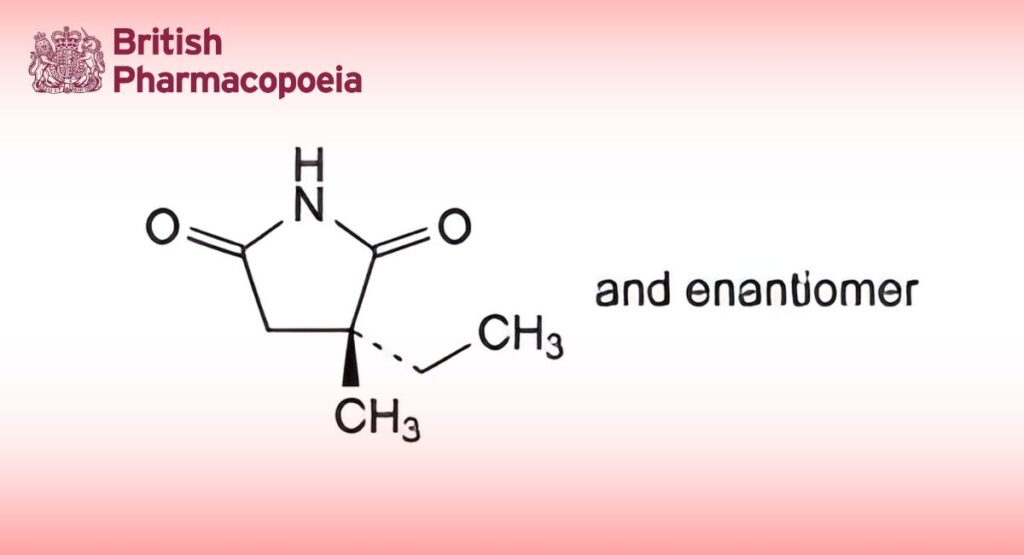
(Ph. Eur. monograph 0764)
C7H11NO2 141.2 77-67-8
Action and use
Antiepileptic.
Preparations
Ethosuximide Capsules
Ethosuximide Oral Solution
DEFINITION
(3RS)-3-Ethyl-3-methylpyrrolidine-2,5-dione.
Content
99.0 per cent to 101.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, powder or waxy solid.
Solubility
Freely soluble in water, very soluble in ethanol (96 per cent) and in methylene chloride.
It shows polymorphism (5.9).
IDENTIFICATION
First identification: A, B.
Second identification: A, C.
A. Melting point (2.2.14): 45 °C to 50 °C.
B. Infrared absorption spectrophotometry (2.2.24).
Comparison: ethosuximide CRS.
If the spectra obtained in the solid state show differences, dissolve the substance to be examined and the reference substance separately in methylene chloride R, evaporate to dryness and record new spectra using the residues.
C. Thin-layer chromatography (2.2.27).
Test solution: Dissolve 20 mg of the substance to be examined in 1 mL of methanol R.
Reference solution: Dissolve 20 mg of ethosuximide CRS in 1 mL of methanol R.
Plate: TLC silica gel F254 plate R.
Mobile phase: glacial acetic acid R, water R, butanol R (17:17:66 V/V/V).
Application: 5 μL.
Development: Over 3/4 of the plate.
Drying: At 100-105 °C for 10 min.
Detection: Examine in ultraviolet light at 254 nm.
Results: The principal spot in the chromatogram obtained with the test solution is similar in position and size to the principal spot in the chromatogram obtained with the reference solution.
TESTS
Appearance of solution
The solution is clear (2.2.1) and colourless (2.2.2, Method II).
Dissolve 2.5 g in water R and dilute to 25 mL with the same solvent.
Cyanide
Liquid chromatography (2.2.29).
Test solution: Dissolve 0.50 g of the substance to be examined in water R and dilute to 10.0 mL with the same solvent.
Reference solution (a): Dissolve 0.125 g of potassium cyanide R in water R and dilute to 50.0 mL with the same solvent. Dilute 1.0 mL of the solution to 100.0 mL with water R. Dilute 0.5 mL of this solution to 10.0 mL with water R.
Reference solution (b): Dissolve 0.50 g of the substance to be examined in water R, add 0.5 mL of reference solution (a) and dilute to 10.0 mL with water R.
Column:
— size: l = 0.075 m, Ø = 7.5 mm,
— stationary phase: spherical weak anion-exchange resin R (10 μm).
Mobile phase: Dissolve 2.1 g of lithium hydroxide R and 85 mg of sodium edetate R in water for chromatography R, and dilute to 1000 mL with the same solvent.
Flow rate: 2.0 mL/min.
Detection: Electrochemical detector (direct amperometry) with a silver working electrode, a silver-silver chloride reference electrode, held at + 0.05 V oxidation potential, and a detector sensitivity of 20 nA full scale.
Injection: 20 μL of the test solution and reference solution (b).
System suitability: Reference solution (b):
— peak-to-valley ratio: minimum 3, where Hp = height above the baseline of the peak due to cyanide and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to ethosuximide.
Limit:
— cyanide: not more than 0.5 times the height of the corresponding peak in the chromatogram obtained with reference solution (b) (0.5 ppm).
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 0.250 g of the substance to be examined in mobile phase A and dilute to 10.0 mL with mobile phase A. Store the solution at room temperature for at least 30 min before injection (in situ transformation of impurity B to impurity A).
Reference solution (a): Dissolve 5.0 mg of ethosuximide impurity A CRS in mobile phase A and dilute to 10.0 mL with mobile phase A.
Reference solution (b): Dilute 1.0 mL of reference solution (a) to 20.0 mL with mobile phase A.
Reference solution (c): Mix 1 mL of reference solution (a) and 4 mL of the test solution.
Column:
— size: l = 0.10 m, Ø = 2.1 mm;
— stationary phase: end-capped solid core octadecylsilyl organosilica polymer compatible with 100 per cent aqueous mobile phases R (2.6 μm);
— temperature: 25 °C.
Mobile phase:
— mobile phase A: 15.6 g/L solution of sodium dihydrogen phosphate R previously adjusted to pH 2.0 with phosphoric acid R;
— mobile phase B: acetonitrile for chromatography R;
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 10 | 90 | 10 |
| 10 – 11 | 90 → 30 | 10 → 70 |
| 11 – 15 | 30 | 70 |
Flow rate: 0.25 mL/min.
Detection: Spectrophotometer at 220 nm.
Injection: 3 μL of the test solution and reference solutions (b) and (c).
Identification of impurities: Use the chromatogram obtained with reference solution (b) to identify the peak due to impurity A.
Relative retention: With reference to ethosuximide (retention time = about 4 min): impurity A = about 1.7.
System suitability: Reference solution (c):
— resolution: minimum 3.0 between the peaks due to ethosuximide and impurity A.
Calculation of percentage contents:
— for each impurity, use the concentration of impurity A in reference solution (b).
Limits:
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 0.2 per cent;
— reporting threshold: 0.05 per cent.
Water (2.5.12)
Maximum 0.5 per cent, determined on 1.00 g.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.120 g in 20 mL of dimethylformamide R and carry out a potentiometric titration (2.2.20) using 0.1 M tetrabutylammonium hydroxide. Protect the solution from atmospheric carbon dioxide throughout the titration. Carry out a blank titration.
1 mL of 0.1 M tetrabutylammonium hydroxide is equivalent to 14.12 mg of C7H11NO2.
STORAGE
Protected from light.
IMPURITIES
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph
Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, B.
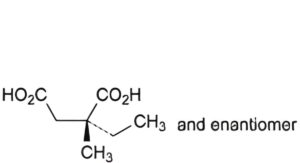
A. (2RS)-2-ethyl-2-methylbutanedioic acid,
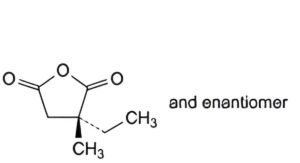
B. (3RS)-3-ethyl-3-methyldihydrofuran-2,5-dione.