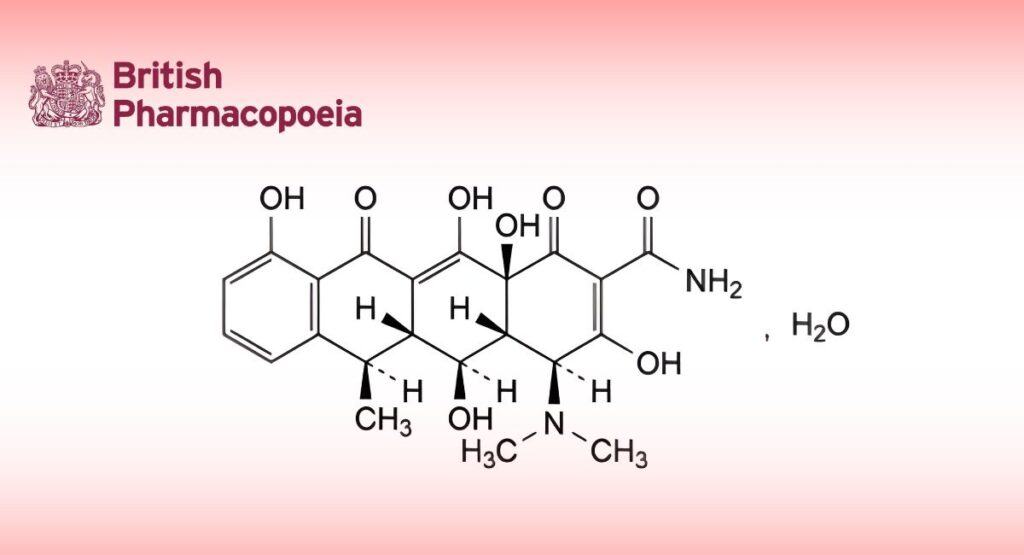
(Ph. Eur. monograph 0820)
C22H24N2O8,H2O 462.5 17086-28-1
Action and use
Tetracycline antibacterial.
Preparations
Doxycycline Dispersible Tablets
Doxycycline Prolonged-release Capsules
DEFINITION
(4S,4aR,5S,5aR,6R,12aS)-4-(Dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide monohydrate.
Substance obtained from oxytetracycline or metacycline or by any other means.
Semi-synthetic product derived from a fermentation product.
Content
95.0 per cent to 102.0 per cent of C22H24N2O8(anhydrous substance).
CHARACTERS
Appearance
Yellow, crystalline powder.
Solubility
Very slightly soluble in water and in ethanol (96 per cent). It dissolves in dilute solutions of mineral acids and in solutions of alkali hydroxides and carbonates.
IDENTIFICATION
A. Examine the chromatograms obtained in the assay.
Results The principal peak in the chromatogram obtained with the test solution is similar in retention time to the principal peak in the chromatogram obtained with reference solution (a).
B. To about 2 mg add 5 mL of sulfuric acid R. A yellow colour develops.
C. Water (see Tests).
TESTS
pH (2.2.3)
5.0 to 6.5.
Suspend 0.1 g in carbon dioxide-free water R and dilute to 10 mL with the same solvent.
Absorbance (2.2.25)
Maximum 0.07 at 490 nm (anhydrous substance).
Dissolve 0.10 g in a mixture of 0.5 volumes of hydrochloric acid R and 99.5 volumes of methanol R and dilute to 10.0 mL with the same mixture of solvents. Carry out the measurement within 1 h of preparing the solution.
Related substances
Liquid chromatography (2.2.29). Prepare the solutions immediately before use.
Solution A: Suspend 111.6 g of sodium edetate R in 900 mL of water R. Adjust to pH 7.0 with concentrated ammonia R to achieve complete dissolution, then dilute to 1 L with water R.
Test solution: Dissolve 20.0 mg of the substance to be examined in a 1 g/L solution of hydrochloric acid R and dilute to 25.0 mL with the same solution.
Reference solution (a): Dissolve 20.0 mg of doxycycline hyclate CRS in a 1 g/L solution of hydrochloric acid R and dilute to 25.0 mL with the same solution.
Reference solution (b): Dissolve 5 mg of doxycycline for system suitability CRS (containing impurities A, B, C and F) in a 1 g/L solution of hydrochloric acid R and dilute to 10 mL with the same solution.
Reference solution (c): Dilute 1.0 mL of the test solution to 100.0 mL with a 1 g/L solution of hydrochloric acid R. Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: end-capped polar-embedded octadecylsilyl amorphous organosilica polymer R (5 μm);
— temperature: 35 °C.
Mobile phase: Mix 13 volumes of acetonitrile R, 17 volumes of water R, 35 volumes of a 67.9 g/L solution of tetrabutylammonium hydrogen sulfate R previously adjusted to pH 7.0 with concentrated ammonia R and 35 volumes of solution A.
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 280 nm.
Injection 20 μL of the test solution and reference solutions (b) and (c).
Run time Twice the retention time of doxycycline.
Identification of impurities Use the chromatogram supplied with doxycycline for system suitability CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities A, B, C and F.
Relative retention With reference to doxycycline (retention time = about 21 min): impurity C = about 0.4;
impurity A = about 0.7; impurity B = about 0.8; impurity F = about 1.3.
System suitability Reference solution (b):
— resolution: minimum 2.0 between the peaks due to impurities A and B; minimum 2.0 between the peaks due to impurity B and doxycycline.
Calculation of percentage contents:
— for each impurity, use the concentration of doxycycline monohydrate in reference solution (c).
Limits:
— impurity A: maximum 2.0 per cent;
— impurity F: maximum 1.2 per cent;
— impurity B: maximum 0.5 per cent;
— impurity C: maximum 0.2 per cent;
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 3.0 per cent;
— reporting threshold: 0.05 per cent.
Water (2.5.12)
3.6 per cent to 4.6 per cent, determined on 0.200 g.
Sulfated ash (2.4.14)
Maximum 0.4 per cent, determined on 1.0 g.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modification.
Injection Test solution and reference solution (a).
Calculate the percentage content of C22H24N2O8 taking into account the assigned content of doxycycline hyclate CRS.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, F.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) D, E.
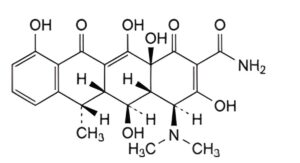
A. (4S,4aR,5S,5aR,6S,12aS)-4-(dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide (6-epidoxycycline),
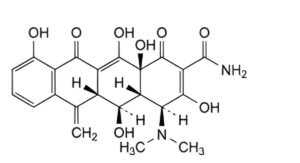
B. (4S,4aR,5S,5aR,12aS)-4-(dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methylene-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide (metacycline),
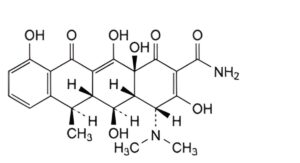
C. (4R,4aR,5S,5aR,6R,12aS)-4-(dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide (4-epidoxycycline),
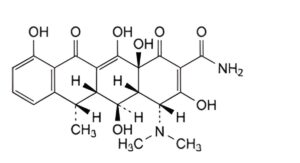
D. (4R,4aR,5S,5aR,6S,12aS)-4-(dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide (4-epi-6-epidoxycycline),
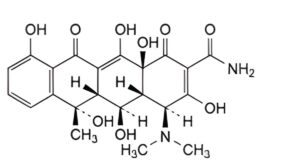
E. (4S,4aR,5S,5aR,6S,12aS)-4-(dimethylamino)-3,5,6,10,12,12a-hexahydroxy-6-methyl-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide (oxytetracycline),
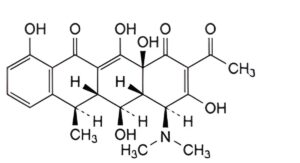
F. (4S,4aR,5S,5aR,6R,12aS)-2-acetyl-4-(dimethylamino)-3,5,10,12,12a-pentahydroxy-6-methyl-4a,5a,6,12a-tetrahydrotetracene-1,11(4H,5H)-dione (2-acetyl-2-decarbamoyldoxycycline).