(Ph. Eur. monograph 0823)
C29H32O13 588.6 33419-42-0
Action and use
Inhibitor of DNA topoisomerase type II; cytotoxic.
Preparations
Etoposide Capsules
Etoposide Infusion
Etoposide Oral Solution
DEFINITION
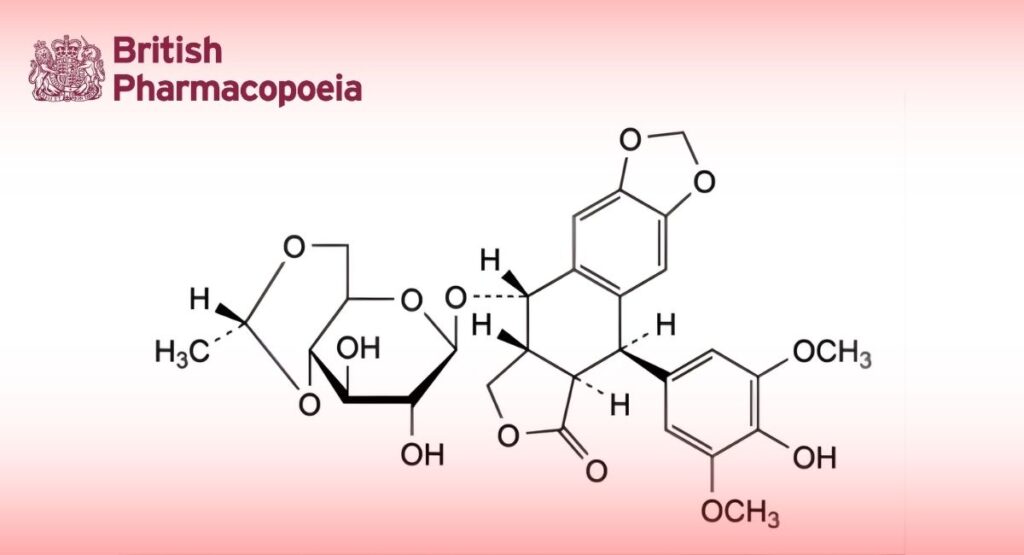
(5R,5aR,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D-glucopyranosyl]oxy]-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9- tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one.
Content
98.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, crystalline powder, slightly hygroscopic.
Solubility
Practically insoluble in water, sparingly soluble in methanol, slightly soluble in ethanol (96 per cent) and in methylene chloride.
IDENTIFICATION
First identification: A, B.
Second identification: C, D.
A. Specific optical rotation (see Tests).
B. Infrared absorption spectrophotometry (2.2.24).
Comparison: etoposide CRS.
C. Thin-layer chromatography (2.2.27).
Test solution: Dissolve 10 mg of the substance to be examined in a mixture of 1 volume of methanol R and 9 volumes of methylene chloride R and dilute to 2 mL with the same mixture of solvents.
Reference solution: Dissolve 10 mg of etoposide CRS in a mixture of 1 volume of methanol R and 9 volumes of methylene chloride R and dilute to 2 mL with the same mixture of solvents.
Plate: silica gel H R as the coating substance.
Mobile phase: water R, glacial acetic acid R, acetone R, methylene chloride R (1.5:8:20:100 V/V/V/V).
Application: 5 μL as bands of 10 mm.
Development: Immediately, over 6/7 of the plate.
Drying: In a current of warm air for 5 min.
Detection: Spray with a mixture of 1 volume of sulfuric acid R and 9 volumes of ethanol (96 per cent) R and heat at 140 °C for 15 min. Cover the plate immediately with a glass plate of the same size. Examine in daylight.
Results The principal zone in the chromatogram obtained with the test solution is similar in position, colour and size to the principal zone in the chromatogram obtained with the reference solution.
D. In a test-tube dissolve about 5 mg in 5 mL of glacial acetic acid R and add 0.05 mL of ferric chloride solution R1. Mix and cautiously add 2 mL of sulfuric acid R. Avoid mixing the 2 layers. Allow to stand for about 30 min; a pink to reddish-brown ring develops at the interface and the upper layer is yellow.
TESTS
Appearance of solution
The solution is clear (2.2.1) and not more intensely coloured than reference solution Y6 or BY6 (2.2.2, Method II).
Dissolve 0.6 g in a mixture of 1 volume of methanol R and 9 volumes of methylene chloride R and dilute to 20 mL with the same mixture of solvents.
Specific optical rotation (2.2.7)
-114 to -106 (anhydrous substance).
Dissolve 50.0 mg in a mixture of 1 volume of methanol R and 9 volumes of methylene chloride R and dilute to 10.0 mL with the same mixture of solvents.
Related substances
Liquid chromatography (2.2.29).
Solvent mixture: Mobile phase A, mobile phase B (50:50 V/V).
Test solution (a): Dissolve 40 mg of the substance to be examined in the solvent mixture and dilute to 10.0 mL with the solvent mixture.
Test solution (b): Dissolve 50.0 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a): Dilute 1.0 mL of test solution (a) to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Reference solution (b): Dissolve 4 mg of etoposide for system suitability CRS (containing impurities B, C, D, E, N and O) in 1.0 mL of the solvent mixture.
Reference solution (c): Dissolve 50.0 mg of etoposide CRS in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Column:
— size: l = 0.125 m, Ø = 4.6 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R (5 μm);
— temperature: 40 °C.
Mobile phase:
— mobile phase A: anhydrous formic acid R, triethylamine R, water for chromatography R (1:1:998 V/V/V);
— mobile phase B: anhydrous formic acid R, triethylamine R, acetonitrile R (1:1:998 V/V/V);
| Time
(min) |
Mobile phase A
(per cent V/V) |
Mobile phase B
(per cent V/V) |
| 0 – 7 | 75 | 25 |
| 7 – 23 | 75 → 27 | 25 → 73 |
Flow rate: 1 mL/min.
Detection: Spectrophotometer at 285 nm.
Injection: 10 μL of test solution (a) and reference solutions (a) and (b).
Identification of impurities: Use the chromatogram supplied with etoposide for system suitability CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities B, C, D, E, N and O.
Relative retention: With reference to etoposide (retention time = about 5 min): impurity D = about 0.4; impurity E = about 0.8; impurity C = about 1.1; impurity B = about 1.2; impurity N = about 3.1; impurity O = about 4.2.
System suitability: Reference solution (b):
— peak-to-valley ratio: minimum 2.0, where Hp = height above the baseline of the peak due to impurity C and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to etoposide; and minimum 3.0, where Hp = height above the baseline of the peak due to impurity B and Hv = height above the
baseline of the lowest point of the curve separating this peak from the peak due to impurity C.
Limits:
— correction factor: for the calculation of content, multiply the peak area of impurity O by 1.7;
— impurities B, C, D, E, N: for each impurity, not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
— impurity O: not more than 1.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.15 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
— total: not more than 10 times the area of the principal peak in the chromatogram obtained with reference solution (a) (1.0 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent); disregard any peak due to the solvent.
Water
(2.5.32)Maximum 6.0 per cent, determined on 0.150 g using the evaporation technique at 130 °C.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection: Test solution (b) and reference solution (c).
System suitability:
— repeatability: maximum relative standard deviation of 1.0 per cent determined on 6 injections of reference solution (c).
Calculate the percentage content of C29H32O13 taking into account the assigned content of etoposide CRS.
STORAGE
In an airtight container.
IMPURITIES
Specified impurities B, C, D, E, N, O.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, F, G, H, I, J, K, L, M, P, Q, R.
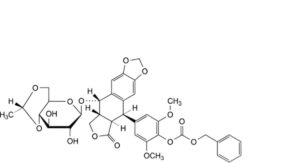
A. (5R,5aR,8aR,9S)-5-[4-[[(benzyloxy)carbonyl]oxy]-3,5-dimethoxyphenyl]-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D- glucopyranosyl]oxy]-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-carbobenzoyloxyethylidene-lignan P),
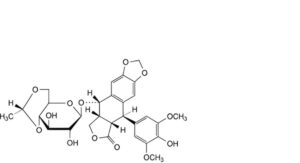
B. (5R,5aS,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D-glucopyranosyl]oxy]-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (picroethylidene-lignan P; cis-etoposide),
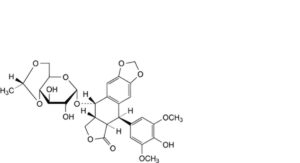
C. (5R,5aR,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-α-D-glucopyranosyl]oxy]-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (α-etoposide),
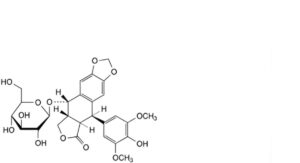
D. (5R,5aR,8aR,9S)-9-(β-D-glucopyranosyloxy)-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro-[5,6-f][1,3]benzodioxol-6(5aH)-one (lignan P),
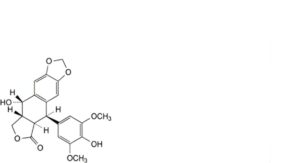
E. (5R,5aR,8aR,9S)-9-hydroxy-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-desmethylepipodophyllotoxin),
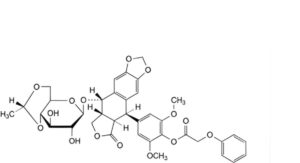
F. (5R,5aR,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D-glucopyranosyl]oxy]-5-[4-[(phenoxyacetyl)oxy]-3,5-dimethoxyphenyl]-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-phenoxyacetyletoposide),
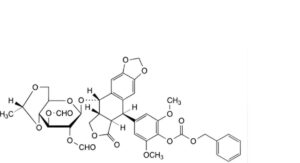
G. (5R,5aR,8aR,9S)-5-[4-[[(benzyloxy)carbonyl]oxy]-3,5-dimethoxyphenyl]-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-2,3-di-O-formyl-β-D-glucopyranosyl]oxy]-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-carbobenzoyloxydiformyl-ethylidene-lignan P),
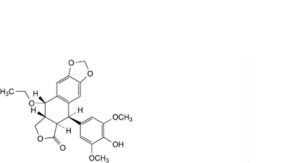
H. (5R,5aR,8aR,9S)-9-ethoxy-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-O-desmethyl-1-O ethylepipodophyllotoxin),
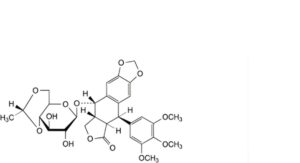
I. (5R,5aR,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D-glucopyranosyl]oxy]-5-(3,4,5-trimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4-O-methylethylidene-lignan P),
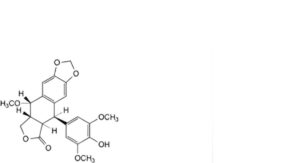
J. (5R,5aR,8aR,9S)-5-(4-hydroxy-3,5-dimethoxyphenyl)-9-methoxy-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-O-desmethyl-1-O-methylepipodophyllotoxin),
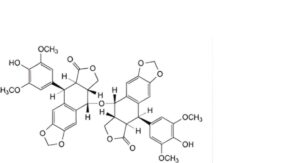
K. 9,9′-oxybis[(5R,5aR,8aR,9S)-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro-[5,6-f][1,3]benzodioxol-6(5aH)-one] (di-4′-O-desmethylepipodophyllotoxin),
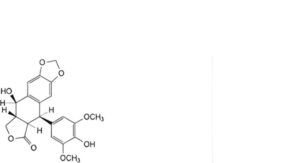
L. (5R,5aR,8aR,9R)-9-hydroxy-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (4′-O-desmethylpodophyllotoxin),
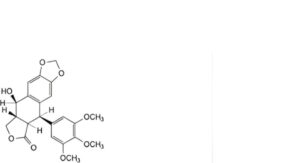
M. (5R,5aR,8aR,9R)-9-hydroxy-5-(3,4,5-trimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (podophyllotoxin),
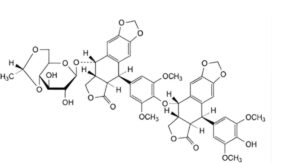
N. (5R,5aR,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D-glucopyranosyl]oxy]-5-[4-[[(5R,5aR,8aR,9S)-5-(4-hydroxy-3,5-dimethoxyphenyl)-6-oxo-5,5a,6,8,8a,9-hexahydro[2]benzofuro[5,6-f][1,3]benzodioxol-9-yl]oxy]-3,5- dimethoxyphenyl]-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one,
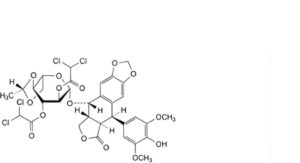
O. (5R,5aR,8aR,9S)-9-[[2,3-bis-O-(dichloroacetyl)-4,6-O-[(1S)-ethane-1,1-diyl]-β-L-glucopyranosyl]oxy]-5-(4-hydroxy-3,5-dimethoxyphenyl)-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one,
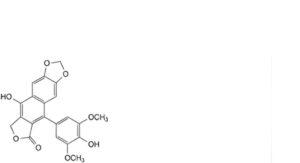
P. 9-hydroxy-5-(4-hydroxy-3,5-dimethoxyphenyl)[2]benzofuro[5,6-f][1,3]benzodioxol-6(8H)-one,
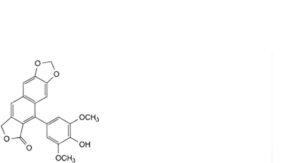
Q. 5-(4-hydroxy-3,5-dimethoxyphenyl)[2]benzofuro[5,6-f][1,3]benzodioxol-6(8H)-one,
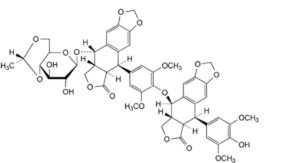
R. (5R,5aR,8aR,9S)-9-[[4,6-O-[(1R)-ethane-1,1-diyl]-β-D-glucopyranosyl]oxy]-5-[4-[[(5R,5aR,8aR,9R)-5-(4-hydroxy-3,5-dimethoxyphenyl)-6-oxo-5,5a,6,8,8a,9-hexahydro[2]benzofuro[5,6-f][1,3]benzodioxol-9-yl]oxy]-3,5-dimethoxyphenyl]-5,8,8a,9-tetrahydro[2]benzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one.