of the absorbances at the absorption maxima at 291 nm and 319 nm to the absorbance at the absorption maximum at 305 nm are 0.61 to 0.73 and 0.83 to 0.96, respectively. The ratio of the absorbance measured at the absorption maximum at 230 nm to that measured at the shoulder at 280 nm is 0.83 to 1.25.
B. Infrared absorption spectrophotometry (2.2.24).
Comparison: nystatin CRS.
C. To about 2 mg add 0.1 mL of hydrochloric acid R. A brown colour develops.
D. To about 2 mg add 0.1 mL of sulfuric acid R. A brown colour develops that becomes violet on standing.
E. Examine the chromatograms obtained in the test for composition.
Results: The principal peak in the chromatogram obtained with the test solution is similar in retention time to the principal peak in the chromatogram obtained with reference solution (a).
TESTS
Absorbance (2.2.25)
Dissolve 0.10 g in a mixture of 5.0 mL of glacial acetic acid R and 50 mL of methanol R and dilute to 100.0 mL with methanol R. Dilute 1.0 mL of the solution to 100.0 mL with methanol R. Determined at the maximum at 305 nm within 30 min of preparation of the solution, the absorbance is not less than 0.60.
Composition
Liquid chromatography (2.2.29): use the normalisation procedure. Carry out the test protected from light. Test solution Dissolve 20 mg of the substance to be examined in dimethyl sulfoxide R and dilute to 50 mL
with the same solvent.
Reference solution (a): Dissolve 20 mg of nystatin CRS in dimethyl sulfoxide R and dilute to 50 mL with the same solvent.
Reference solution (b): Dissolve 20 mg of the substance to be examined in 25 mL of methanol R and dilute to 50 mL with water R. To 10.0 mL of the solution add 2.0 mL of dilute hydrochloric acid R. Allow to stand at
room temperature for 1 h.
Reference solution (c): Dilute 1.0 mL of reference solution (a) to 100.0 mL with dimethyl sulfoxide R. Dilute 1.0 mL of this solution to 10.0 mL with dimethyl sulfoxide R.
Column:
— size: l = 0.15 m, Ø = 4.6 mm,
— stationary phase: base-deactivated end-capped octadecylsilyl silica gel for chromatography R (5 μm),
— temperature: 30 °C.
Mobile phase:
— mobile phase A: acetonitrile R, 3.85 g/L solution of ammonium acetate R (29:71 V/V),
— mobile phase B: 3.85 g/L solution of ammonium acetate R, acetonitrile R (40:60 V/V)
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 25 | 100 | 0 |
| 25 – 35 | 100 → 0 | 0 → 100 |
| 35 – 45 | 0 | 100 |
| 45 – 50 | 0 → 100 | 100 → 0 |
Flow rate: 1.0 mL/min.
Detection: Spectrophotometer at 305 nm.
Injection: 20 μL
Retention time: Nystatin A1 = about 14 min.
System suitability: Reference solution (b):
— resolution: minimum 3.5 between the 2 principal peaks (retention time = about 13 min and 19 min).
Composition:
— nystatin A1: minimum 85.0 per cent,
— any other compound: maximum 4.0 per cent,
— disregard limit: the area of the principal peak in the chromatogram obtained with reference solution (c); disregard any peak with a retention time of less than 2 min.
Loss on drying (2.2.32)
Maximum 5.0 per cent, determined on 1.000 g by drying in vacuo at 60 °C at a pressure not exceeding 0.1 kPa for 3 h.
Sulfated ash (2.4.14)
Maximum 3.5 per cent, determined on 1.0 g.
ASSAY
Carry out the microbiological assay of antibiotics (2.7.2). Protect the solutions from light throughout the assay.
Dissolve the substance to be examined and nystatin CRS separately in dimethylformamide R and dilute with a mixture of 5 volumes of dimethylformamide R and 95 volumes of buffer solution pH 6.0.
STORAGE
In an airtight container, protected from light.
LABELLING
The label states where applicable, that the substance is only for cutaneous use.