BP 2025 (Ph. Eur. 11.6 update)
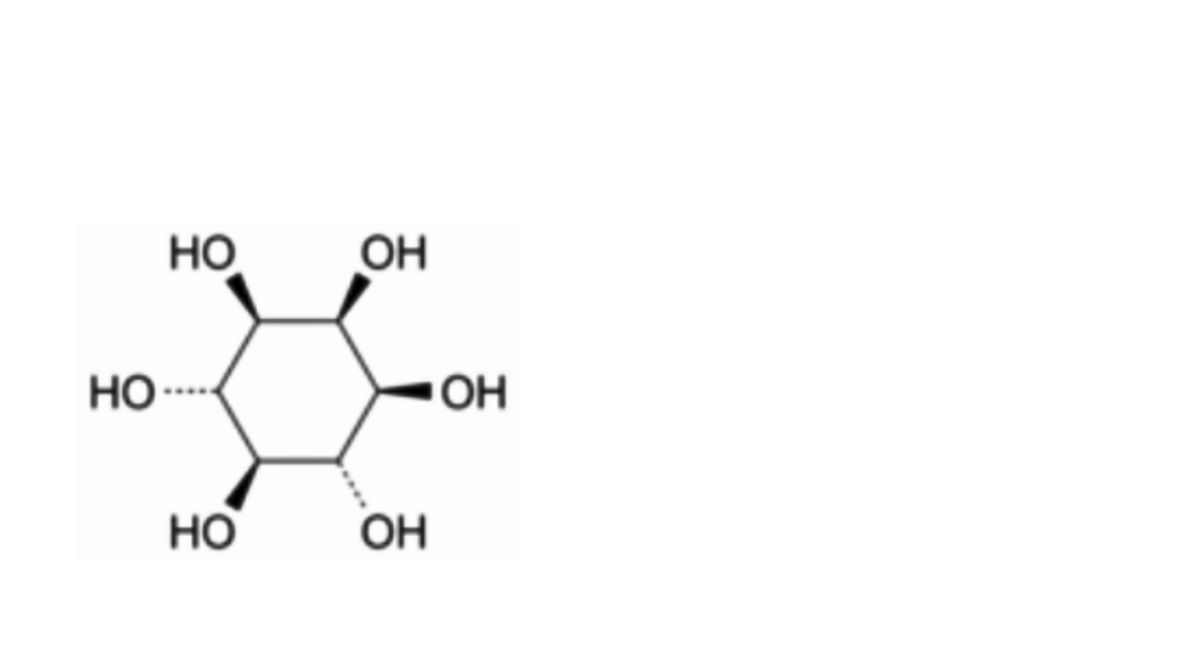
C6H12O6 180.2 87-89-8
Action and use
Vasodilator.
DEFINITION
Cyclohexane-1,2,3,5/4,6-hexol.
Content
97.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Very soluble in water, practically insoluble in ethanol (96 per cent).
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Comparison myo-inositol CRS.
B. Examine the chromatograms obtained in the assay.
Results The principal peak in the chromatogram obtained with the test solution is similar in retention time and size to the principal peak in the chromatogram obtained with reference solution (a).
TESTS
Solution S
Dissolve 10.0 g in distilled water R and dilute to 100.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and colourless (2.2.2, Method II).
Conductivity (2.2.38)
Maximum 30 μS·cm .
Dissolve 10.0 g in carbon dioxide-free water R prepared from distilled water R, with gentle warming if necessary, and dilute to 50.0 mL with the same solvent. Measure the conductivity of the solution while gently stirring with a magnetic stirrer.
Related substances
Liquid chromatography (2.2.29).
Test solution Dissolve 0.500 g of the substance to be examined in water R and dilute to 10.0 mL with the same solvent.
Reference solution (a) Dissolve 0.500 g of myo-inositol CRS in water R and dilute to 10.0 mL with the same solvent.
Reference solution (b) Dilute 2.0 mL of the test solution to 100.0 mL with water R. Dilute 5.0 mL of this solution to 100.0 mL with water R.
Reference solution (c) Dissolve 0.5 g of myo-inositol R, 2 mg of mannitol R (impurity A) and 2 mg of glycerol R (impurity B) in water R and dilute to 10 mL with the same solvent.
Column:
— size: l = 0.3 m, Ø = 7.8 mm;
— stationary phase: strong cation-exchange resin (calcium form) R (9 μm);
— temperature: 85 ± 2 °C.
Mobile phase water for chromatography R.
Flow rate 0.5 mL/min.
Detection Differential refractometer maintained at a constant temperature (e.g. 35 °C).
Injection 20 μL of the test solution and reference solutions (b) and (c).
Run time Twice the retention time of myo-inositol.
Relative retention With reference to myo-inositol (retention time = about 16 min): impurity B = about 1.2; impurity A = about 1.3.
System suitability Reference solution (c):
— resolution: minimum 1.5 between the peaks due to impurity B and impurity A.
Limits:
— impurities A, B: for each impurity, not more than 3 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.3 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.10 per cent);
— total: not more than 10 times the area of the principal peak in the chromatogram obtained with reference solution (b) (1.0 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Water (2.5.12)
Maximum 0.5 per cent, determined on 1.00 g.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modification.
Injection Test solution and reference solution (a).
Calculate the percentage content of C6H12O6 taking into account the assigned content of myo-inositol CRS.
IMPURITIES
Specified impurities A, B.
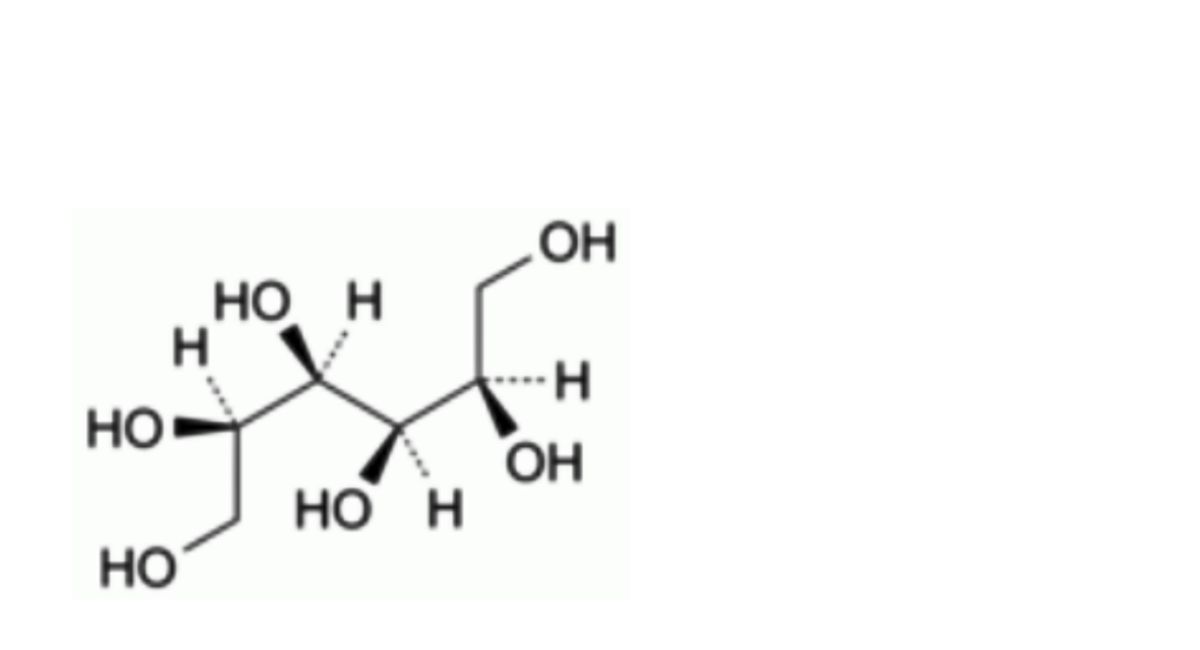
A. D-mannitol,
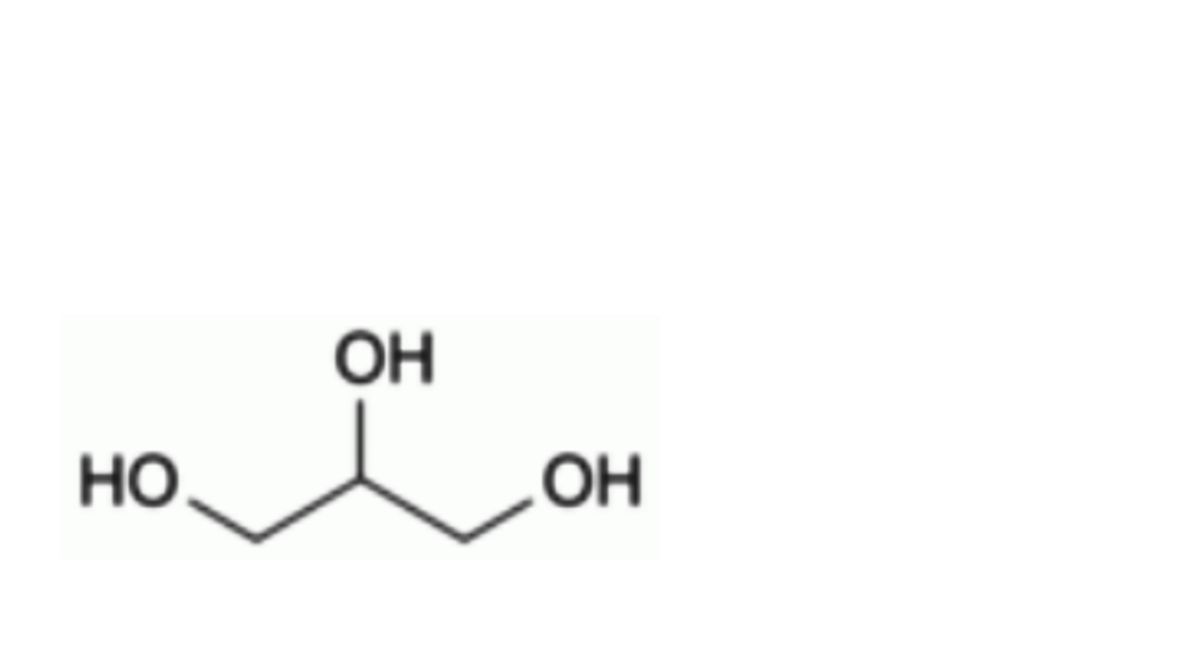
B. propane-1,2,3-triol (glycerol).