Edition: BP 2025 (Ph. Eur. 11.6 update)
General Notices
(Ph. Eur. monograph 3038)
Action and use
Antiarrhythmic
Ph Eur
DEFINITION
Tablets containing Dronedarone hydrochloride (3039), for human use.
They comply with the monograph Tablets (0478) and the following additional requirements.
Content
95.0 per cent to 105.0 per cent of the content of dronedarone (C31H44N2O5S) stated on the label.
IDENTIFICATION
A. Record the UV spectrum of the principal peak in the chromatograms obtained with the solutions used in the assay, with a diode array detector in the range 210-400 nm.
Results The UV spectrum of the principal peak in the chromatogram obtained with test solution (b) is similar to the UV spectrum of the principal peak in the chromatogram obtained with reference solution (c).
B. Examine the chromatograms obtained in the assay.
Results The principal peak in the chromatogram obtained with test solution (b) is similar in retention time and size to the principal peak in the chromatogram obtained with reference solution (c).
TESTS
Related substances
Liquid chromatography (2.2.29).
Solution A To 2.0 mL of triethylamine R add 950 mL of water for chromatography R, adjust to pH 3.0 with phosphoric acid R and dilute to 1000.0 mL with water for chromatography R.
Test solution (a) Crush 20 tablets to obtain a homogeneous powder. Suspend an amount of the powder containing the equivalent of 400 mg of dronedarone in methanol R and dilute to 100.0 mL with the same solvent. Sonicate for 5 min and allow to settle for 10 min. Dilute 5.0 mL of the supernatant to 20.0 mL with the mobile phase and filter.
Test solution (b) Dilute 1.0 mL of test solution (a) to 10.0 mL with the mobile phase.
Reference solution (a) Dilute 1.0 mL of test solution (a) to 100.0 mL with methanol R. Dilute 2.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (b) Dissolve 2 mg of dronedarone impurity A CRS and 2 mg of dronedarone impurity B CRS in methanol R and dilute to 10 mL with the same solvent. Dilute 1 mL of the solution to 20 mL with the mobile phase.
Reference solution (c) Dissolve 53.3 mg of dronedarone hydrochloride CRS in methanol R and dilute to 25.0 mL with the same solvent. Dilute 5.0 mL of the solution to 100.0 mL with the mobile phase.
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: diisopropylcyanosilyl silica gel for chromatography R (5 μm).
Mobile phase acetonitrile for chromatography R, solution A (50:50 V/V).
Flow rate 0.8 mL/min.
Detection Spectrophotometer at 246 nm.
Injection 20 μL of test solution (a) and reference solutions (a) and (b).
Run time 3.5 times the retention time of dronedarone.
Identification of impurities Use the chromatogram obtained with reference solution (b) to identify the peaks due to impurities A and B.
Relative retention With reference to dronedarone (retention time = about 7.4 min): impurity A = about 0.72; impurity B = about 0.77.
System suitability Reference solution (b):
— resolution: minimum 1.5 between the peaks due to impurities A and B.
Calculation of percentage contents:
— for each impurity, use the concentration of dronedarone in reference solution (a).
Limits:
— unspecified impurities: for each impurity, maximum 0.2 per cent;
— total: maximum 0.3 per cent;
— reporting threshold: 0.1 per cent.
Dissolution (2.9.3, Apparatus 2). Use sinker devices.
Dissolution medium Dissolve 13.61 g of potassium dihydrogen phosphate R in 1000 mL of water R. Adjust to pH 4.5 with a 10.3 g/L solution of hydrochloric acid R or a 4 g/L solution of sodium hydroxide R. Use 1000 mL of the medium.
Rotation speed 75 r/min.
Time 30 min and 90 min.
Analysis Ultraviolet and visible absorption spectrophotometry (2.2.25) using a path length of 1 mm.
Test solutions Samples withdrawn from the dissolution vessel and filtered. When a different path length is used, the solutions may be diluted accordingly (e.g. for a path length of 1 cm, 10-fold dilution for 400 mg tablets).
Measure the absorbances of the solutions at the absorption maximum at 288 nm.
Calculate the amount of dissolved dronedarone (C31H44N2O5S), expressed as a percentage of the content stated on the label, taking the specific absorbance to be 315.
Acceptance criteria:
— level 1: 25-50 per cent at 30 min; Q = 80 per cent at 90 min;
— level 2: the average value of the 12 tablets is within the range 25-50 per cent at 30 min and is not less than 80 per cent at 90 min; none of the units is outside the range 15-60 per cent at 30 min and none of the units is less than 65 per cent of the labelled content at 90 min;
— level 3: the average value of the 24 tablets is within the range 25-50 per cent at 30 min and is not less than 80 per cent at 90 min; at 30 min, not more than 2 units are outside the range 15-60 per cent and none is outside the range 5- 70 per cent of the labelled content; at 90 min, not more than 2 units are less than 65 per cent of the labelled content and none is less than 55 per cent of the labelled content.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modifications.
Injection Test solution (b) and reference solution (c).
Detection Spectrophotometer at 288 nm.
Run time Twice the retention time of dronedarone.
System suitability Reference solution (c):
— symmetry factor: maximum 2.1;
— repeatability: maximum relative standard deviation of 1.0 per cent determined on 6 injections.
Calculate the percentage content of dronedarone (C31H44N2O5S) taking into account the assigned content of dronedarone hydrochloride CRS and applying a conversion factor of 0.9385.
IMPURITIES
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph): A, B, C.
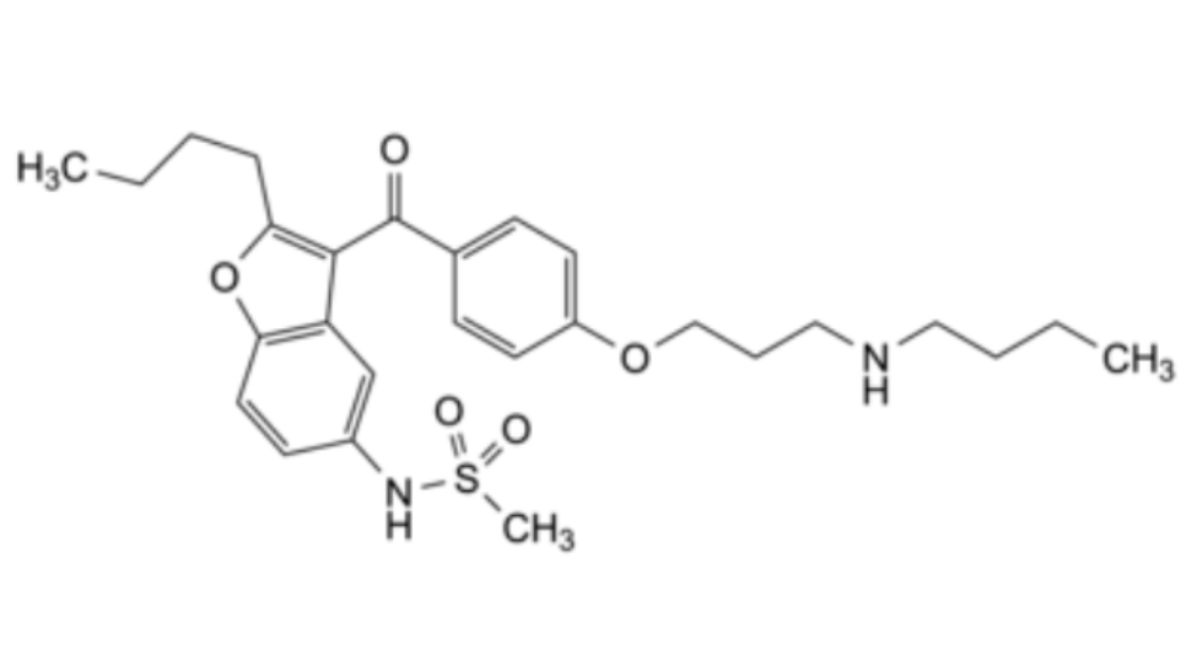
A. N-[2-butyl-3-[4-[3-(butylamino)propoxy]benzoyl]-1-benzofuran-5-yl]methanesulfonamide,
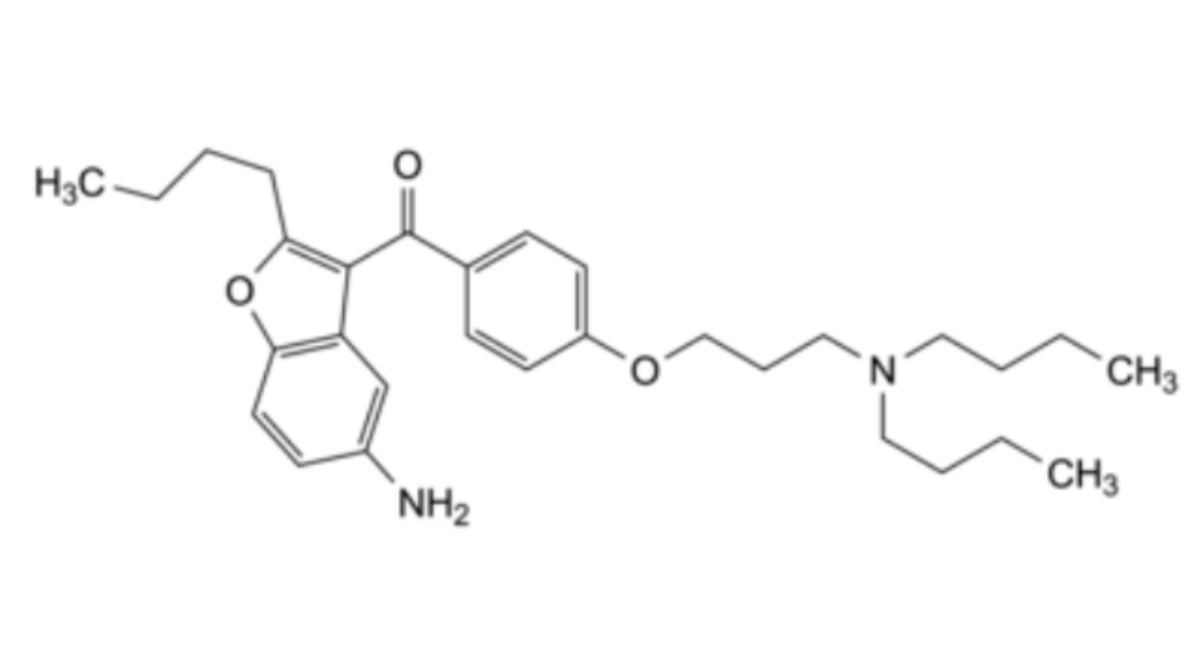
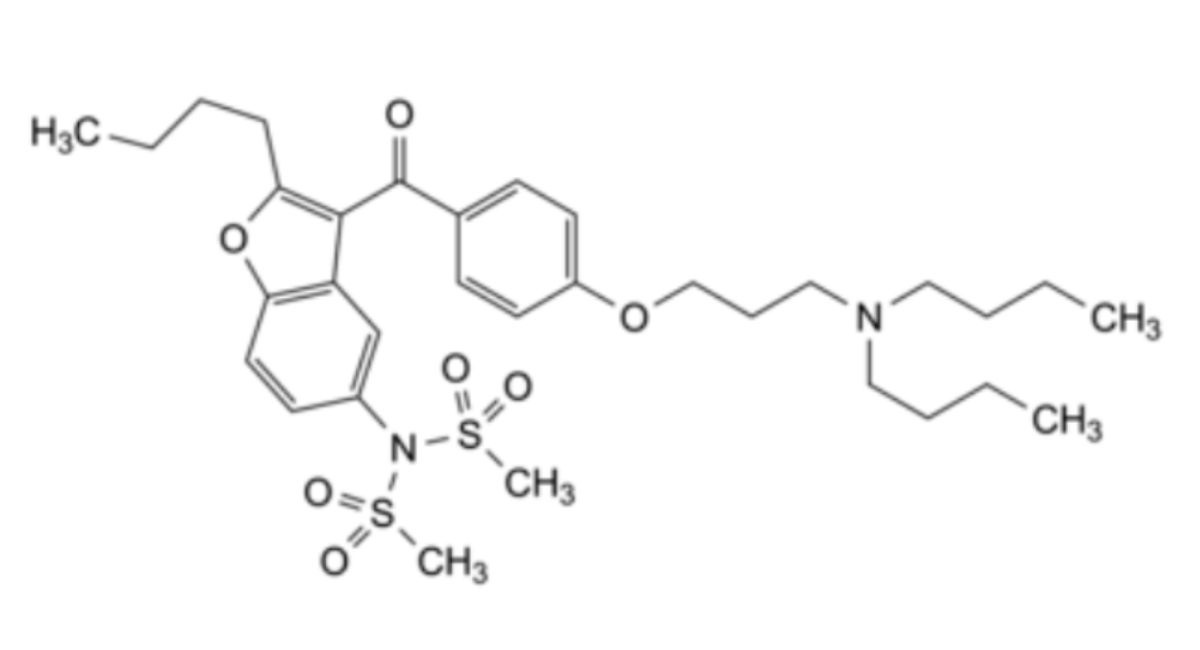
The test approved in the marketing authorisation is to be used for routine quality control to confirm batch-to-batch consistency. For more information please consult Ph. Eur. 1. General Notices.






