Edition: BP 2025 (Ph. Eur. 11.6 update)
General Notices
(Ph. Eur. monograph 1674)
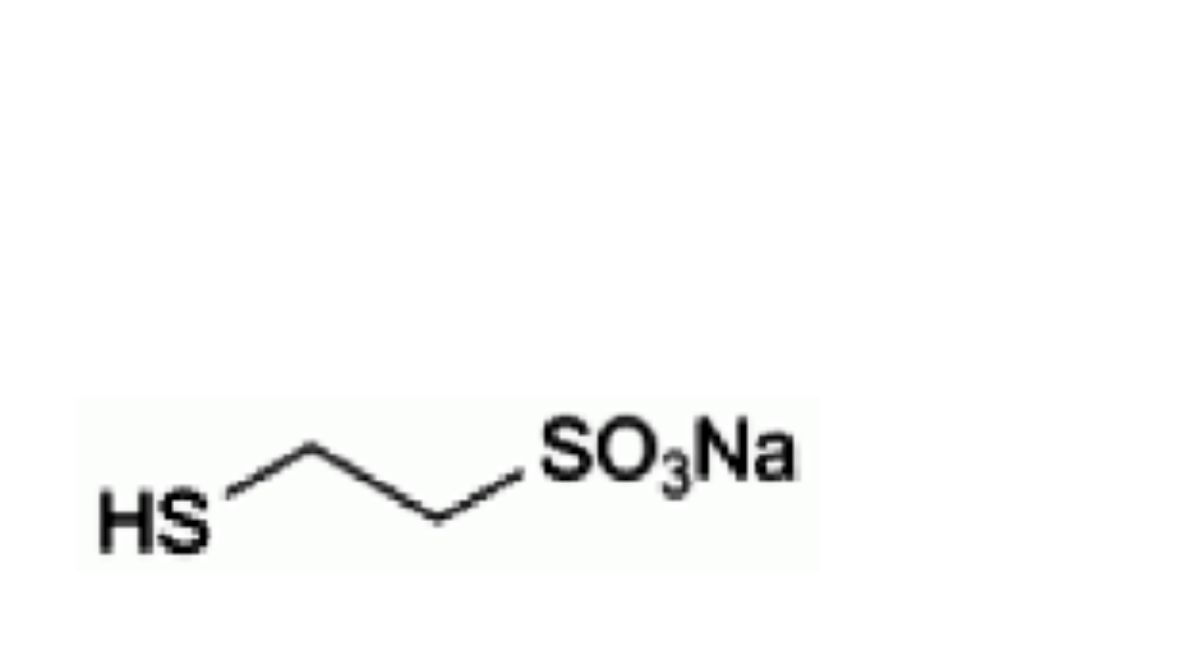
C2H5NaO3S2 164.2 19767-45-4
Action and use
Preventing adverse effects of cyclophosphamide and ifosfamide.
DEFINITION
Sodium 2-sulfanylethanesulfonate.
Content
96.0 per cent to 102.0 per cent (dried substance).
CHARACTERS
Appearance
White or slightly yellow, crystalline powder, hygroscopic.
Solubility
Freely soluble in water, slightly soluble in ethanol (96 per cent), practically insoluble in cyclohexane.
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Comparison Ph. Eur. reference spectrum of mesna.
B. It gives reaction (a) of sodium (2.3.1).
TESTS
Solution S
Dissolve 10.0 g in carbon dioxide-free water R prepared from distilled water R and dilute to 50 mL with the same solvent.
Appearance of solution
Solution S is not more opalescent than reference suspension II (2.2.1) and not more intensely coloured tha reference solution Y7 (2.2.2, Method II).
pH (2.2.3)
4.5 to 6.0.
Dilute 10 mL of solution S to 20 mL with carbon dioxide-free water R.
Related substances
Liquid chromatography (2.2.29).
Test solution Dissolve 0.10 g of the substance to be examined in the mobile phase and dilute to 25.0 mL with the mobile phase.
Reference solution (a) Dissolve 4.0 mg of mesna impurity C CRS in the mobile phase and dilute to 50.0 m with the mobile phase. Dilute 2.0 mL of the solution to 20.0 mL with the mobile phase.
Reference solution (b)
with the mobile phase.
Dissolve 6.0 mg of mesna impurity D CRS in the mobile phase and dilute to 50.0 n
Reference solution (c)
Dilute 3.0 mL of the test solution to 10.0 mL with the mobile phase.
Reference solution (d)
Dilute 1.0 mL of reference solution (c) to 100.0 mL with the mobile phase.
Reference solution (e)
this solution add 10 mL of reference solution (a).
Dilute 6 mL of reference solution (c) to 20 mL with the mobile phase. To 10 mL of
Column:
— size: l = 0.25 m, 0 = 4.6 mm;
— stationary phase: octadecylsilyl silica gel for chromatography R (10 pm).
Mobile phase Dissolve 2.94 g of potassium dihydrogen phosphate R, 2.94 g of dipotassium hydrogen phosphate R and 2.6 g of tetrabutylammonium hydrogen sulfate R in about 600 mL of water for chromatography R. Adjust to pH 2.3 with phosphoric acid R, add 335 mL of methanol R1 and dilute to 1000 mL with water for chromatography R.
Flow rate
1 mL/min.
Injection 20 pL.
Run time 4 times the retention time of mesna.
Relative retention With reference to mesna (retention time = about 4.8 min): impurities A and B = about 0. impurity E = about 0.8; impurity C = about 1.4; impurity D = about 2.3.
System suitability Reference solution (e):
— resolution: minimum 3.0 between the peaks due to mesna and impurity C.
Limits:
— correction factors: for the calculation of content, multiply the peak areas of impurities A, B and E by 0.01;
— impurity C: not more than the area of the corresponding peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
— impurity D: not more than the area of the corresponding peak in the chromatogram obtained with reference solution (b) (3.0 per cent);
— impurities A, B, E: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (d) (0.3 per cent);
— unspecified impurities: for each impurity, not more than one third of the area of the principal peak in the chromatogram obtained with reference solution (d) (0.10 per cent);
— sum of other impurities: not more than the area of the principal peak in the chromatogram obtained with reference solution (d) (0.3 per cent);
— disregard limit: 0.15 times the area of the principal peak in the chromatogram obtained with referenc solution (d) (0.045 per cent).
Chlorides (2.4.4)
Maximum 250 ppm.
Dilute 1 mL of solution S to 15 mL with water R.
Sulfates (2.4.13)
Maximum 300 ppm.
Dilute 5 mL of solution S to 30 mL with distilled water R. 15 mL of the solution complies with the test.
Disodium edetate
Maximum 500 ppm.
Dissolve 4.000 g in 90 mL of water R and adjust to pH 4.5 using 0.1 M hydrochloric acid. Add 10 mL of acetate buffer solution pH 4.5 R and 50 mL of 2-propanol R. Add 2 mL of a 0.25 g/L solution of dithizone R 2-propanol R. Titrate with 0.01 M zinc sulfate until the colour changes from bluish-grey to pink.
1 mL of 0.01 M zinc sulfate is equivalent to 3.72 mg of C10H14N2Na2O8,2H2O.
Loss on drying (2.2.32)
Maximum 1.0 per cent, determined on 1.000 g under vacuum at 60 °C for 2 h.
ASSAY
Dissolve 0.120 g in 10 mL of water R. Add 10 mL of dilute sulfuric acid R and 10.0 mL of 0.1 M iodine. Titra with 0.1 M sodium thiosulfate adding 1 mL of starch solution R near the endpoint. Carry out a blank titration
1 mL of 0.1 M sodium thiosulfate is equivalent to 16.42 mg of C2H5NaO3S2.
STORAGE
In an airtight container.
IMPURITIES
Specified impurities A, B, C, D, E.
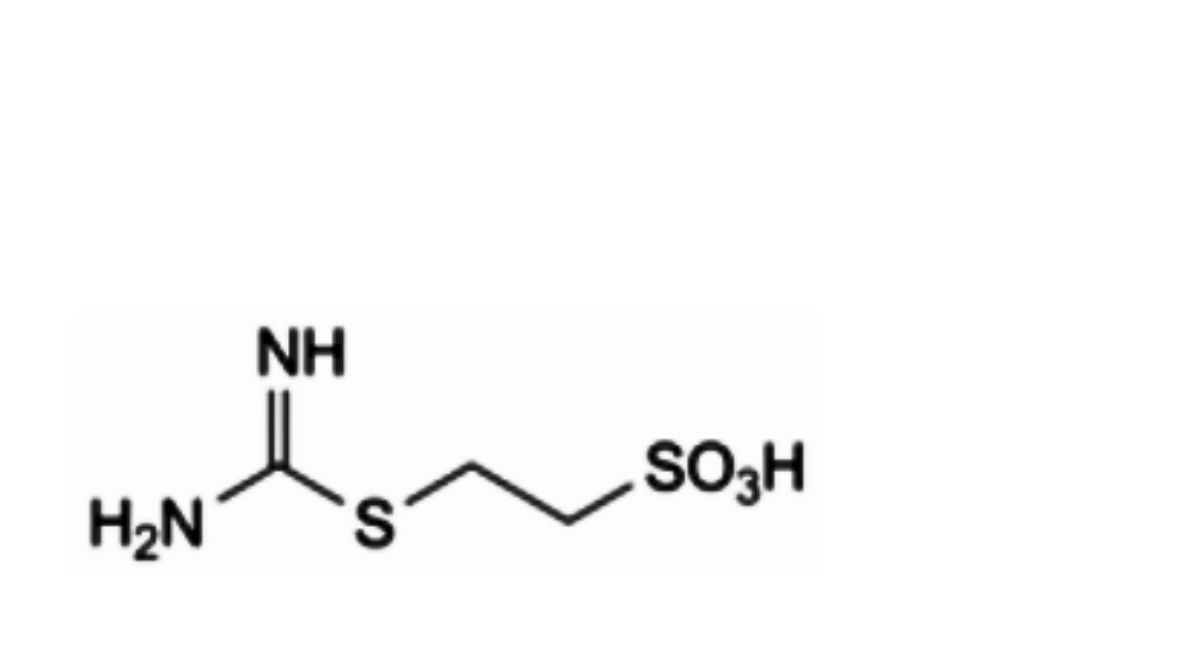
A. 2-(carbamimidoylsulfanyl)ethanesulfonic acid,
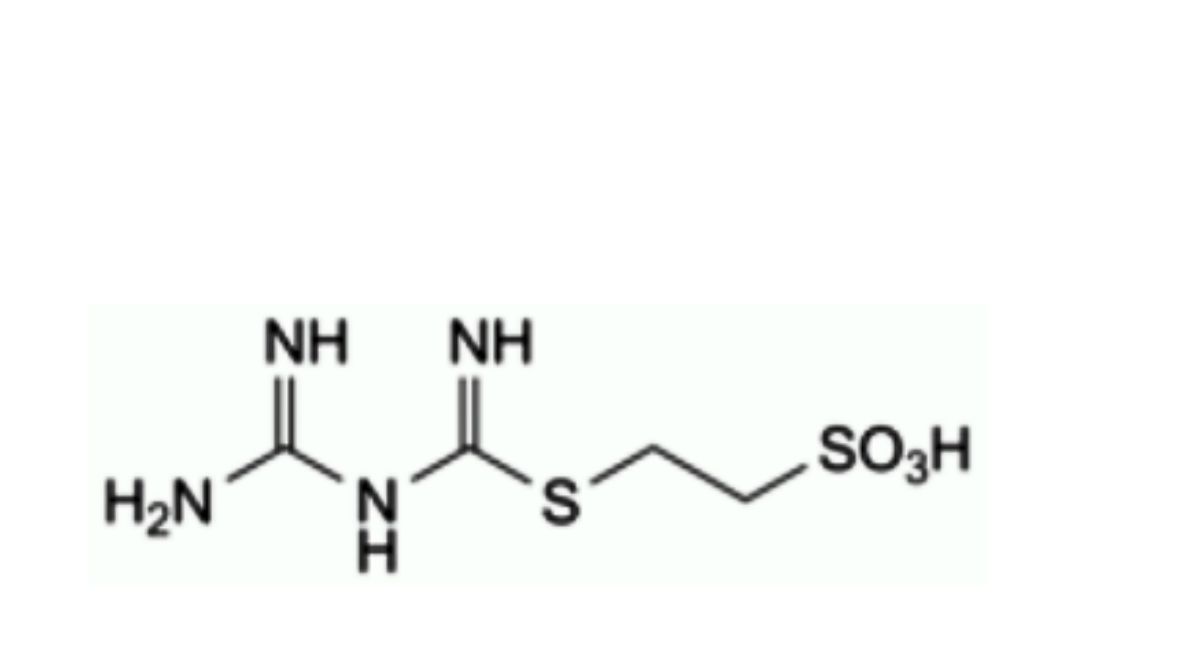
B. 2-[[(guanidino)(imino)methyl]sulfanyl]ethanesulfonic acid,
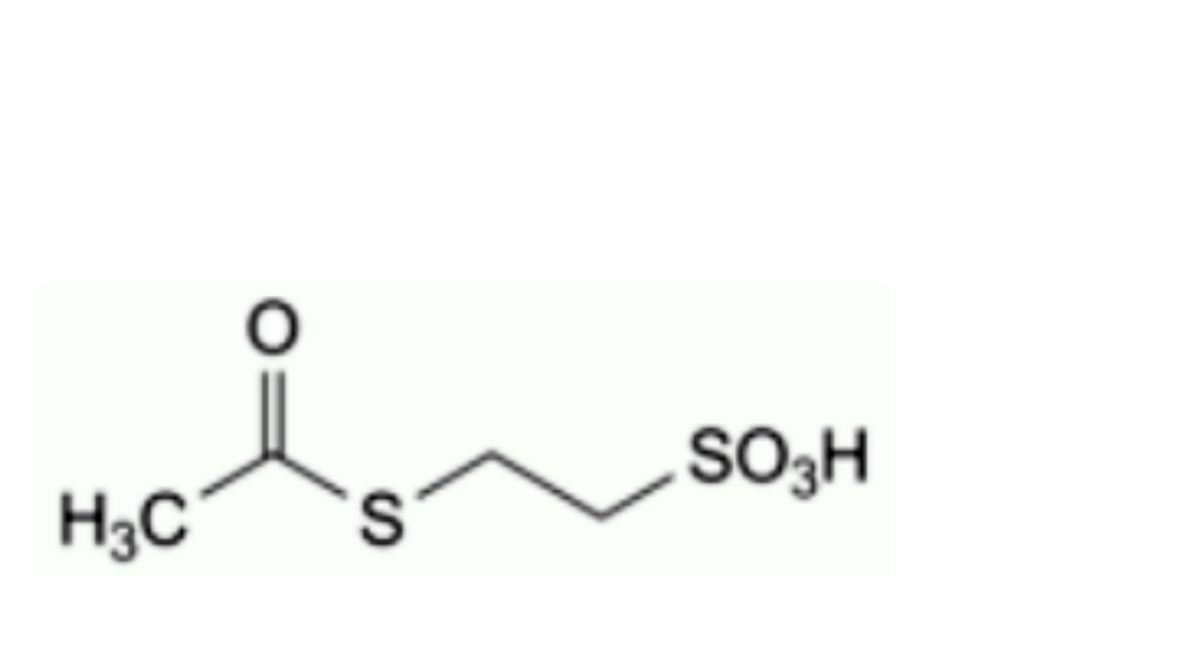
C. 2-(acetylsulfanyl)ethanesulfonic acid,
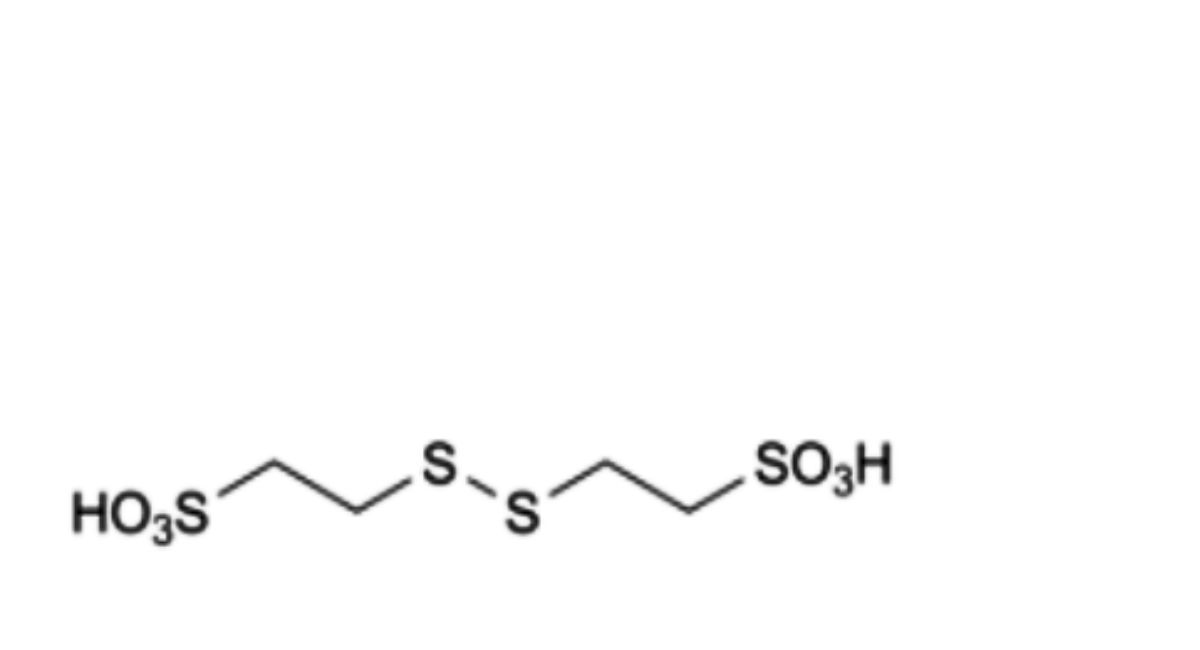
D. 2,2′-(disulfanediyl)bis(ethanesulfonic acid),
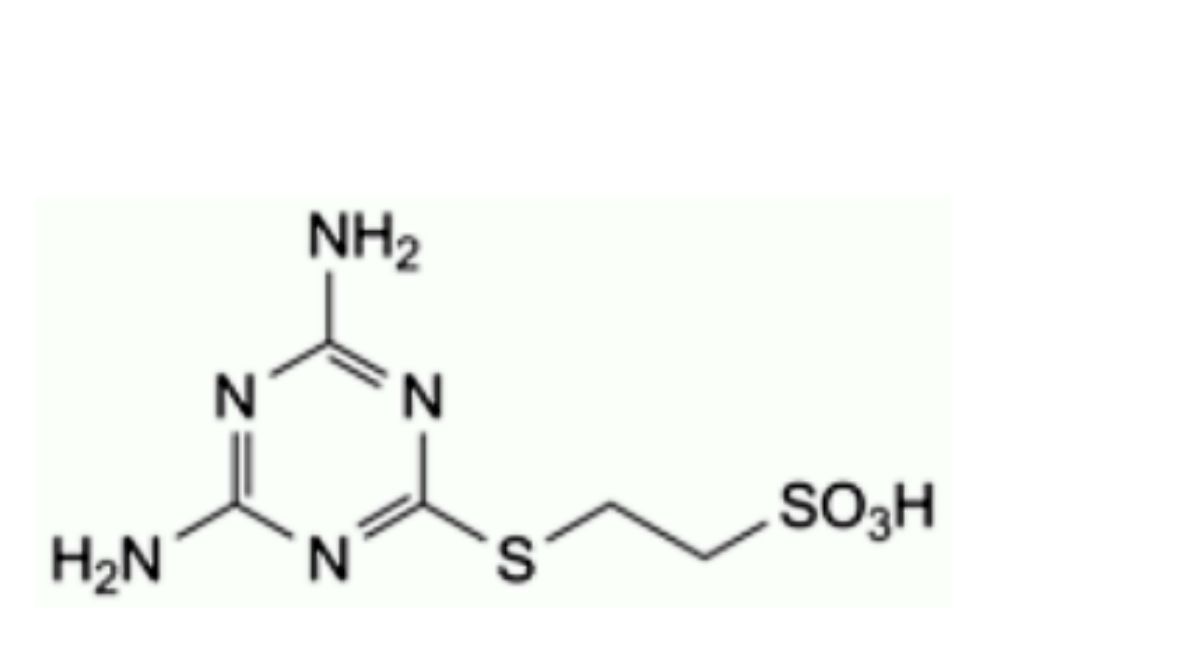
E. 2-(4,6-diamino-1,3,5-triazin-2-yl)sulfanylethanesulfonic acid.






