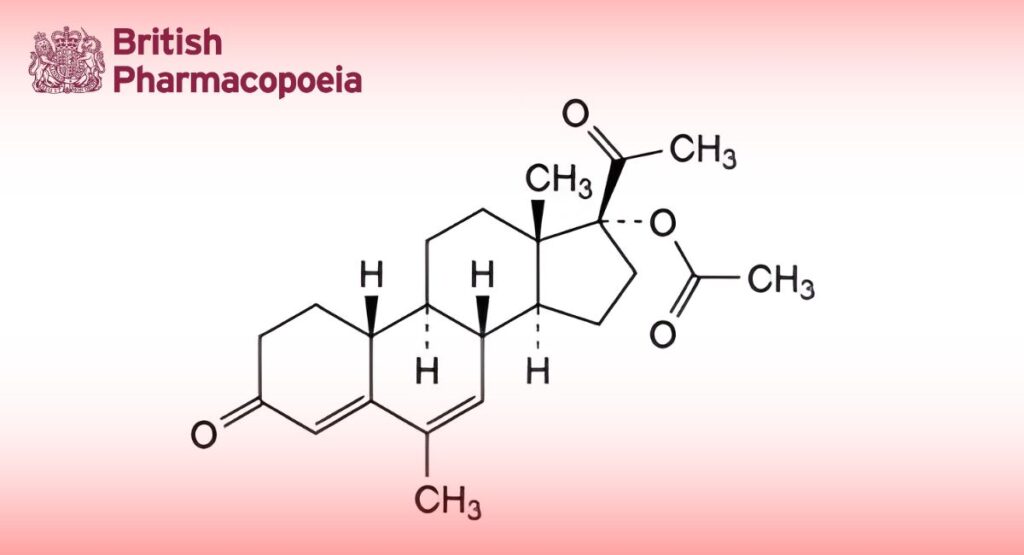
(Ph. Eur. monograph 1551)
C23H30O4 370.5 58652-20-3
Action and use
Progestogen.
DEFINITION
6-Methyl-3,20-dioxo-19-norpregna-4,6-dien-17-yl acetate.
Content
98.0 per cent to 102.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Practically insoluble in water, freely soluble in acetone, soluble in ethanol (96 per cent), practically insoluble in heptane.
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Comparison: nomegestrol acetate for ID and assay CRS.
TESTS
Appearance of solution
The solution is clear (2.2.1) and not more intensely coloured than reference solution Y5 (2.2.2, Method II).
Dissolve 1.0 g in methylene chloride R and dilute to 10 mL with the same solvent.
Specific optical rotation (2.2.7)
-64.0 to -60.0 (dried substance).
Dissolve 0.500 g in anhydrous ethanol R and dilute to 25.0 mL with the same solvent.
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 25.0 mg of the substance to be examined in methanol R and dilute to 50.0 mL with the same solvent.
Reference solution (a): Dissolve 25.0 mg of nomegestrol acetate for ID and assay CRS in methanol R and dilute to 50.0 mL with the same solvent.
Reference solution (b): Dilute 1.0 mL of the test solution to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (c): Dissolve 25.0 mg of nomegestrol acetate impurity A CRS in methanol R and dilute to 100.0 mL with the same solvent. Dilute 0.25 mL of the solution to 25.0 mL with reference solution (a).
Reference solution (d): Dissolve the contents of a vial of nomegestrol acetate impurity B CRS in 1 mL of methanol R.
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R (5 μm).
Mobile phase” acetonitrile for chromatography R, methanol R1, water for chromatography R (24:38:38 V/V/V).
Flow rate: 1.3 mL/min.
Detection: Spectrophotometer at 245 nm and at 290 nm.
Injection: 10 μL of the test solution and reference solutions (b), (c), (d).
Run time: 1.5 times the retention time of nomegestrol acetate.
Identification of impurities: Use the chromatogram obtained with reference solution (c) at 245 nm to identify the peak due to impurity A; use the chromatogram obtained with reference solution (d) at 290 nm to identify the peak due to impurity B.
Relative retention: With reference to nomegestrol acetate (retention time = about 17 min): impurity B = about 0.6; impurity A = about 1.1.
System suitability: Reference solution (c) at 245 nm:
— peak-to-valley ratio: minimum 5, where Hp = height above the baseline of the peak due to impurity A and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to nomegestrol acetate.
Calculation of percentage contents:
— for each impurity at 245 nm, use the concentration of impurity A in reference solution (c);
— for each impurity at 290 nm, use the concentration of nomegestrol acetate in reference solution (b).
Limits:
— impurity A at 245 nm: maximum 0.2 per cent;
— impurity B at 290 nm: maximum 0.15 per cent;
— unspecified impurities at 245 nm and at 290 nm: for each impurity, maximum 0.10 per cent;
— sum of impurities other than A at 245 nm and 290 nm: maximum 0.3 per cent;
— reporting threshold at 245 nm and at 290 nm: 0.05 per cent.
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications:
Detection: Spectrophotometer at 290 nm.
Injection: Test solution and reference solution (a).
Calculate the percentage content of C23H30O4 taking into account the assigned content of nomegestrol acetate for ID and assay CRS.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B.
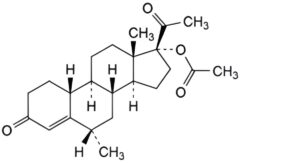
A. 6α-methyl-3,20-dioxo-19-norpregn-4-en-17-yl acetate,
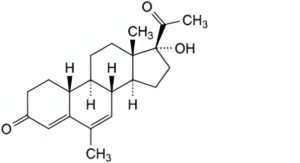
B. 17-hydroxy-6-methyl-19-norpregna-4,6-diene-3,20-dione (nomegestrol).