Edition: BP 2025 (Ph. Eur. 11.6 update)
Action and use
Sweetening agent.
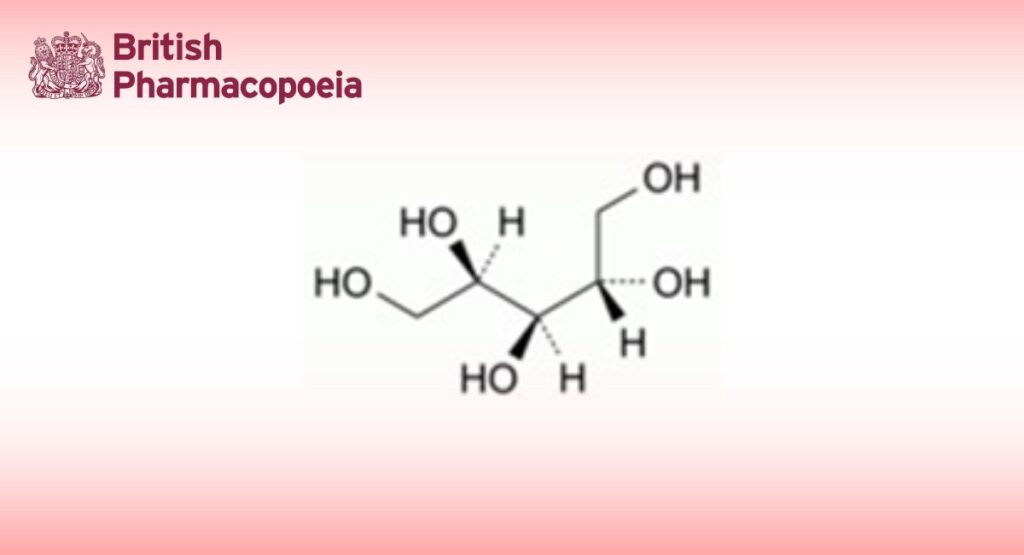
DEFINITION
Meso-xylitol.
Content
98.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, crystalline powder or crystals.
Solubility
Very soluble in water, sparingly soluble in ethanol (96 per cent).
IDENTIFICATION
First identification: B.
Second identification: A, C.
A. Melting point (2.2.14): 92 °C to 96 °C.
B. Infrared absorption spectrophotometry (2.2.24).
Preparation Mulls in liquid paraffin R. Comparison xylitol CRS.
C. Thin-layer chromatography (2.2.27).
Test solution Dissolve 25 mg of the substance to be examined in water R and dilute to 10 mL with the same solvent.
Reference solution (a) Dissolve 25 mg of xylitol CRS in water R and dilute to 10 mL with the same solvent.
Reference solution (b) Dissolve 25 mg of mannitol R and 25 mg of xylitol R in water R and dilute to 10 mL with the same solvent.
Plate TLC silica gel plate R.
Mobile phase water R, ethyl acetate R, propanol R (10:20:70 V/V/V). Application 2 µL.
Development Over 3/4 of the plate.
Drying In air.
Detection Spray with 4-aminobenzoic acid solution R, dry in a current of cold air until the acetone is removed, then heat at 100 °C for 15 min; allow to cool, spray with a 2 g/L solution of sodium periodate R, dry in a current of cold air, then heat at 100 °C for 15 min.
System suitability Reference solution (b):
— the chromatogram shows 2 clearly separated spots.
Results The principal spot in the chromatogram obtained with the test solution is similar in position, colour and size to the principal spot in the chromatogram obtained with reference solution (a).
TESTS
Appearance of solution
The solution is not more opalescent than reference suspension IV (2.2.1) and not more intensely coloured than reference solution BY7 (2.2.2, Method II).
Dissolve 2.5 g in water R and dilute to 50.0 mL with the same solvent.
Conductivity (2.2.38)
Maximum 20 µS·cm-1.
Dissolve 20.0 g in carbon dioxide-free water R prepared from distilled water R and dilute to 100.0 mL with the same solvent. Measure the conductivity of the solution while gently stirring with a magnetic stirrer.
Reducing sugars
Maximum 0.2 per cent, calculated as glucose equivalent.
Dissolve 5.0 g in 6 mL of water R with the aid of gentle heat. Cool and add 20 mL of cupri-citric solution R and a few glass beads. Heat so that boiling begins after 4 min and maintain boiling for 3 min. Cool rapidly and add 100 mL of a 2.4 per cent V/V solution of glacial acetic acid R and 20.0 mL of 0.025 M iodine. With continuous shaking, add 25 mL of a mixture of 6 volumes of hydrochloric acid R and 94 volumes of water R and, when the precipitate has dissolved, titrate the excess of iodine with 0.05 M sodium thiosulfate using 1 mL of starch solution R, added towards the end of the titration, as indicator. Not less than 12.8 mL of 0.05 M sodium thiosulfate is required.
Related substances
Gas chromatography (2.2.28).
Internal standard solution Dissolve 5 mg of erythritol R in water R and dilute to 25.0 mL with the same solvent.
Test solution (a) Dissolve 5.000 g of the substance to be examined in water R and dilute to 100.0 mL with the same solvent.
Test solution (b) Dilute 1.0 mL of test solution (a) to 10.0 mL with water R.
Reference solution (a) Dissolve 5.0 mg each of L-arabinitol CRS (impurity A), galactitol CRS (impurity B), mannitol CRS (impurity C) and sorbitol CRS (impurity D) in water R and dilute to 20.0 mL with the same solvent.
Reference solution (b) Dissolve 50.0 mg of xylitol CRS in water R and dilute to 10.0 mL with the same solvent.
Pipette 1.0 mL of test solutions (a) and (b) and reference solutions (a) and (b) into 4 separate 100 mL round-bottomed flasks. Add 1.0 mL of the internal standard solution to each of the flasks containing test solution (a) or reference solution (a), and 5.0 mL of the internal standard solution to each of the flasks containing test solution (b) or reference solution (b). Evaporate each mixture to dryness in a water-bath at 60 °C by suitable means. Dissolve each dry residue in 1 mL of anhydrous pyridine R, add 1 mL of acetic anhydride R to each flask and boil each solution under reflux for 1 h to complete acetylation.
Column:
— size: l = 30 m, Ø = 0.25 mm;
— stationary phase: cyanopropyl(7)phenyl(7)methyl(86)polysiloxane R (0.25 µm).
Carrier gas nitrogen R. Flow rate 1 mL/min.
Split ratio 1:50 to 1:100.
Temperature:
| Time (min) | Temperature (°C) | |
| Column | 0 – 1 | 170 |
| 1 – 6 | 170 → 230 | |
| 6 – 30 | 230 | |
| Injection port | 250 | |
| Detector | 250 | |
Detection Flame ionisation.
Injection 1 µL of test solution (a) and reference solution (a) (solutions obtained after derivatisation).
Relative retention With reference to xylitol (retention time = about 15 min): internal standard = about 0.6; impurity A = about 0.9; impurity C = about 1.4; impurity B = about 1.45; impurity D = about 1.5.
System suitability Reference solution (a):
— resolution: minimum 2.0 between the peaks due to impurities B and D.
Calculate the percentage content of each related substance in the substance to be examined using the following expression:
100 × (ms/mu) × (Ru/Rs)
ms = mass of the particular component in 1 mL of reference solution (a), in milligrams;
mu = mass of the substance to be examined in 1 mL of test solution (a), in milligrams;
Rs = ratio of the area of the peak due to the particular derivatised component to the area of the peak due to the derivatised internal standard in the chromatogram obtained with reference solution (a);
Ru = ratio of the area of the peak due to the particular derivatised component to the area of the peak due to the derivatised internal standard in the chromatogram obtained with test solution (a).
The sum of the percentage contents of the related substances in the chromatogram obtained with test solution (a) is not greater than 2.0 per cent. Disregard any peak with an area corresponding to a percentage content of 0.05 per cent or less.
Lead (2.4.10)
Maximum 0.5 ppm.
Dissolve the substance to be examined in 150.0 mL of the prescribed mixture of solvents.
Nickel (2.4.15)
Maximum 1 ppm.
Dissolve the substance to be examined in 150.0 mL of the prescribed mixture of solvents.
Water (2.5.12)
Maximum 1.0 per cent, determined on 1.00 g.
Bacterial endotoxins (2.6.14)
Less than 4 IU/g if the concentration is less than 100 g/L of xylitol and less than 2.5 IU/g if the concentration is 100 g/L or more of xylitol, when intended for use in the manufacture of parenteral preparations without a further appropriate procedure for the removal of bacterial endotoxins.
ASSAY
Gas chromatography (2.2.28) as described in the test for related substances with the following modifications.
Injection 1 µL of test solution (b) and reference solution (b) (solutions obtained after derivatisation). Calculate the percentage content of C5H12O5 using the following expression:
T × (mt/mv) × (Rv/Rt)
T = declared percentage content of xylitol CRS;
mt = mass of xylitol CRS in 1 mL of reference solution (b), in milligrams;
mv = mass of the substance to be examined in 1 mL of test solution (b), in milligrams;
Rt = ratio of the area of the peak due to derivatised xylitol to the area of the peak due to the derivatised internal standard in the chromatogram obtained with reference solution (b);
Rv = ratio of the area of the peak due to derivatised xylitol to the area of the peak due to the derivatised internal standard in the chromatogram obtained with test solution (b).
LABELLING
The label states:
— where applicable, the maximum concentration of bacterial endotoxins;
— where applicable, that the substance is suitable for use in the manufacture of parenteral preparations.
IMPURITIES
A. L-arabinitol,
B. meso-galactitol,
C. D-mannitol,
D. D-glucitol (D-sorbitol).