Edition: BP 2025 (Ph. Eur. 11.6 update)
General Notices
Action and use
Antifungal.
DEFINITION
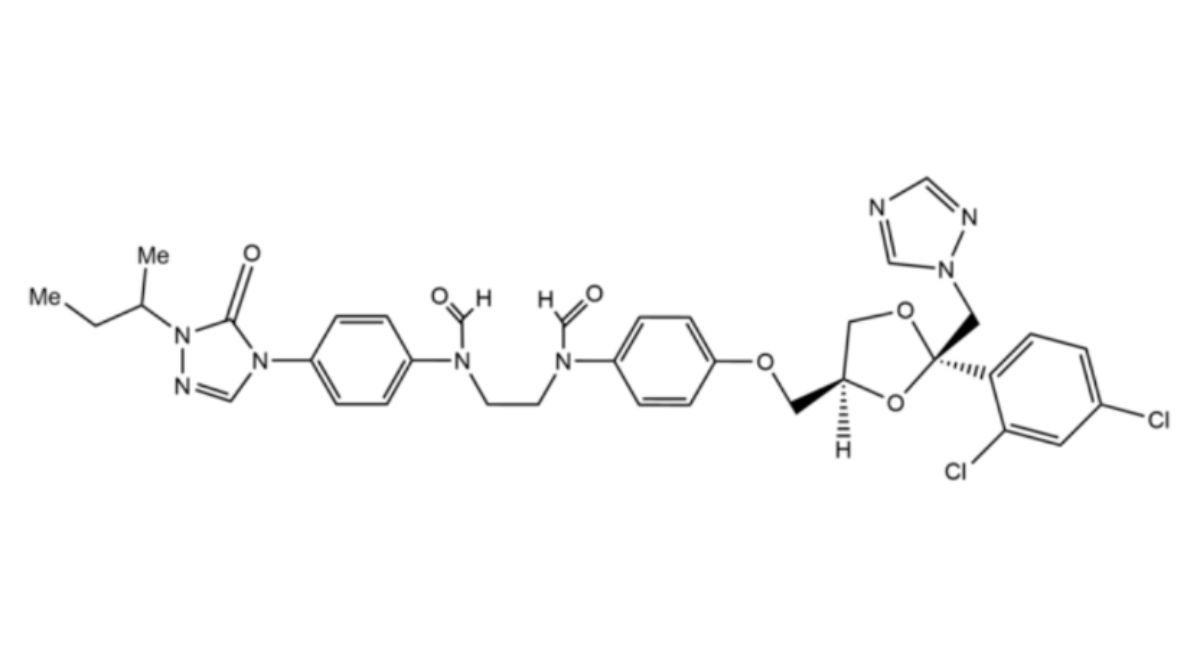
Itraconazole Capsules contain Itraconazole.
The capsules comply with the requirements stated under Capsules and with the following requirements.
Content of itraconazole, C35H38Cl2N8O4
95.0 to 105.0% of the stated amount.
IDENTIFICATION
A. Carry out the method for thin-layer chromatography, Appendix III A, using the following solutions.
(1) Shake a quantity of capsule contents containing 50 mg of Itraconazole with 50 mL of water and filter. Shake the residue with 50 mL of dichloromethane and filter (a 0.45-μm PVDF filter is suitable) and use the filtrate.
(2) 0.1% w/v of itraconazole BPCRS in dichloromethane.
(3) 0.1% w/v of itraconazole BPCRS and 0.2% w/v of fluconazole BPCRS in dichloromethane.
CHROMATOGRAPHIC CONDITIONS
(a) Use as the coating silica gel F254 (Merck TLC silica gel 60 F254 plates are suitable).
(b) Use the mobile phase as described below.
(c) Apply 5 μL of each solution.
(d) Develop the plate to 15 cm.
(e) After removal of the plate, dry in air and examine under ultraviolet light (254 nm).
MOBILE PHASE
20 volumes of a solution containing 0.015% w/v of ammonium acetate and 0.0003% v/v of glacial acetic acid in water, 40 volumes of 1,4-dioxan and 40 volumes of methanol.
SYSTEM SUITABILITY
The test is not valid unless the chromatogram obtained with solution (3) shows two clearly separated spots.
CONFIRMATION
The principal spot in the chromatogram obtained with solution (1) corresponds to that in the chromatogram obtained with solution (2).
B. In the Assay, the retention time of the principal peak in the chromatogram obtained with solution (1) is similar to that of the principal peak in the chromatogram obtained with solution (2).
TESTS
Dissolution
Comply with the dissolution test for tablets and capsules, Appendix XII B1.
TEST CONDITIONS
(a) Use Apparatus 2, and rotating the paddle at 75 revolutions per minute.
(b) Use 900 mL of 0.5% w/v of sodium dodecyl sulfate in water, at a temperature of 37°, as the medium.
PROCEDURE
(1) After 60 minutes withdraw a sample of the medium and filter. Use the filtered medium, diluted with the dissolution medium if necessary, to produce a solution expected to contain 0.0055% w/v of Itraconazole.
(2) Dissolve 27.5 mg of itraconazole BPCRS in 10 mL of acetonitrile (60%) and 1 drop of orthophosphoric acid, and dilute to 100 mL with acetonitrile (60%). Dilute 1 volume to 5 volumes with dissolution medium to produce a solution containing 0.0055% w/v of itraconazole BPCRS.
CHROMATOGRAPHIC CONDITIONS
(a) Use a stainless steel column (15 cm × 4.6 mm) packed with end-capped octadecylsilyl silica gel for chromatography (5 μm) (Phenomenex Prodigy ODS-2 is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 2 mL per minute.
(d) Use a column temperature of 40°.
(e) Use a detection wavelength of 254 nm.
(f) Inject 15 μL of each solution.
MOBILE PHASE
45 volumes of 0.01M potassium dihydrogen orthophosphate, adjusted to pH 3.0 with orthophosphoric acid and 55 volumes of acetonitrile.
When the chromatograms are recorded under the prescribed conditions, the retention time of itraconazole is about 8 minutes.
DETERMINATION OF CONTENT
Calculate the total content of itraconazole, C35H38Cl2N8O4, in the medium from the chromatograms obtained and using the declared content of C35H38Cl2N8O4 in itraconazole BPCRS.
LIMITS
The amount of itraconazole released is not less than 70% (Q) of the stated amount.
Related substances
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
Solution A: 0.1 volumes of hydrochloric acid and 50 volumes of mobile phase.
(1) To a quantity of mixed capsule contents containing 0.1 g of Itraconazole, add 5 mL of mobile phase and 0.2 mL of hydrochloric acid, and mix with the aid of ultrasound. Add 50 mL of solution A and shake. Dilute to produce 100 mL with the mobile phase and filter (a 0.45-μm PVDF filter is suitable), discarding the first 6 mL.
(2) Dilute 1 volume of solution (1) to 100 volumes with solution A and further dilute 1 volume to 5 volumes with solution A.
(3) 0.1% w/v of itraconazole for system suitability EPCRS in solution A.
CHROMATOGRAPHIC CONDITIONS
(a) Use a stainless steel column (15 cm × 4.6 mm) packed with end-capped octadecylsilyl silica gel for chromatography (5 μm) (Phenomenex Prodigy ODS-2 is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 1.5 mL per minute.
(d) Use a column temperature of 40°.
(e) Use a detection wavelength of 254 nm.
(f) Inject 15 μL of each solution.
(g) Allow the chromatography to proceed for 3 times the retention time of itraconazole.
MOBILE PHASE
48 volumes of acetonitrile and 52 volumes of 0.01M potassium dihydrogen orthophosphate, adjusted to pH 3.0 with orthophosphoric acid.
When the chromatograms are recorded under the prescribed conditions, the retention times relative to itraconazole (retention time about 23 minutes) are: impurity A, about 0.3; impurity 1, about 0.4; impurity B, about 0.6; impurity C + D, about 0.7; impurity E, about 0.8; impurity F, about 1.1; and impurity G, about 1.5.
SYSTEM SUITABILITY
The test is not valid unless, in the chromatogram obtained with solution (3), the peak-to-valley ratio is at least 2.0, where Hp is the height above the baseline of the peak due to impurity F and Hv is the height above the baseline of the lowest point of the curve separating this peak from the peak due to itraconazole.
The test is not valid unless, in the chromatogram obtained with solution (2), the signal-to-noise ratio of the principal peak is at least 30.
LIMITS
Identify any peak corresponding to Impurity 1 and multiply the area of this peak by a correction factor of 1.3.
In the chromatogram obtained with solution (1):
the area of any peak due to impurity 1 is not greater than five times the area of the principal peak in the chromatogram obtained with solution (2) (1%);
the area of any peak due to impurity B, impurities C + D, or impurity G is not greater than 1.5 times the area of the principal peak in the chromatogram obtained with solution (2) (0.3% of each);
the area of any other secondary peak is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.2%);
the sum of the areas of any secondary peaks, excluding impurity 1, is not greater than 5 times the area of the principal peak in the chromatogram obtained with solution (2) (1%).
Disregard any peak with an area less than half the area of the principal peak in the chromatogram obtained with solution (2) (0.1%).
ASSAY
Mix the contents of 20 capsules. Mix, and powder if necessary. Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
Solution A: 0.1 volumes of hydrochloric acid and 50 volumes of mobile phase.
(1) To a quantity of capsule contents containing 0.1 g of Itraconazole, add 5 mL of mobile phase and 0.2 mL of hydrochloric acid, and mix with the aid of ultrasound. Add 50 mL of mobile phase and shake. Dilute to produce 100 mL with mobile phase and filter (a 0.45-μm PVDF filter is suitable), discarding the first 6 mL. Dilute 1 volume of the filtrate to 10 volumes with mobile phase.
(2) 0.01% w/v of itraconazole BPCRS in solution A.
(3) 0.1% w/v of itraconazole for system suitability EPCRS in solution A.
CHROMATOGRAPHIC CONDITIONS
The chromatographic conditions described under Related substances may be used with a flow rate of 2 mL per minute.
MOBILE PHASE
45 volumes of 0.01M potassium dihydrogen orthophosphate, adjusted to pH 3.0 with orthophosphoric acid and 55 volumes of acetonitrile.
SYSTEM SUITABILITY
The test is not valid unless, in the chromatogram obtained with solution (3), the peak-to-valley ratio is at least 2.0, where Hp is the height above the baseline of the peak due to impurity F and Hv is the height above the baseline of the lowest point of the curve separating this peak from the peak due to itraconazole.
DETERMINATION OF CONTENT
Calculate the content of C35H38Cl2N8O4 in the capsules using the declared content of C35H38Cl2N8O4 in itraconazole BPCRS.
IMPURITIES
The impurities limited by the requirements of this monograph include impurities listed under Itraconazole and the following: