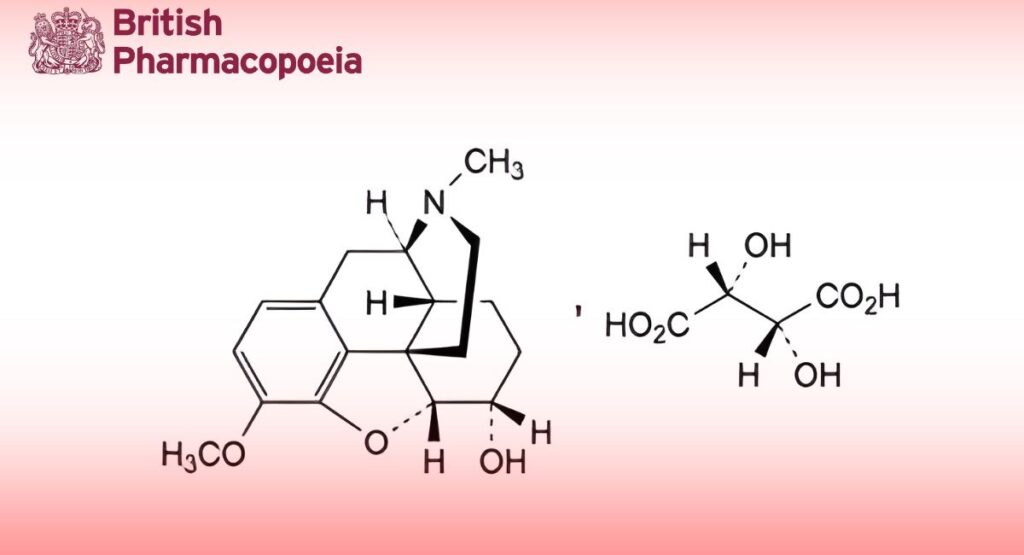
(Dihydrocodeine Hydrogen Tartrate, Ph. Eur. monograph 1776)
C22H29NO9 451.5 5965-13-9
Action and use
Opioid receptor agonist; analgesic.
Preparations
Co-dydramol Tablets
Dihydrocodeine Injection
Dihydrocodeine Oral Solution
Dihydrocodeine Tablets
Dihydrocodeine Prolonged-release Tablets
DEFINITION
4,5α-Epoxy-3-methoxy-17-methylmorphinan-6α-ol hydrogen (2R,3R)-2,3-dihydroxybutanedioate.
Content
98.5 per cent to 101.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Freely soluble in water, sparingly soluble in ethanol (96 per cent), practically insoluble in cyclohexane.
IDENTIFICATION
First identification: A.
Second identification: B, C, D.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison: Ph. Eur. reference spectrum of dihydrocodeine hydrogen tartrate.
B. To about 0.1 g add 1 mL of sulfuric acid R and 0.05 mL of ferric chloride solution R1 and heat on a water-bath. A brownish-yellow colour develops. Add 0.05 mL of dilute nitric acid R. The colour does not become red.
C. To 1 mL of solution S (see Tests) add 5 mL of picric acid solution R. Heat on a water-bath until a clear solution is obtained. Allow to cool. A precipitate is formed. Filter, wash with 5 mL of water R and dry at 100-105 °C. The crystals melt (2.2.14) at 220 °C to 223 °C.
D. It gives reaction (b) of tartrates (2.3.1).
TESTS
Solution S
Dissolve 2.50 g in carbon dioxide-free water R and dilute to 25.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and not more intensely coloured than reference solution BY5 (2.2.2, Method II).
pH (2.2.3)
3.2 to 4.2 for solution S.
Specific optical rotation (2.2.7)
-70.5 to -73.5 (anhydrous substance).
Dilute 10.0 mL of solution S to 20.0 mL with water R.
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 10.0 mg of the substance to be examined in the mobile phase and dilute to 10.0 mL with the mobile phase.
Reference solution (a): Dissolve 2.0 mg of codeine phosphate R in 2.0 mL of the test solution and dilute to 25.0 mL with the mobile phase.
Reference solution (b): Dilute 1.0 mL of the test solution to 200 mL with the mobile phase.
Column:
— size: l = 0.25 m, Ø = 4.6 mm,
— stationary phase: octylsilyl silica gel for chromatography R (5 μm).
Mobile phase: To 1.0 g of sodium heptanesulfonate R, add 10.0 mL of glacial acetic acid R and 4.0 mL of a solution of 5.0 mL of triethylamine R diluted to 25.0 mL with a mixture of equal volumes of water R and acetonitrile R. Add 170 mL of acetonitrile R and dilute to 1000 mL with water R.
Flow rate: 1 mL/min.
Detection: Spectrophotometer at 284 nm.
Injection: 20 μL.
Run time: 5 times the retention time of dihydrocodeine.
Retention time: Dihydrocodeine = about 14 min.
System suitability: Reference solution (a):
— resolution: minimum of 2 between the peaks due to dihydrocodeine and to impurity A.
Limits:
— impurity A: not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.5 per cent),
— any other peak: not more than 0.6 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.3 per cent),
— total: not more than twice the area of the principal peak in the chromatogram obtained with reference solution (b) (1 per cent); disregard any peak due to tartaric acid (relative retention with reference to dihydrocodeine = about 0.25),
— disregard limit: 0.1 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Water (2.5.12)
Maximum 0.7 per cent, determined on 1.00 g.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.350 g in 60 mL of anhydrous acetic acid R. Titrate with 0.1 M perchloric acid determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 45.15 mg of C22H29NO9.
STORAGE
Protected from light.
IMPURITIES
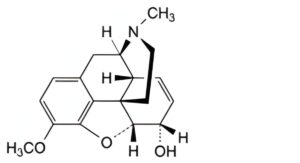
A. 7,8-didehydro-4,5α-epoxy-3-methoxy-17-methylmorphinan-6α-ol (codeine),
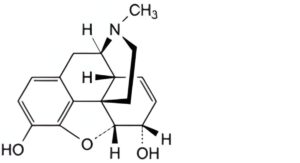
B. 7,8-didehydro-4,5α-epoxy-17-methylmorphinan-3,6α-diol (morphine),
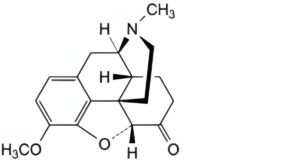
C. 4,5α-epoxy-3-methoxy-17-methylmorphinan-6-one (hydrocodone),
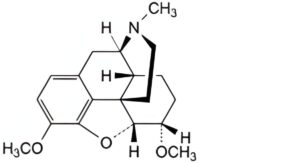
D. 4,5α-epoxy-3,6α-dimethoxy-17-methylmorphinan (tetrahydrothebaine).