Edition: BP 2025 (Ph. Eur. 11.6 update)
(Ph. Eur. monograph 1028)
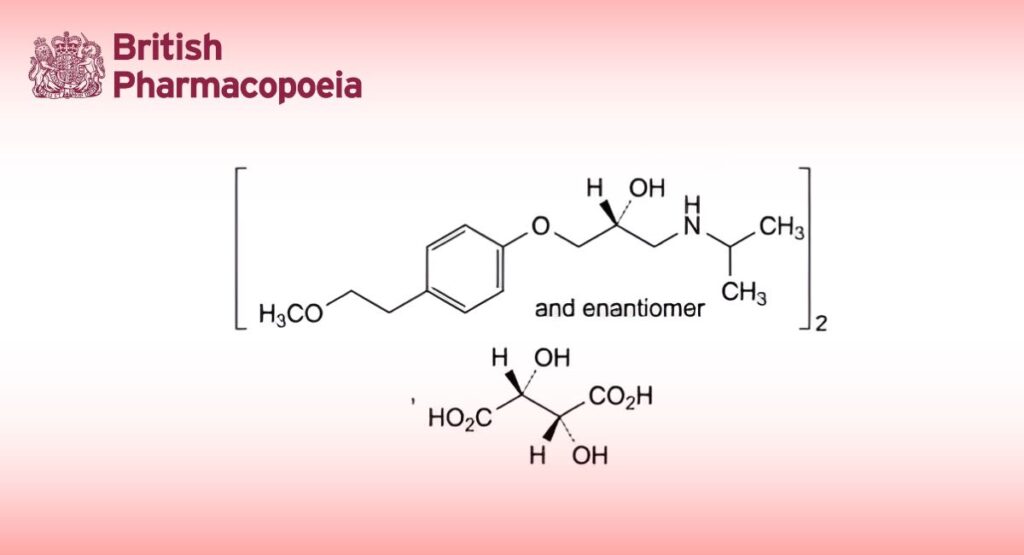
C34H56N2O12 685 56392-17-7
Action and use
Beta-adrenoceptor antagonist.
Preparations
Metoprolol Injection
Metoprolol Oral Suspension
Metoprolol Tartrate Prolonged-release Tablets
Metoprolol Tartrate Tablets
Ph Eur
DEFINITION
Bis[(2RS)-1-[4-(2-methoxyethyl)phenoxy]-3-[(1-methylethyl)amino]propan-2-ol] (2R,3R)-2,3 dihydroxybutanedioate.
Content
99.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder or colourless crystals.
Solubility
Very soluble in water, freely soluble in ethanol (96 per cent).
It shows polymorphism (5.9).
IDENTIFICATION
A. Specific optical rotation (see Tests).
B. Infrared absorption spectrophotometry (2.2.24).
Comparison metoprolol tartrate CRS.
If the spectra obtained in the solid state show differences, record further spectra using discs prepared by placing 25 μL of a 100 g/L solution in methylene chloride R on a disc of potassium bromide R and evaporating the solvent. Examine immediately.
TESTS
Solution S
Dissolve 0.500 g in carbon dioxide-free water R and dilute to 25.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and not more intensely coloured than reference solution B8
(2.2.2, Method II).
pH (2.2.3)
6.0 to 7.0 for solution S.
Specific optical rotation (2.2.7)
+ 7.0 to + 10.0 (dried substance), determined on solution S.
Impurities M, N, O
Thin-layer chromatography (2.2.27).
Test solution Dissolve 0.50 g of the substance to be examined in methanol R and dilute to 10 mL with the same solvent.
Reference solution (a) Dilute 1 mL of the test solution to 20 mL with methanol R. Dilute 5 mL of this solution to 50 mL with methanol R.
Reference solution (b) Dilute 4 mL of reference solution (a) to 10 mL with methanol R.
Plate TLC silica gel plate R.
Mobile phase Place 2 beakers, each containing 30 volumes of concentrated ammonia R, at the bottom of a chromatographic tank containing a mixture of 20 volumes of methanol R and 80 volumes of ethyl acetate R.
Application 5 μL.
Development Over 2/3 of the plate in a tank saturated for at least 1 h.
Drying In air for at least 3 h.
Detection Expose the plate to iodine vapour for at least 15 h.
Limits:
– any impurity: any spot, apart from the principal spot, is not more intense than the spot in the chromatogram obtained with reference solution (a) (0.5 per cent) and at most 1 such spot is more intense than the spot in the chromatogram obtained with reference solution (b) (0.2 per cent);
– disregard any spot on the line of application.
Related substances
Liquid chromatography (2.2.29).
Test solution Dissolve 20.0 mg of the substance to be examined in the mobile phase and dilute to 10.0 mL with the mobile phase.
Reference solution (a) Dissolve 1.5 mg of metoprolol impurity A CRS and 2.5 mg of metoprolol tartrate CRS in the mobile phase and dilute to 50.0 mL with the mobile phase.
Reference solution (b) Dilute 1.0 mL of the test solution to 20.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 50.0 mL with the mobile phase.
Column:
– size: l = 0.15 m, Ø = 3.9 mm;
– stationary phase: end-capped octadecylsilyl silica gel for chromatography R (5 μm).
Mobile phase Dissolve 3.9 g of ammonium acetate R in 810 mL of water R, add 2.0 mL of triethylamine R, 3.0 mL of phosphoric acid R, 10.0 mL of glacial acetic acid R and 146 mL of acetonitrile R and mix.
Flow rate 1 mL/min.
Detection Spectrophotometer at 280 nm.
Injection 20 μL.
Run time 3 times the retention time of metoprolol.
Relative retention With reference to metoprolol (retention time = about 7 min): impurity H = about 0.3; impurity C = about 0.4; impurity G = about 0.45; impurity F = about 0.7; impurity A = about 0.8; impurity J = about 1.4; impurity D = about 1.6; impurity E = about 1.8; impurity B = about 2.
System suitability Reference solution (a):
– resolution: minimum 6.0 between the peaks due to impurity A and metoprolol.
Limits:
– correction factor: for the calculation of content, multiply the peak area of impurity C by 0.1;
– impurities A, B, C, D, E, F, G, H, J: for each impurity, not more than 3 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.3 per cent);
– unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.10 per cent);
– total: not more than 5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.5 per cent);
– disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent); disregard any peak due to tartaric acid.
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in vacuo over anhydrous calcium chloride R for 4 h.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.250 g in 30 mL of anhydrous acetic acid R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 34.24 mg of C34H56N2O12.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D, E, F, G, H, J, M, N, O.
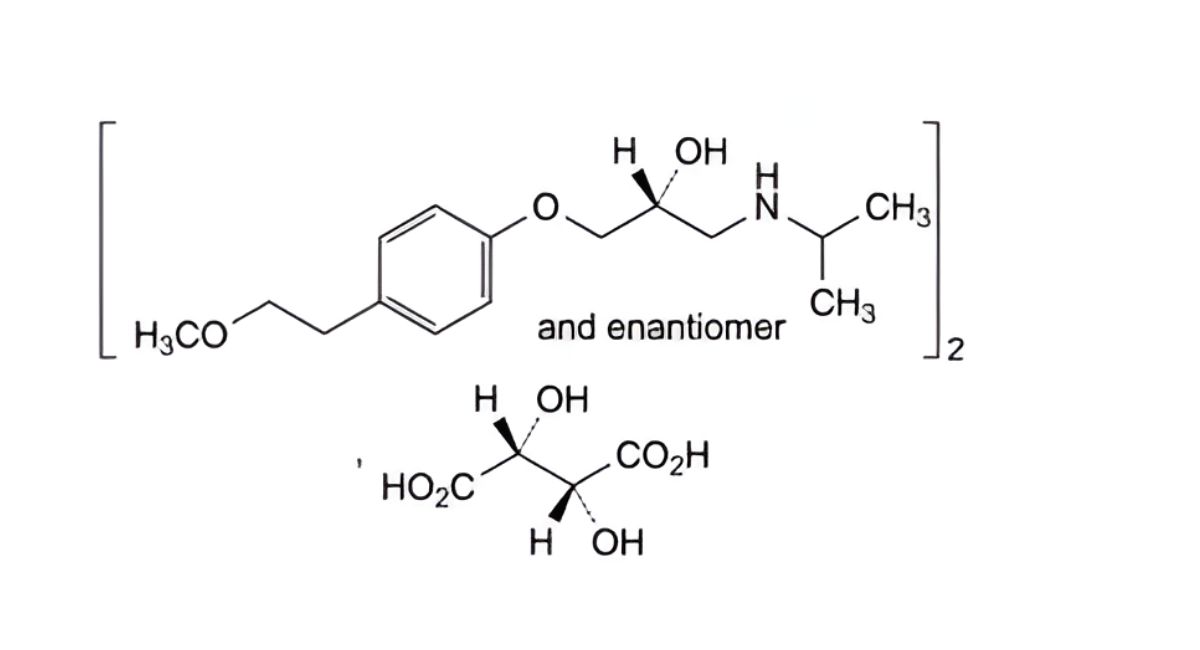
A. (2RS)-1-(ethylamino)-3-[4-(2-methoxyethyl)phenoxy]propan-2-ol,
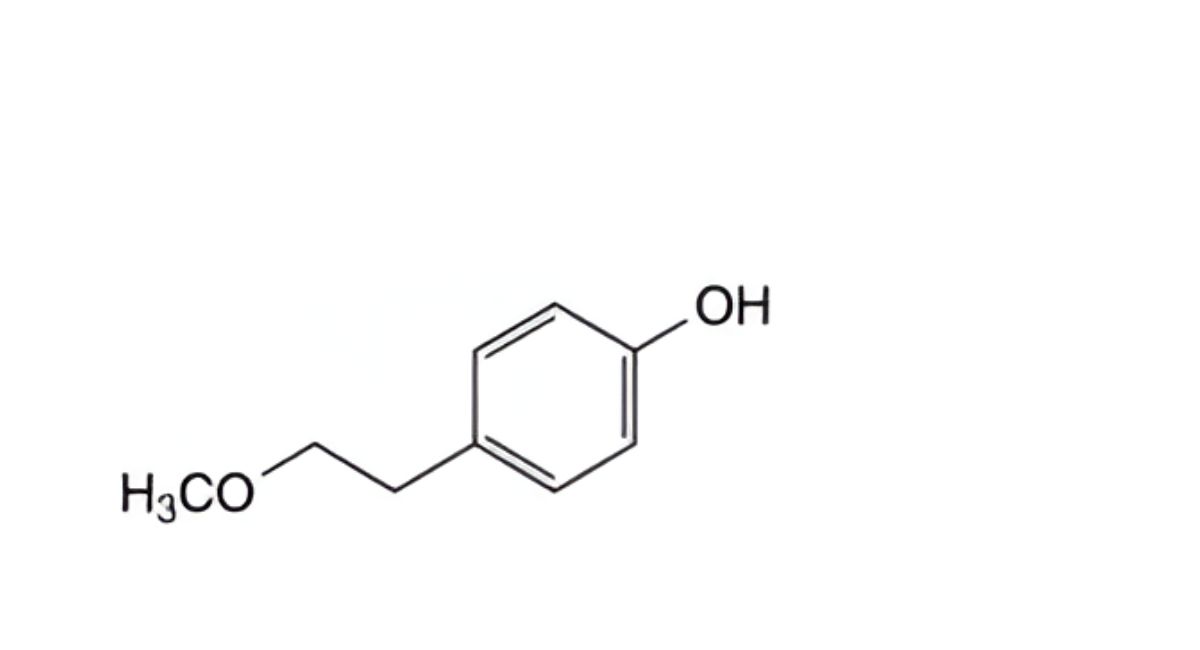
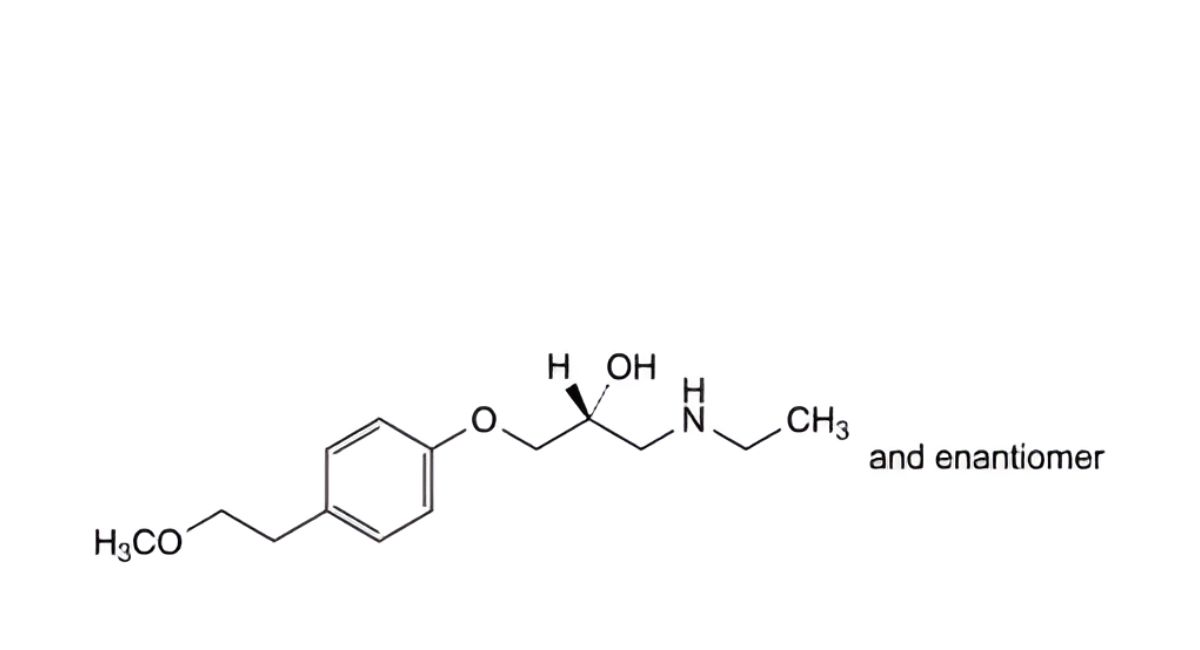
B. 4-(2-methoxyethyl)phenol,
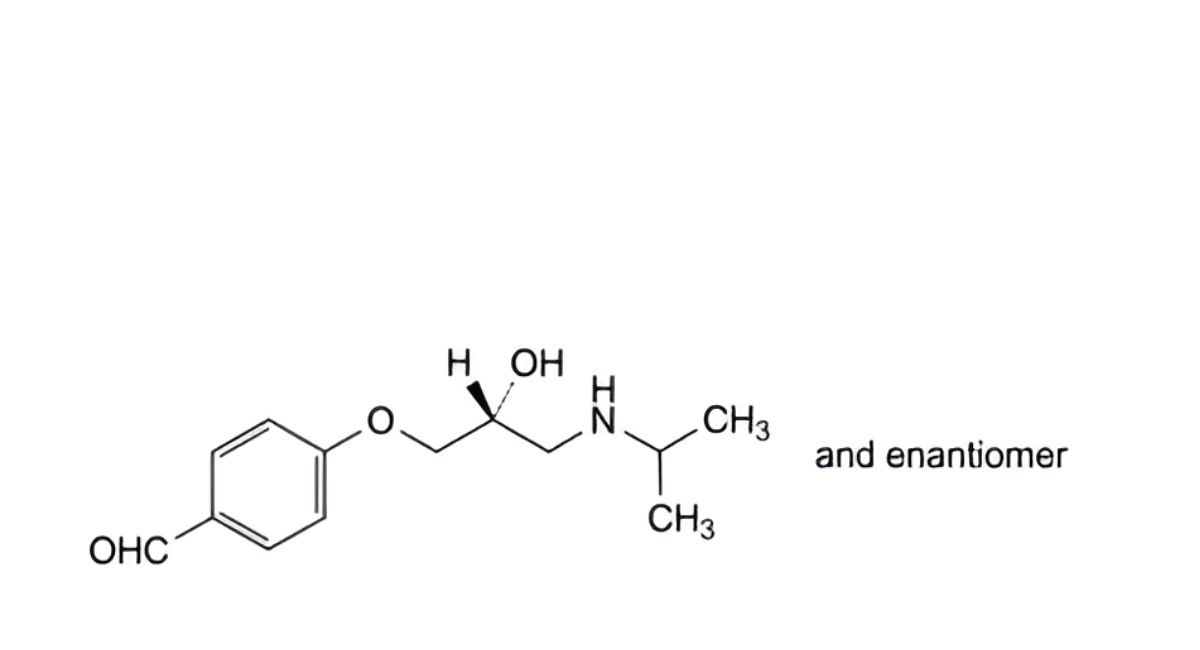
C. 4-[(2RS)-2-hydroxy-3-[(1-methylethyl)amino]propoxy]benzaldehyde,
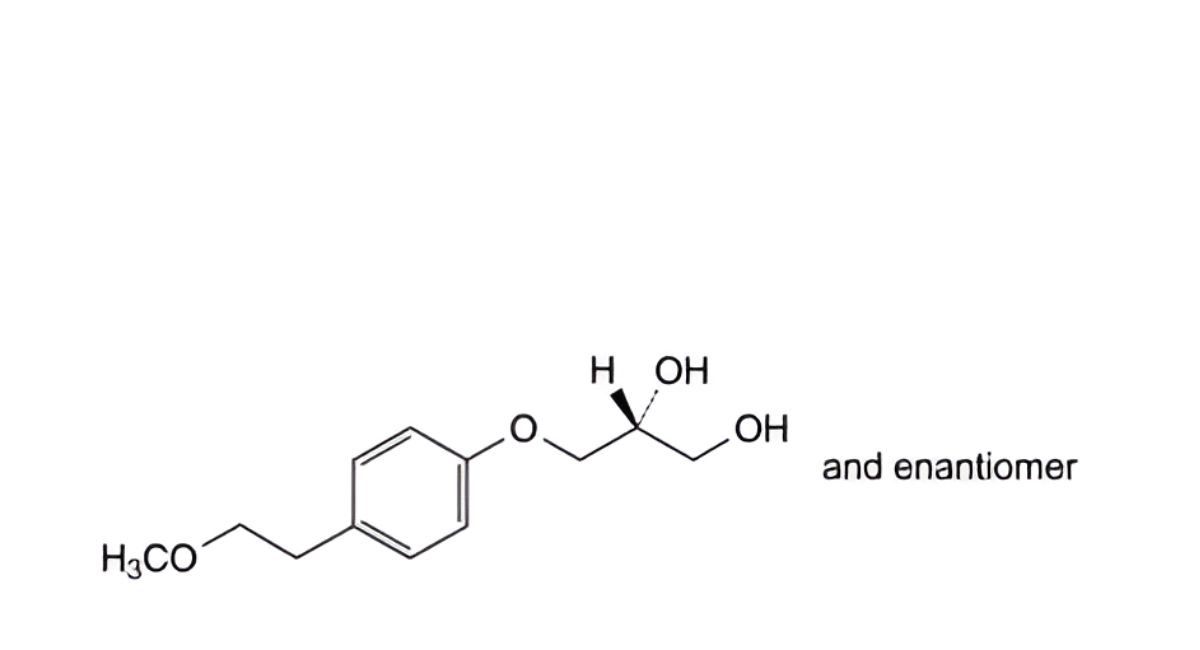
D. (2RS)-3-[4-(2-methoxyethyl)phenoxy]propane-1,2-diol,
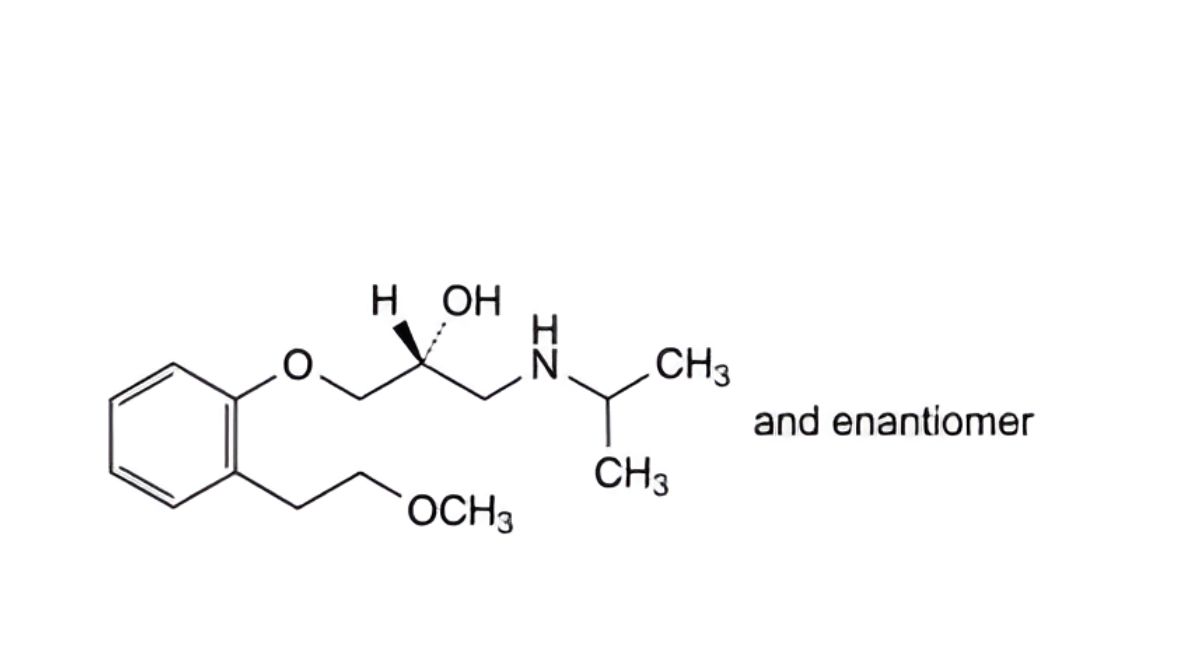
E. (2RS)-1-[2-(2-methoxyethyl)phenoxy]-3-[(1-methylethyl)amino]propan-2-ol,
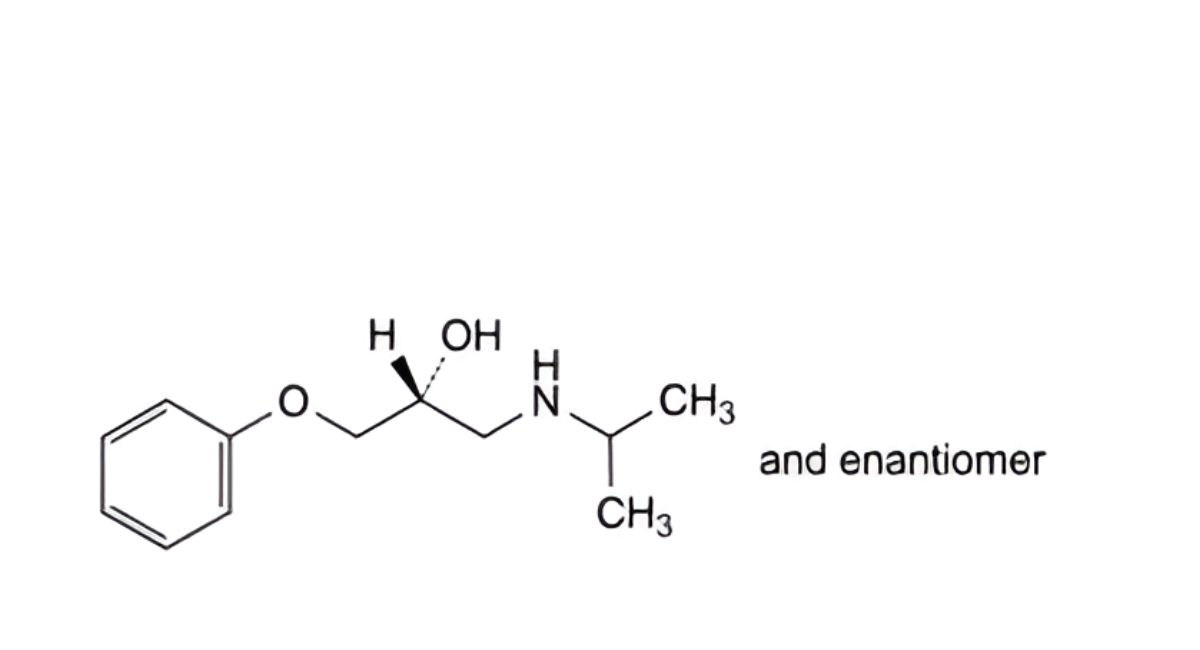
F. (2RS)-1-[(1-methylethyl)amino]-3-phenoxypropan-2-ol,
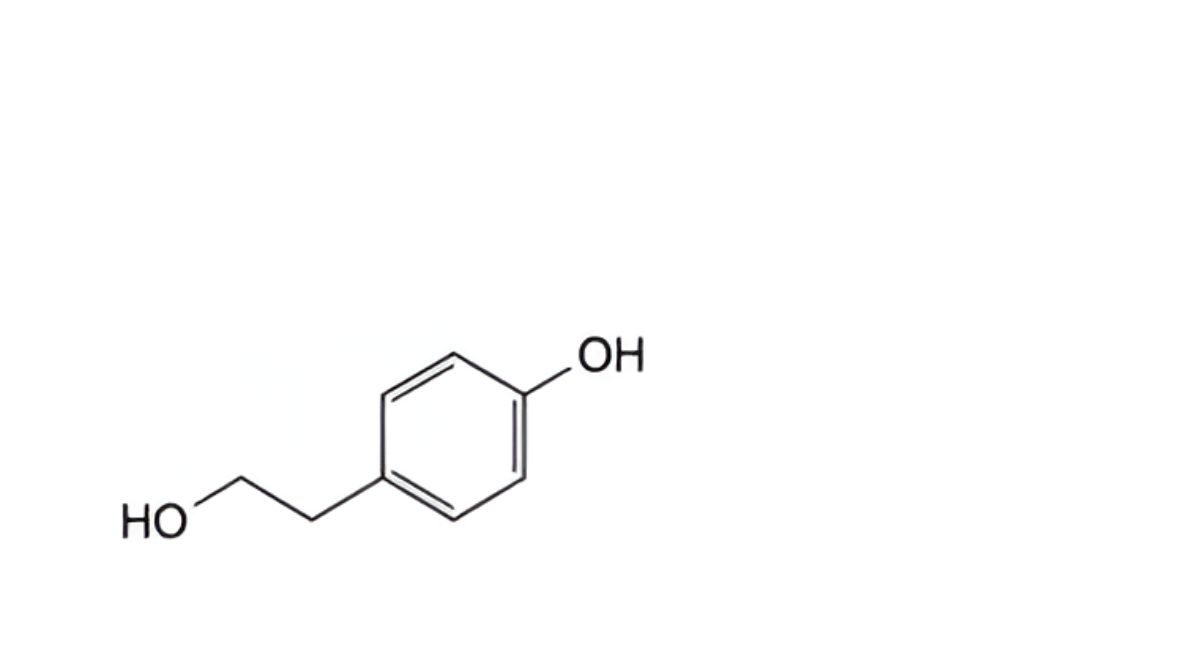
G. 2-(4-hydroxyphenyl)ethanol,
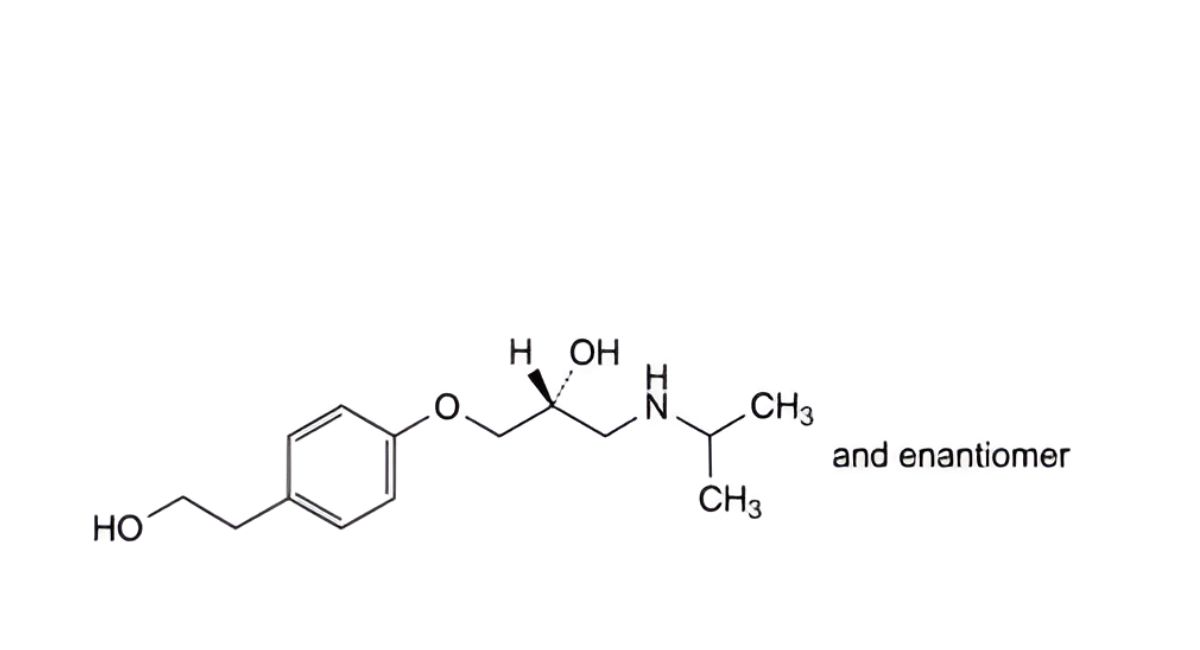
H. (2RS)-1-[4-(2-hydroxyethyl)phenoxy]-3-[(1-methylethyl)amino]propan-2-ol,
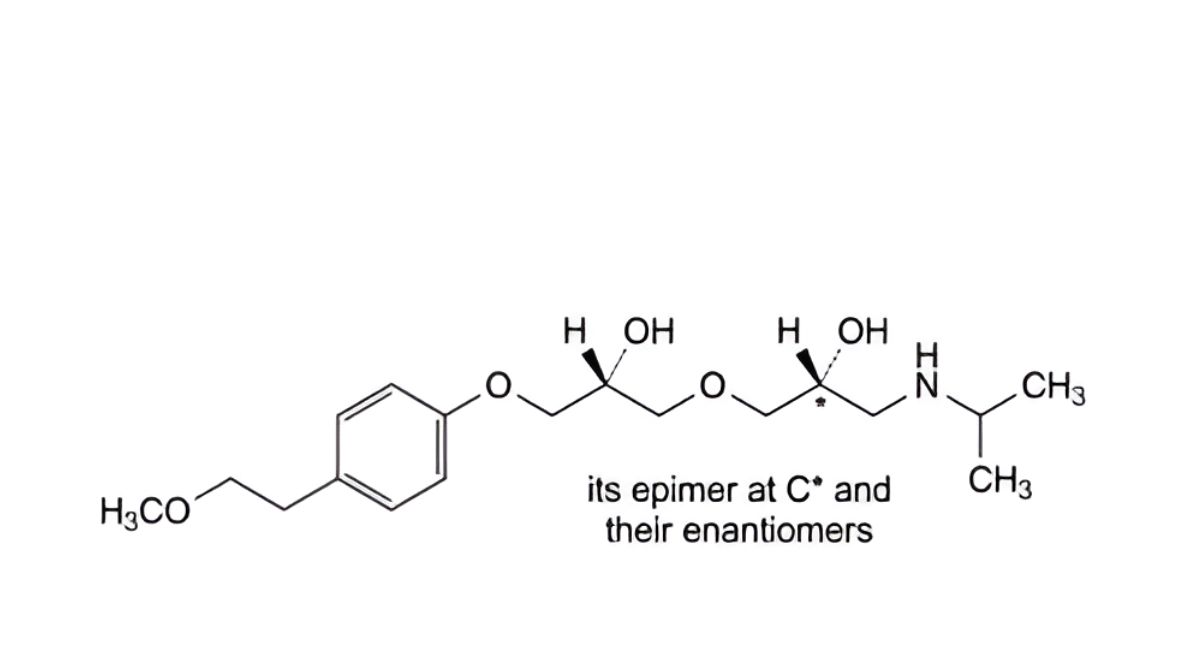
J. mixture of the 4 stereoisomers of 1-[2-hydroxy-3-[(1-methylethyl)amino]propoxy]-3-[4-(2-methoxyethyl)phenoxy]propan-2-ol,
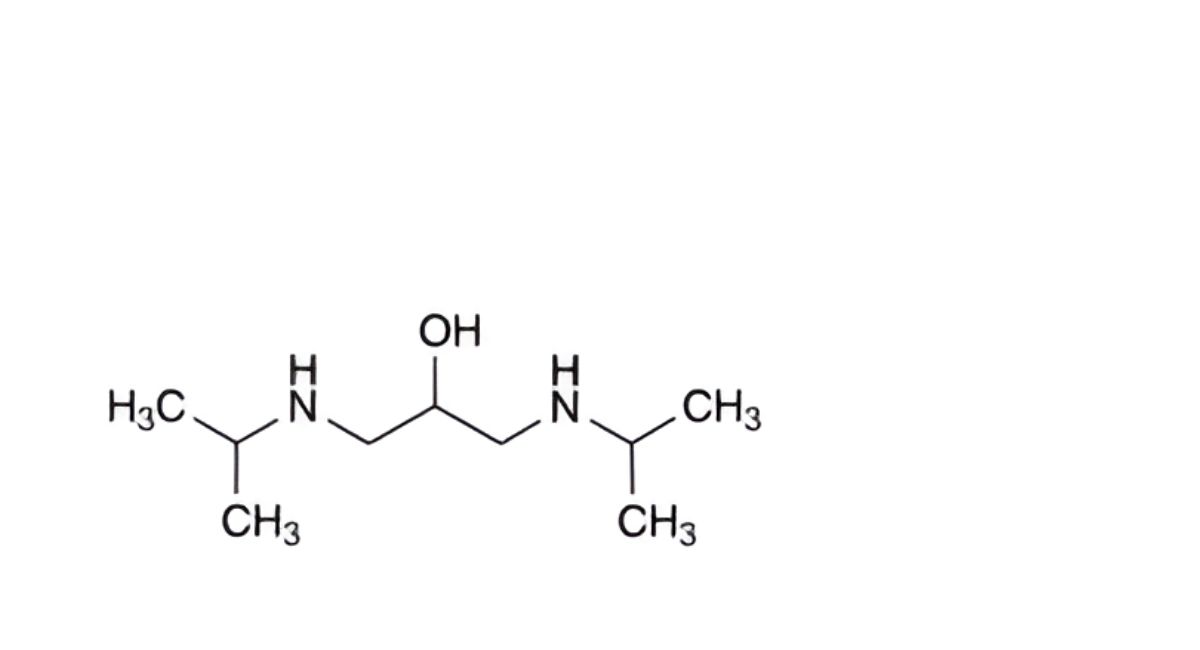
M. 1,3-bis[(1-methylethyl)amino]propan-2-ol,
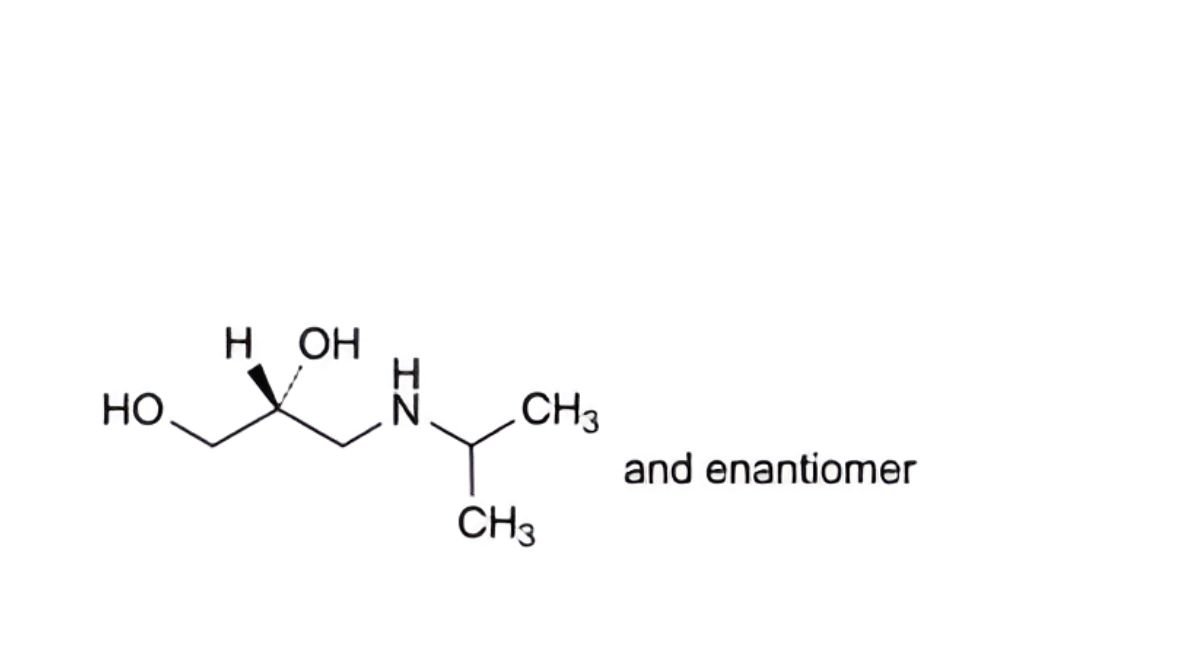
N. (2RS)-3-[(1-methylethyl)amino]propane-1,2-diol,
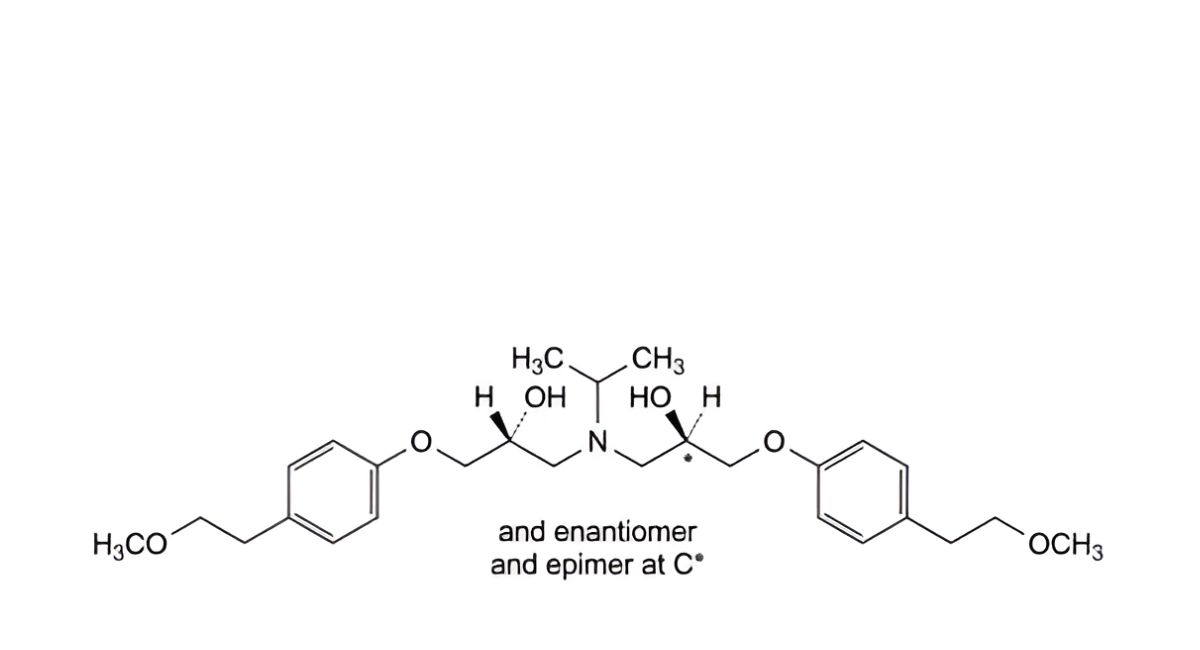
O. mixture of the 3 stereoisomers of 1,1′-[(1-methylethyl)imino]bis[3-[4-(2-methoxyethyl)phenoxy]propan-2-ol].
Ph Eur