Edition: BP 2025 (Ph. Eur. 11.6 update)
(Ph. Eur. monograph 1731)
C22H38O5 382.5 59122-46-2
Action and use
Prostaglandin (PGE1) analogue.
Ph Eur
DEFINITION
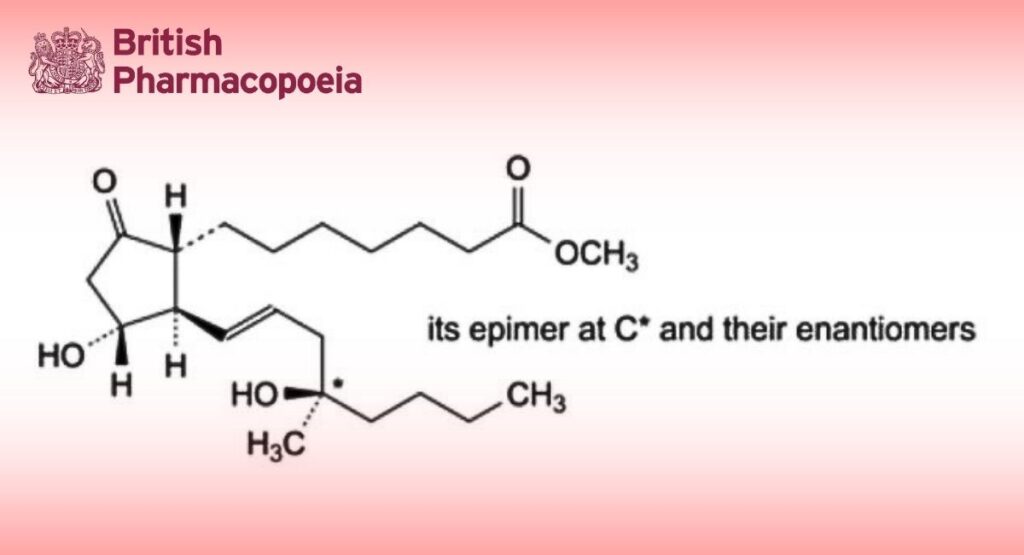
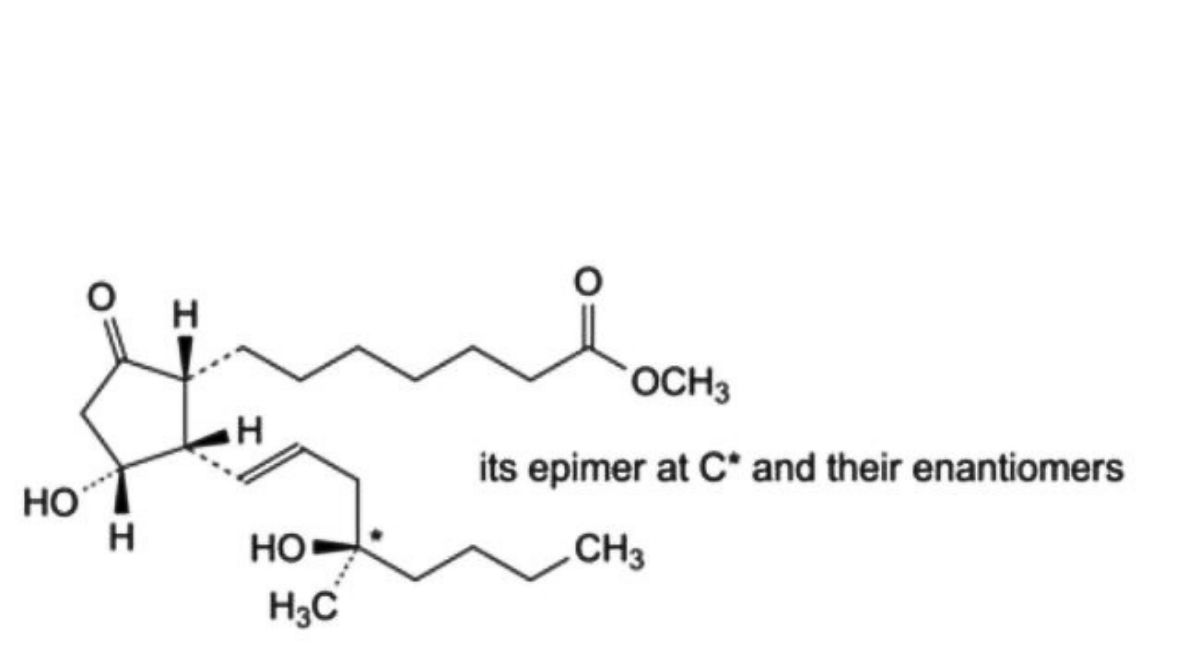
Mixture of methyl 7-[(1RS,2RS,3RS)-3-hydroxy-2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5- oxocyclopentyl]heptanoate and methyl 7-[(1RS,2RS,3RS)-3-hydroxy-2-[(1E,4SR)-4-hydroxy-4-methyloct-1- enyl]-5-oxocyclopentyl]heptanoate.
The 4 stereoisomers are present in approximately equal proportions.
Content
96.5 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
Clear, colourless or yellowish, oily liquid, hygroscopic.
Solubility
Practically insoluble in water, soluble in ethanol (96 per cent), sparingly soluble in acetonitrile.
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Comparison misoprostol CRS.
TESTS
Related substances
Liquid chromatography (2.2.29). Use freshly prepared solutions.
Test solution Dissolve 25.0 mg of the substance to be examined in the mobile phase and dilute to 5.0 mL with the mobile phase.
Reference solution (a) Dissolve 25.0 mg of misoprostol CRS in the mobile phase and dilute to 5.0 mL with the mobile phase.
Reference solution (b) Dilute 1.0 mL of reference solution (a) to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (c) Dissolve 5 mg of misoprostol for system suitability CRS (containing impurities A, B and C) in 1 mL of the mobile phase.
Column:
- size: l = 0.25 m, Ø = 4.6 mm;
- stationary phase: silica gel for chromatography R (5 µm).
Mobile phase Mix 5 volumes of acetonitrile R1, 215 volumes of dioxan R, 780 volumes of heptane R and sonicate for 10 min. Flow rate 2.0 mL/min.
Detection Spectrophotometer at 210 nm.
Injection 20 µL of the test solution and reference solutions (b) and (c).
Run time 1.5 times the retention time of misoprostol.
Identification of impurities Use the chromatogram supplied with misoprostol for system suitability CRS and the chromatogram obtained with reference solution (c) to identify the peaks due to impurities A, B and C.
Relative retention With reference to misoprostol (retention time = about 18 min): impurity C = about 0.2; st nd impurity A = about 0.7; impurity B (1 peak) = about 0.85; impurity B (2 peak) = about 0.91.
System suitability Reference solution (c):
- resolution: minimum 1.2 between the peaks due to impurity B (2nd peak) and misoprostol.
Limits:
- correction factor: for the calculation of content, multiply the peak area of impurity C by 0.13; st nd
- impurity B (sum of 1 and 2 peaks): not more than 10 times the area of the principal peak in the chromatogram obtained with reference solution (b) (1.0 per cent);
- impurity A: not more than 3 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.3 per cent);
- impurity C: not more than 1.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.15 per cent);
- unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.10 per cent);
- total: not more than 15 times the area of the principal peak in the chromatogram obtained with reference solution (b) (1.5 per cent);
- disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Diastereoisomers
Liquid chromatography (2.2.29). Use freshly prepared solutions.
Test solution Dissolve 10.0 mg of the substance to be examined in the mobile phase and dilute to 10.0 mL with the mobile phase.
Column:
- size: l = 0.25 m, Ø = 4.6 mm;
- stationary phase: silica gel for chromatography R (5 µm);
- temperature: 40 °C.
Mobile phase Mix 20 volumes of 2-propanol R, 40 volumes of anhydrous ethanol R, 940 volumes of heptane R and sonicate for 10 min.
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 205 nm.
Injection 20 µL.
st Run time 1.5 times the retention time of the 1 peak of misoprostol.
st nd Retention time Misoprostol 1 peak = about 19 min; misoprostol 2 peak = about 21 min.
System suitability Test solution:
- st nd resolution: minimum 2.0 between the 1 and 2 peaks of misoprostol.
Limit:
- st 1 peak of misoprostol: 45 per cent to 55 per cent of the sum of the areas of the 2 peaks due to misoprostol.
Water (2.5.32)
Maximum 1.0 per cent.
Use 1.0 mL of a 10 mg/mL solution of the substance to be examined in methanol R.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection 20 µL of the test solution and reference solution (a).
System suitability Reference solution (a):
- symmetry factor: maximum 3.7 for the peak due to misoprostol.
Calculate the percentage content of C22H38O5 using the declared content of misoprostol CRS.
STORAGE
In an airtight container, at -20 °C.
IMPURITIES
Specified impurities A, B, C.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) D, E, F.
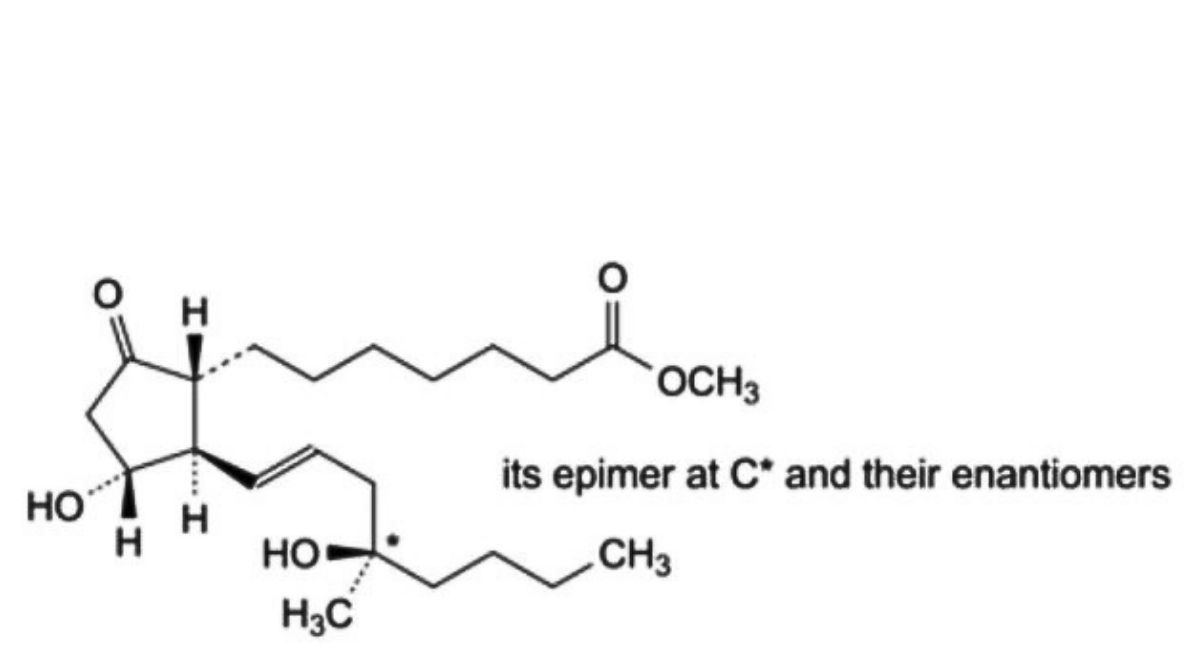
A. mixture of methyl 7-[(1RS,2SR,3SR)-3-hydroxy-2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5- oxocyclopentyl]heptanoate and methyl 7-[(1RS,2SR,3SR)-3-hydroxy-2-[(1E,4SR)-4-hydroxy-4-methyloct-1- enyl]-5-oxocyclopentyl]heptanoate (8-epimisoprostol),
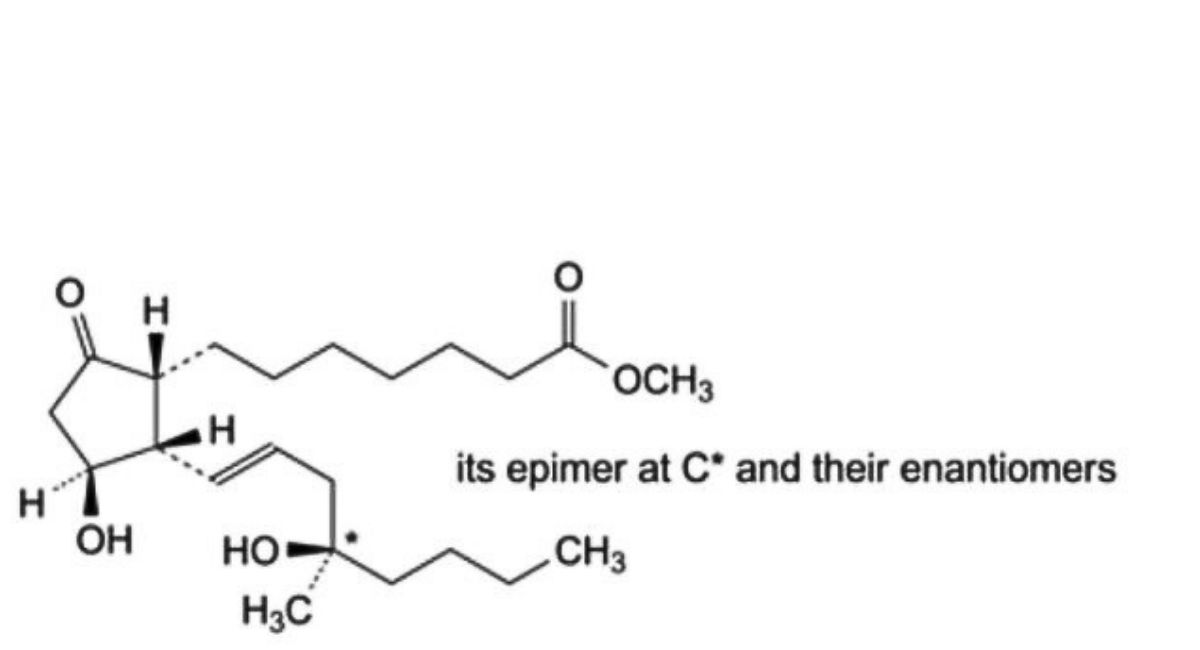
B. mixture of methyl 7-[(1RS,2SR,3RS)-3-hydroxy-2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5- oxocyclopentyl]heptanoate and methyl 7-[(1RS,2SR,3RS)-3-hydroxy-2-[(1E,4SR)-4-hydroxy-4-methyloct-1- enyl]-5-oxocyclopentyl]heptanoate (12 epimisoprostol),
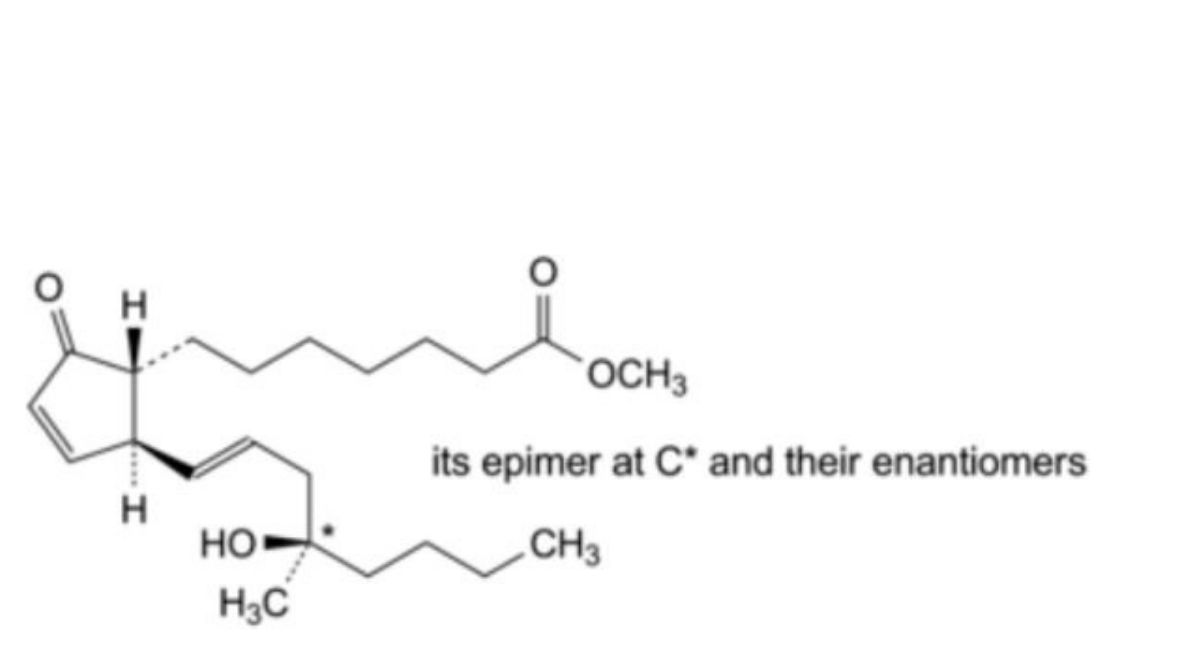
C. mixture of methyl 7-[(1RS,2SR)-2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5-oxocyclopent-3- enyl]heptanoate and methyl 7-[(1RS,2SR)-2-[(1E,4SR)-4-hydroxy-4-methyloct-1-enyl]-5-oxocyclopent-3- enyl]heptanoate (misoprostol A),
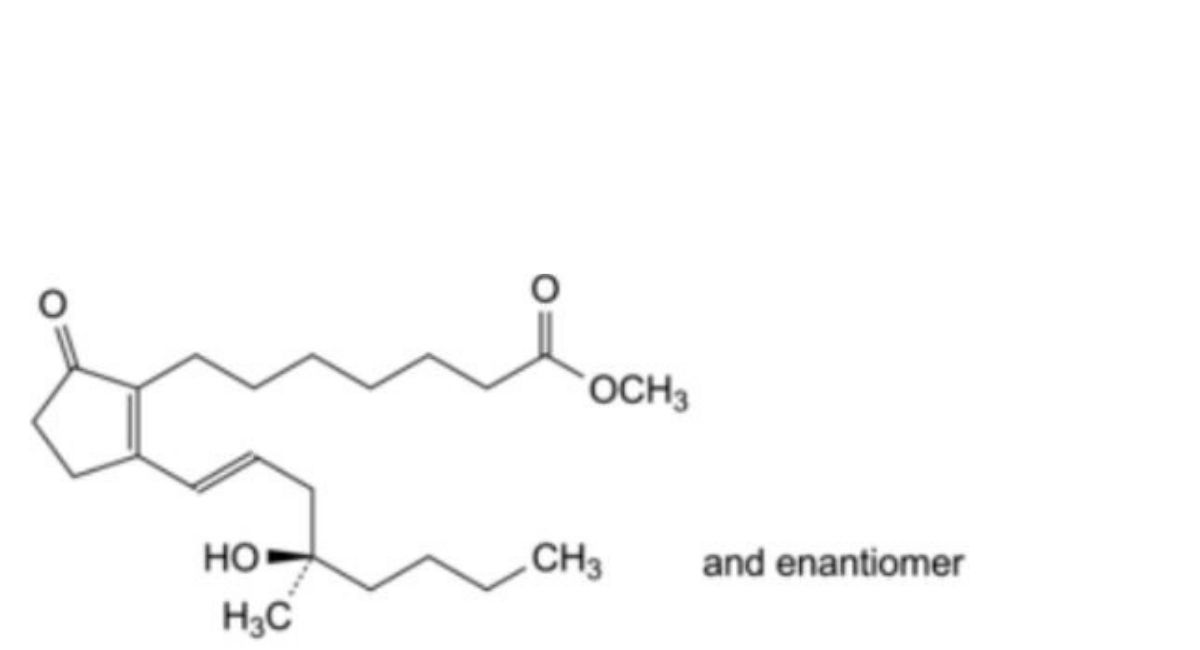
D. methyl 7-[2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5-oxocyclopent-1-enyl]heptanoate (misoprostol B),
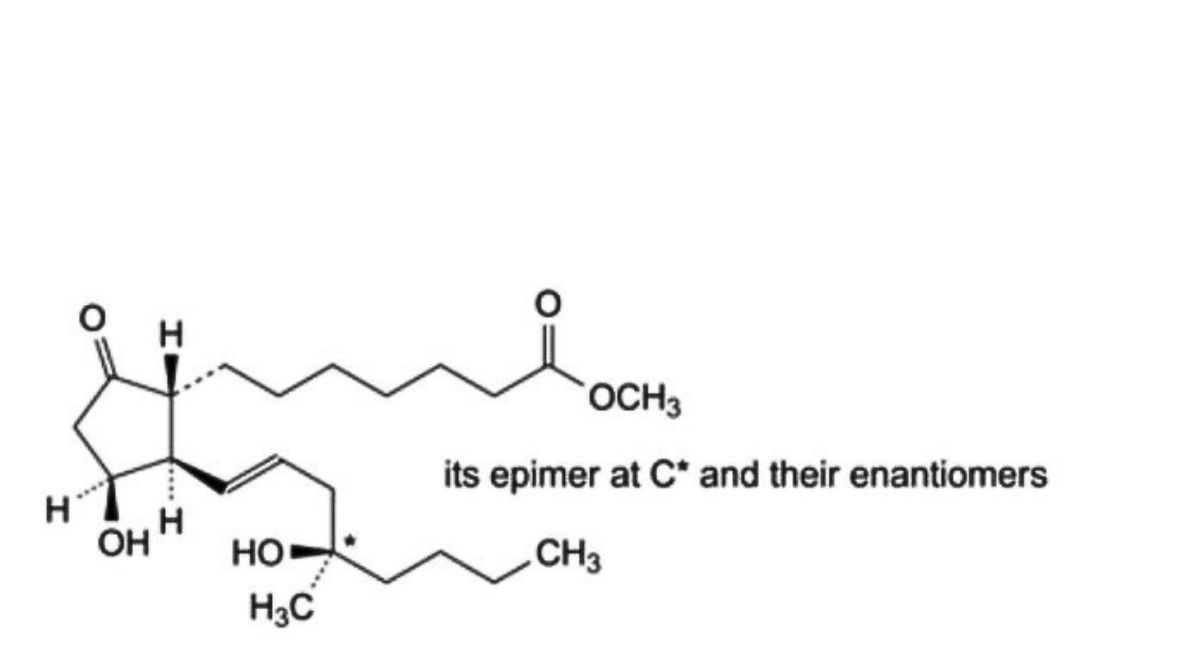
E. mixture of methyl 7-[(1RS,2RS,3SR)-3-hydroxy-2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5- oxocyclopentyl]heptanoate and methyl 7-[(1RS,2RS,3SR)-3-hydroxy-2-[(1E,4SR)-4-hydroxy-4-methyloct-1- enyl]-5-oxocyclopentyl]heptanoate (11-epi misoprostol),
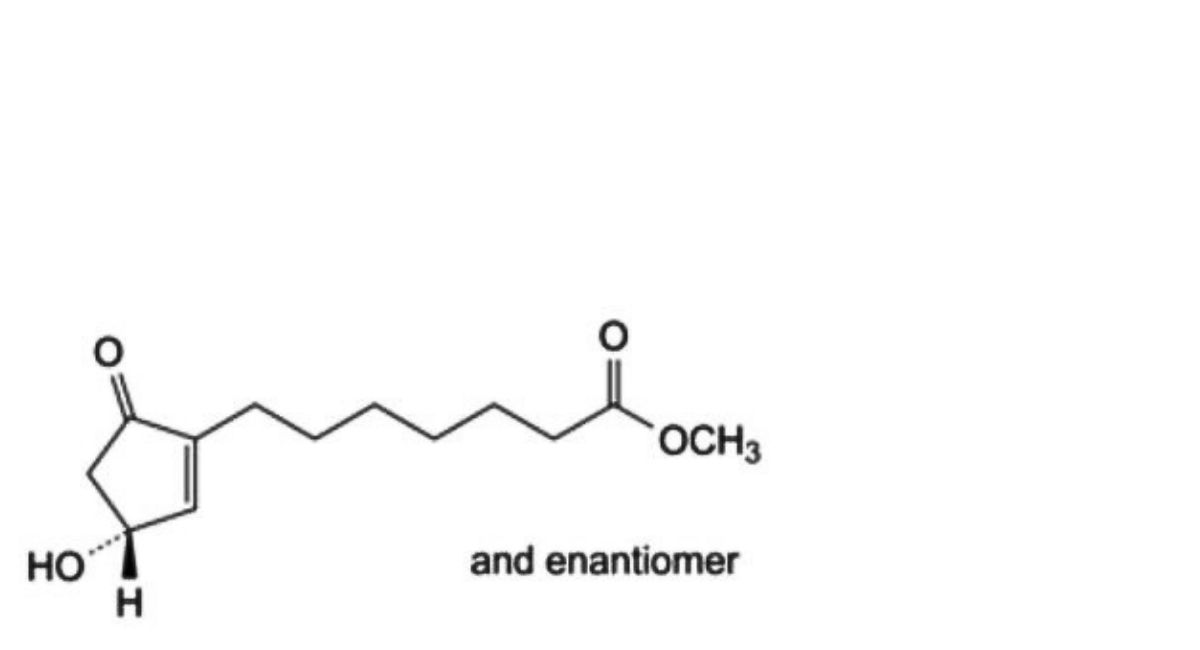
F. methyl 7-[(3RS)-3-hydroxy-5-oxocyclopent-1-enyl]heptanoate.
Ph Eur