BP 2025 (Ph. Eur. 11.6 update)
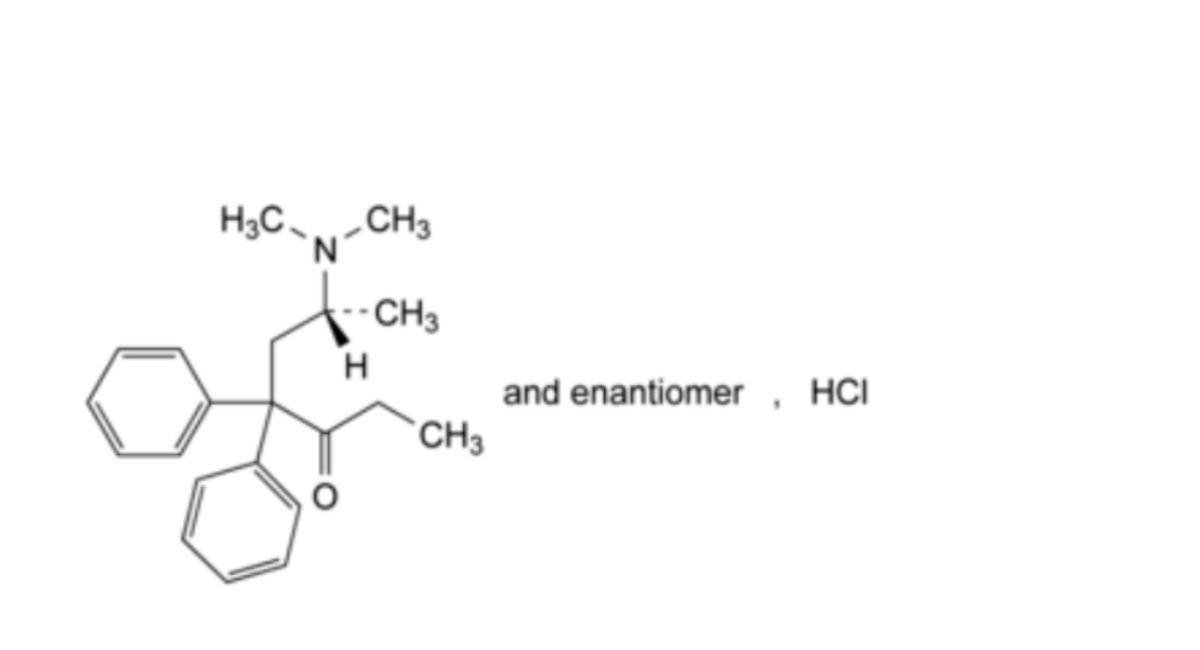
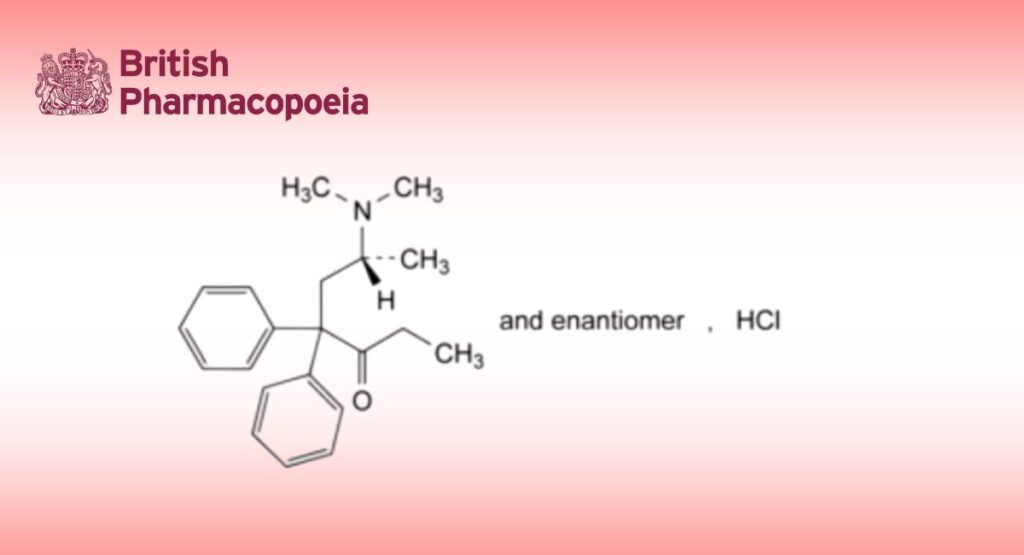
C21H28ClNO 345.9 1095-90-5
Action and use
Opioid receptor agonist; analgesic; treatment of opioid dependence.
Preparations
Methadone Concentrate for Oral Solution
Methadone Injection
Methadone Oral Solution (1 mg per ml)
Methadone Tablets
DEFINITION
(6RS)-6-(Dimethylamino)-4,4-diphenylheptan-3-one hydrochloride.
Content
99.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Soluble in water, freely soluble in ethanol (96 per cent).
IDENTIFICATION
First identification: A, C, D.
Second identification: A, B, D.
A. Optical rotation (see Tests).
B. Melting point (2.2.14): 233 °C to 236 °C.
C. Infrared absorption spectrophotometry (2.2.24).
Comparison Ph. Eur. reference spectrum of methadone hydrochloride.
D. Dilute 1 mL of solution S (see Tests) to 5 mL with water R and add 1 mL of dilute ammonia R1. Mix, allow to stand for 5 min and filter. The filtrate gives reaction (a) of chlorides (2.3.1).
TESTS
Solution S
Dissolve 2.50 g in carbon dioxide-free water R and dilute to 50.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and colourless (2.2.2, Method II).
Acidity or alkalinity
Dilute 10 mL of solution S to 25 mL with carbon dioxide-free water R. To 10 mL of the solution add 0.2 mL of methyl red solution R and 0.2 mL of 0.01 M sodium hydroxide. The solution is yellow. Add 0.4 mL of 0.01 M hydrochloric acid. The solution is red.
Optical rotation (2.2.7)
-0.05° to + 0.05°, determined on solution S in a 2 dm tube.
Related substances
Gas chromatography (2.2.28).
Test solution Dissolve 0.100 g of the substance to be examined in methanol R and dilute to 10.0 mL with the same solvent.
Reference solution (a) Dilute 1.0 mL of the test solution to 10.0 mL with methanol R. Dilute 1.0 mL of this solution to 100.0 mL with methanol R.
Reference solution (b) Dissolve 5 mg of imipramine hydrochloride CRS and 5 mg of cyclobenzaprine hydrochloride CRS in 100.0 mL of methanol R.
Column:
— material: fused silica;
— size: l = 50 m, Ø = 0.32 mm;
— stationary phase: phenyl(5)methyl(95)polysiloxane R (film thickness 1.05 μm).
Carrier gas helium for chromatography R.
Flow rate 1.2 mL/min.
Injection liner Packed with deactivated glass wool to wipe the needle.
Split ratio 1:100.
Temperature:
| Time (min) |
Temperature (°C) |
|
| Column | 0 – 4 | 150 → 250 |
| 4 – 35 | 250 | |
| Injection | 200 | |
| Detector | 250 |
Detection Flame ionisation.
Injection 2 μL.
Run time 1.5 times the retention time of methadone.
Relative retention With reference to methadone (retention time = about 25 min): impurity E = about 0.44; impurity C = about 0.81; impurity B = about 0.89; impurity D = about 0.98; impurity A = about 1.14; imipramine = about 1.19; cyclobenzaprine = about 1.24.
System suitability Reference solution (b):
— resolution: minimum 3.0 between the peaks due to imipramine and cyclobenzaprine.
Limits:
— impurities A, B, C, D, E: for each impurity, not more than the area of the principal peak in the
chromatogram obtained with reference solution (a) (0.1 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the
chromatogram obtained with reference solution (a) (0.10 per cent);
— total: not more than 3 times the area of the principal peak in the chromatogram obtained with
reference solution (a) (0.3 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference
solution (a) (0.05 per cent).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.300 g in a mixture of 5 mL of 0.01 M hydrochloric acid and 50 mL of anhydrous ethanol R. Carry out a potentiometric titration (2.2.20), using 0.1 M sodium hydroxide. Read the volume added between the 2 points of inflexion. Carry out a blank titration.
1 mL of 0.1 M sodium hydroxide is equivalent to 34.59 mg of C21H28ClNO.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D, E.
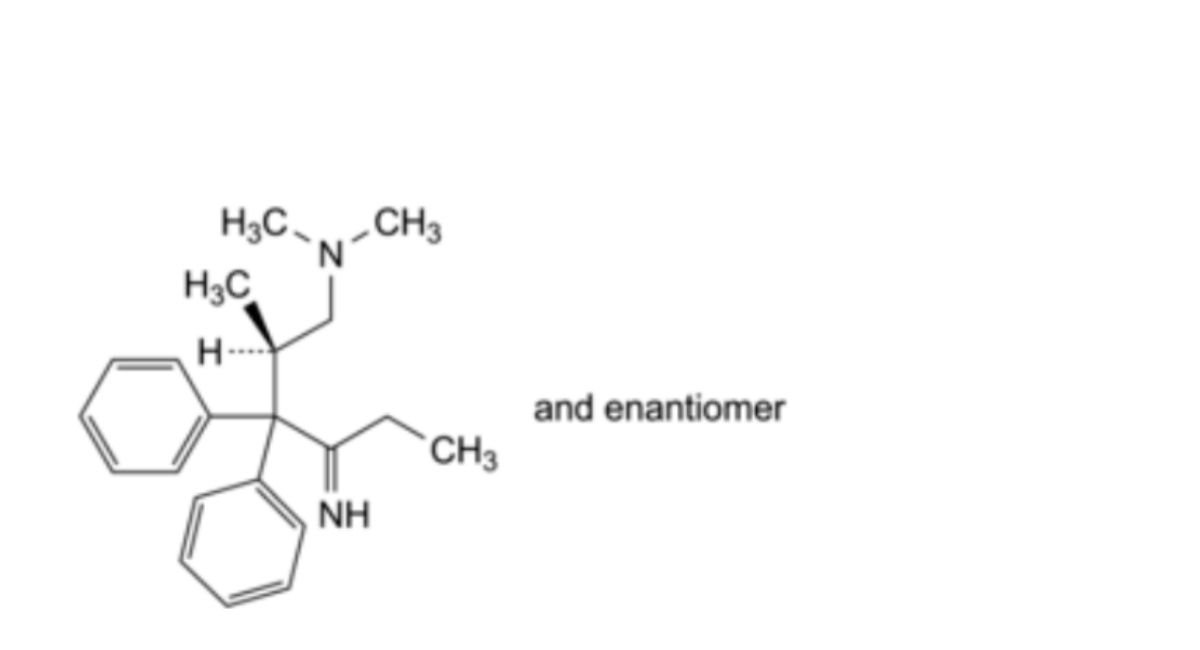
A. (2RS)-4-imino-N,N,2-trimethyl-3,3-diphenylhexan-1-amine (isomethadone ketimine),
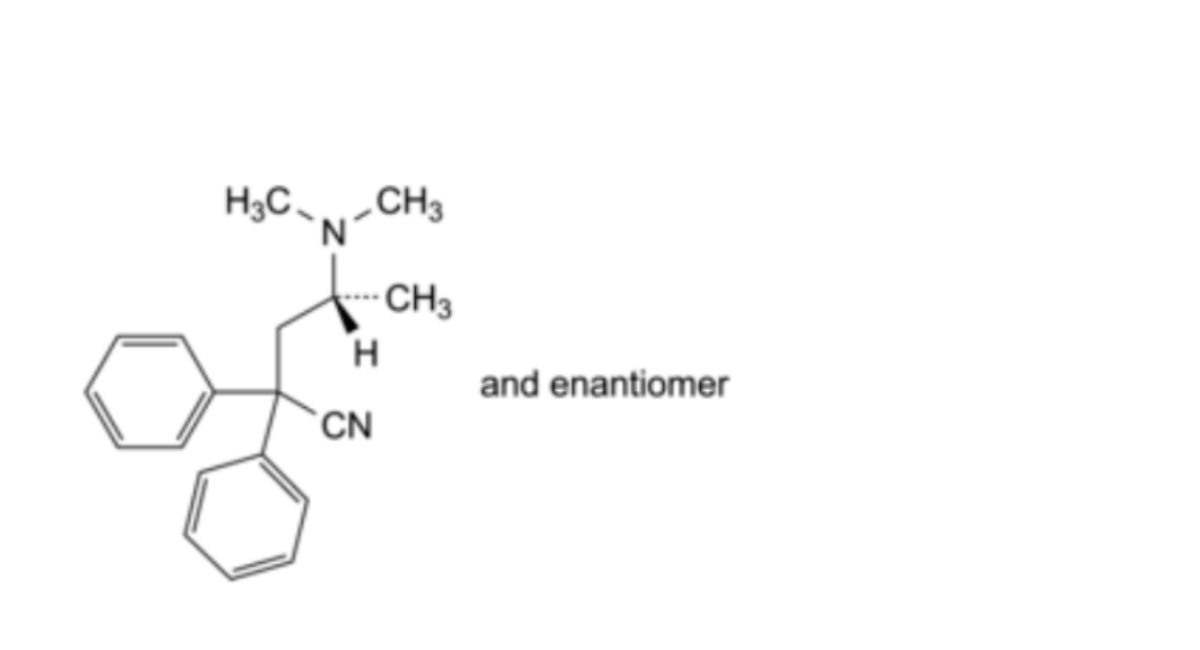
B. (4RS)-4-(dimethylamino)-2,2-diphenylpentanenitrile (didiavalo),
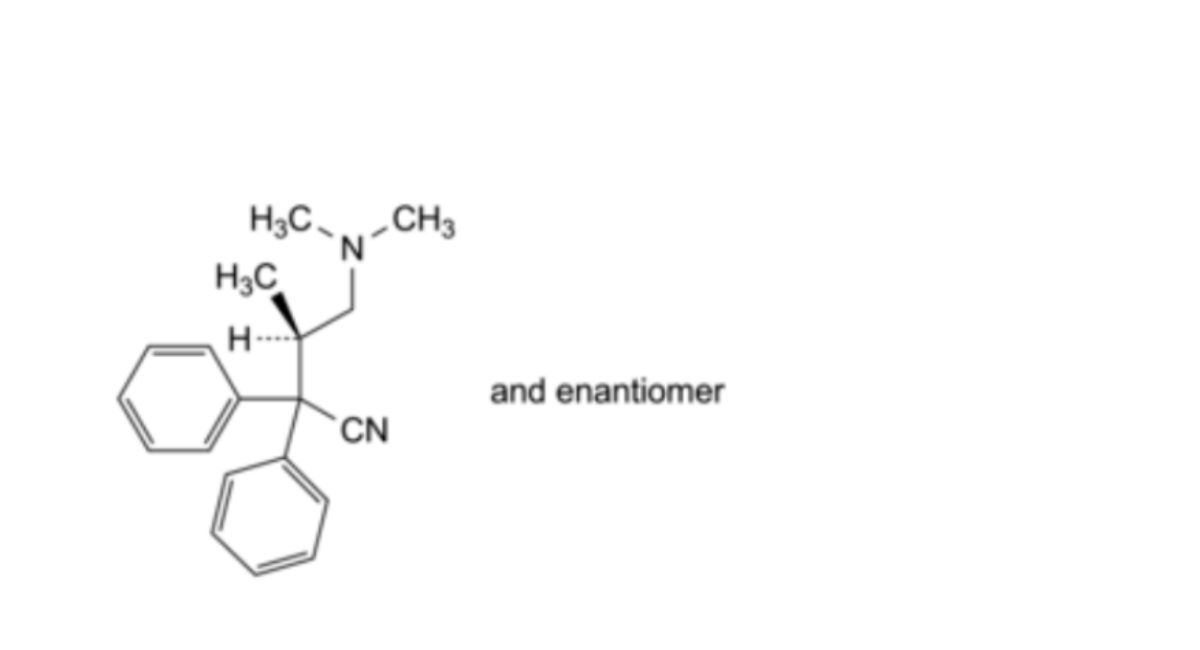
C. (3RS)-4-(dimethylamino)-3-methyl-2,2-diphenylbutanenitrile (isodidiavalo),
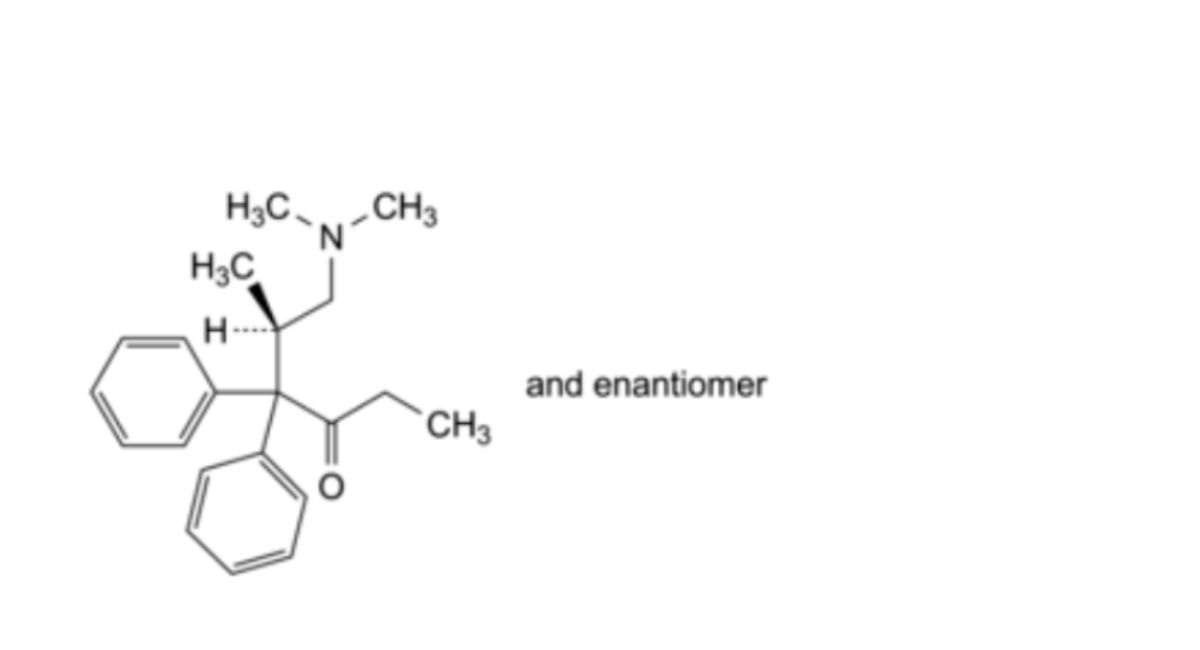
D. (5RS)-6-(dimethylamino)-5-methyl-4,4-diphenylhexan-3-one (isomethadone),
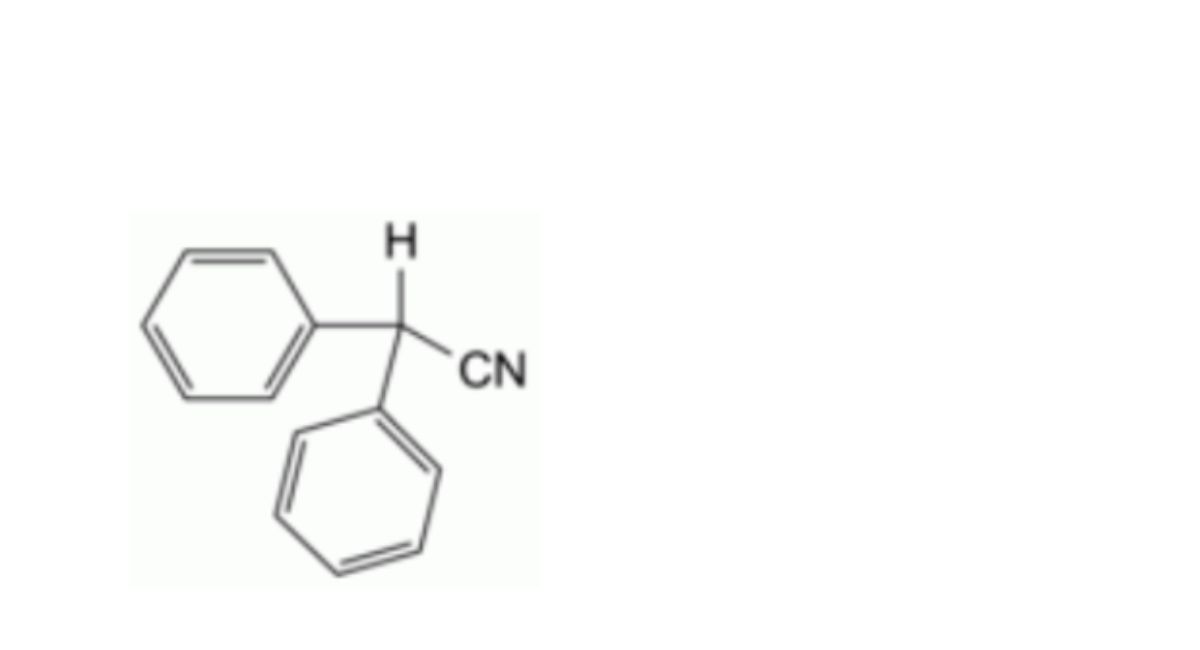
E. diphenylacetonitrile.