Edition: BP 2025 (Ph. Eur. 11.6 update)
(Ph. Eur. monograph 1377)
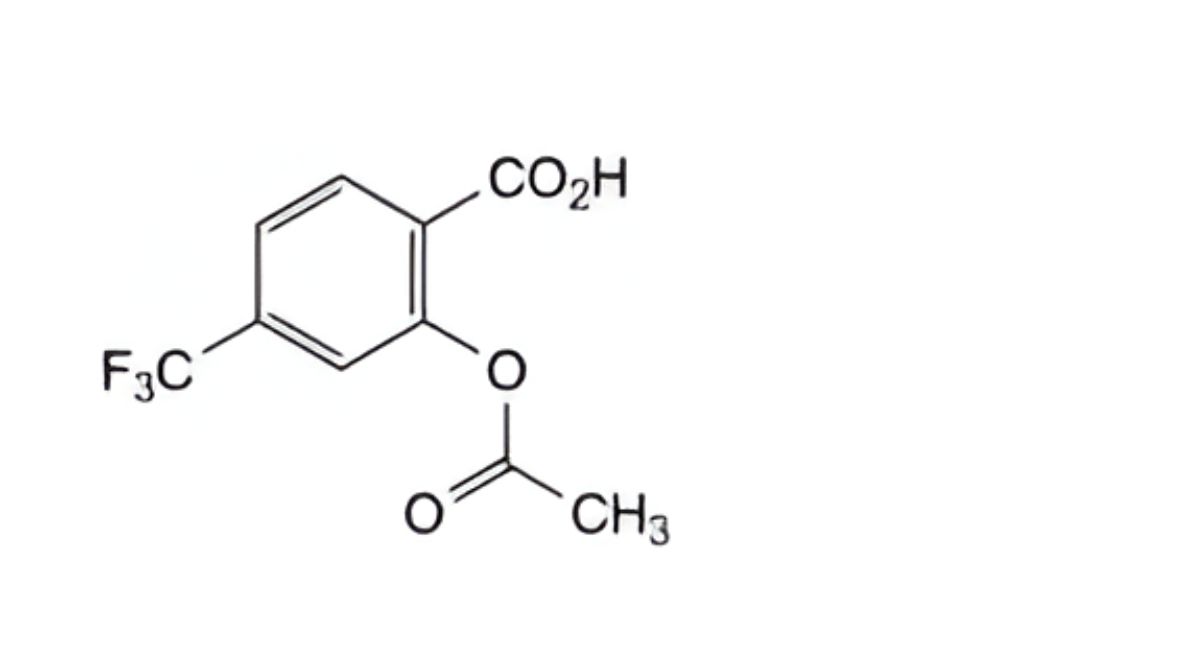
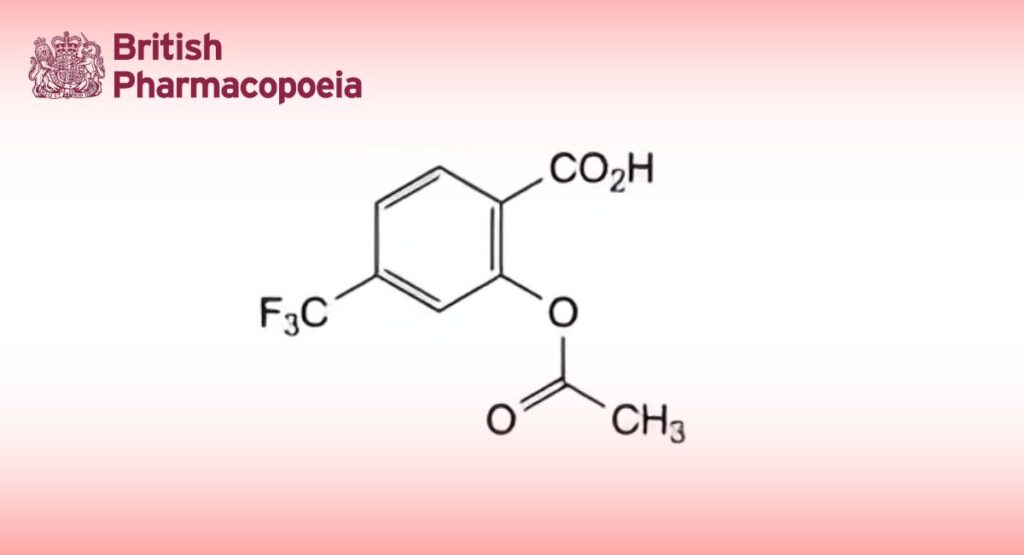
C10H7F3O4 248.2 322-79-2
Action and use
Thromboxane synthesis inhibitor; antiplatelet drug.
Ph Eur
DEFINITION
2-(Acetyloxy)-4-(trifluoromethyl)benzoic acid.
Content
98.5 per cent to 101.5 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Practically insoluble in water, very soluble in anhydrous ethanol, freely soluble in methylene chloride.
mp
About 118 °C, with decomposition.
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Comparison triflusal CRS.
TESTS
Related substances
Liquid chromatography (2.2.29).
Test solution Dissolve 0.200 g of the substance to be examined in acetonitrile R and dilute to 20.0 mL with the same solvent. Prepare the solution immediately before use.
Reference solution (a) Dissolve 5.0 mg of triflusal impurity B CRS in acetonitrile R and dilute to 10.0 mL with the same solvent.
Reference solution (b) Dilute 1.0 mL of reference solution (a) to 25.0 mL with acetonitrile R.
Reference solution (c) Dissolve 2.5 mg of the substance to be examined in acetonitrile R, add 5 mL of reference solution (a) and dilute to 10.0 mL with acetonitrile R. Prepare the solution immediately before use.
Column:
– size: l = 0.15 m, Ø = 4.0 mm;
– stationary phase: end-capped octadecylsilyl silica gel for chromatography R (4-5 μm).
Mobile phase:
– mobile phase A: 0.5 per cent V/V solution of phosphoric acid R;
– mobile phase B: acetonitrile R;
| Time (min) | Mobile phase A (per cent V/V) | Mobile phase B (per cent V/V) |
|---|---|---|
| 0 – 20 | 80 → 30 | 20 → 70 |
| 20 – 25 | 30 | 70 |
Flow rate 1.2 mL/min.
Detection Spectrophotometer at 237 nm.
Injection 10 μL of the test solution and reference solutions (b) and (c).
Identification of impurities Use the chromatogram obtained with reference solution (b) to identify the peak due to impurity B.
Relative retention With reference to triflusal (retention time = about 11 min): impurity B = about 1.2.
System suitability Reference solution (c):
– resolution: minimum 3.0 between the peaks due to triflusal and impurity B.
Limits:
– impurity B: not more than 1.5 times the area of the corresponding peak in the chromatogram obtained with reference solution (b) (0.3 per cent);
– unspecified impurities: for each impurity, not more than 0.5 times the area of the peak due to impurity B in the chromatogram obtained with reference solution (b) (0.10 per cent);
– sum of impurities other than B: not more than 0.5 times the area of the peak due to impurity B in the chromatogram obtained with reference solution (b) (0.1 per cent);
– disregard limit: 0.25 times the area of the peak due to impurity B in the chromatogram obtained with reference solution (b) (0.05 per cent).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in vacuo.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g in a platinum crucible.
ASSAY
Dissolve 0.200 g in 50 mL of anhydrous ethanol R. Titrate with 0.1 M sodium hydroxide, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M sodium hydroxide is equivalent to 24.82 mg of C10H7F3O4.
STORAGE
In an airtight container, at a temperature not exceeding 25 °C.
IMPURITIES
Specified impurities B.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, C, D.
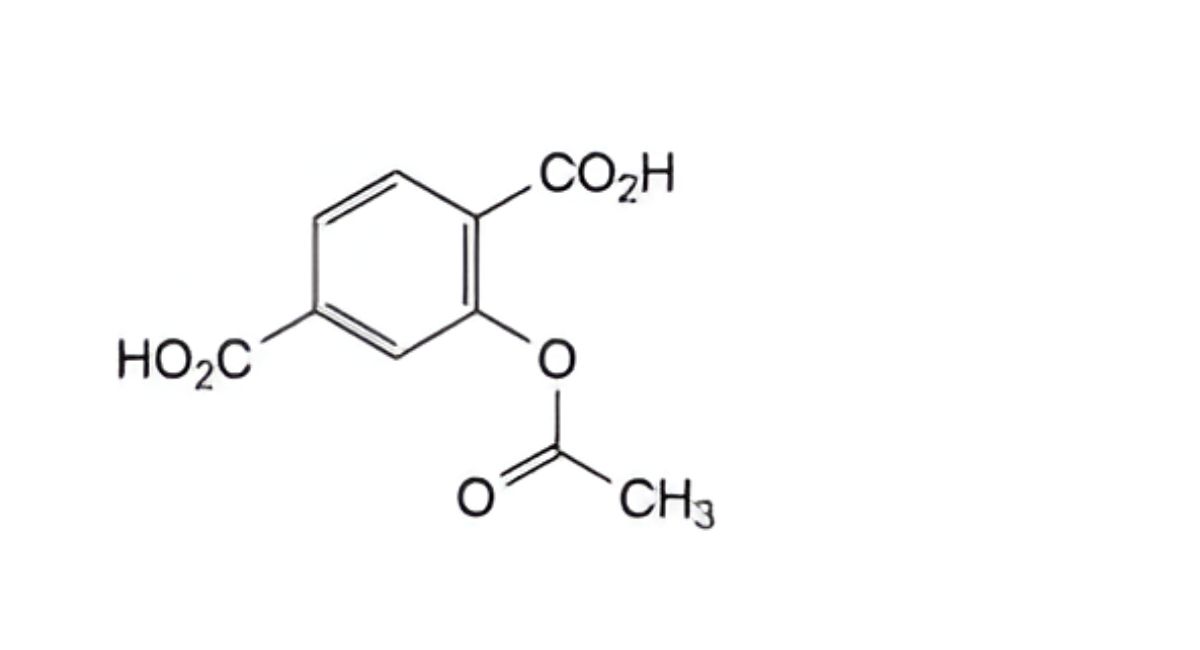
A. 2-(acetyloxy)benzene-1,4-dicarboxylic acid (2-acetoxyterephthalic acid),
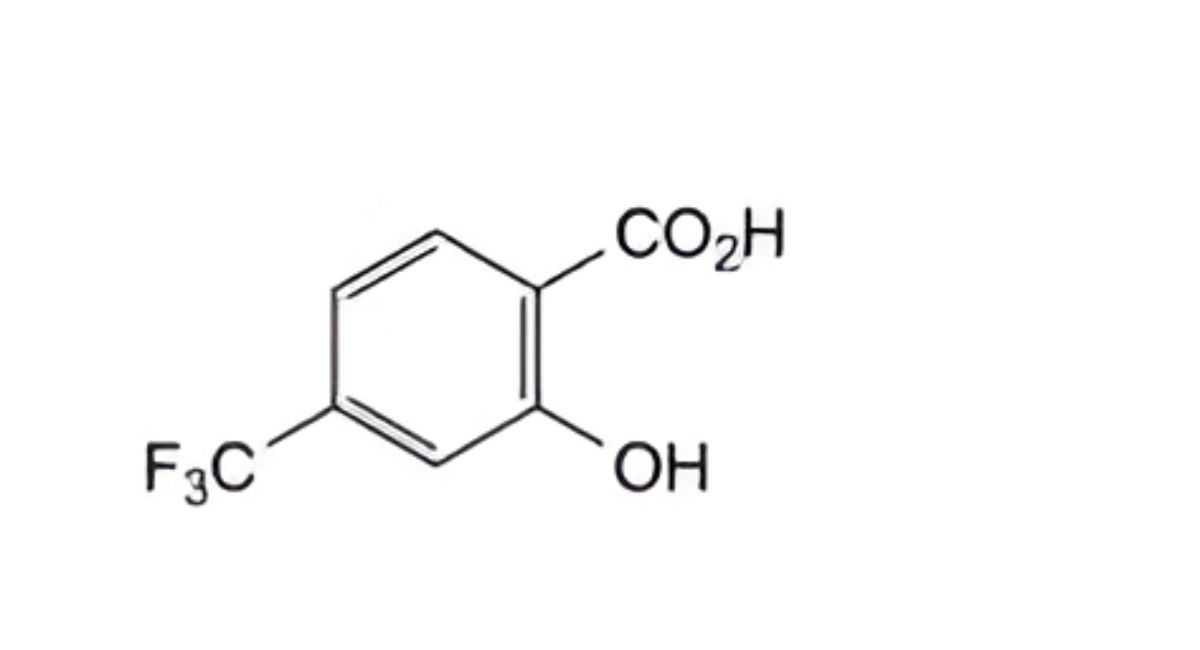
B. 2-hydroxy-4-(trifluoromethyl)benzoic acid (4-(trifluoromethyl)salicylic acid),
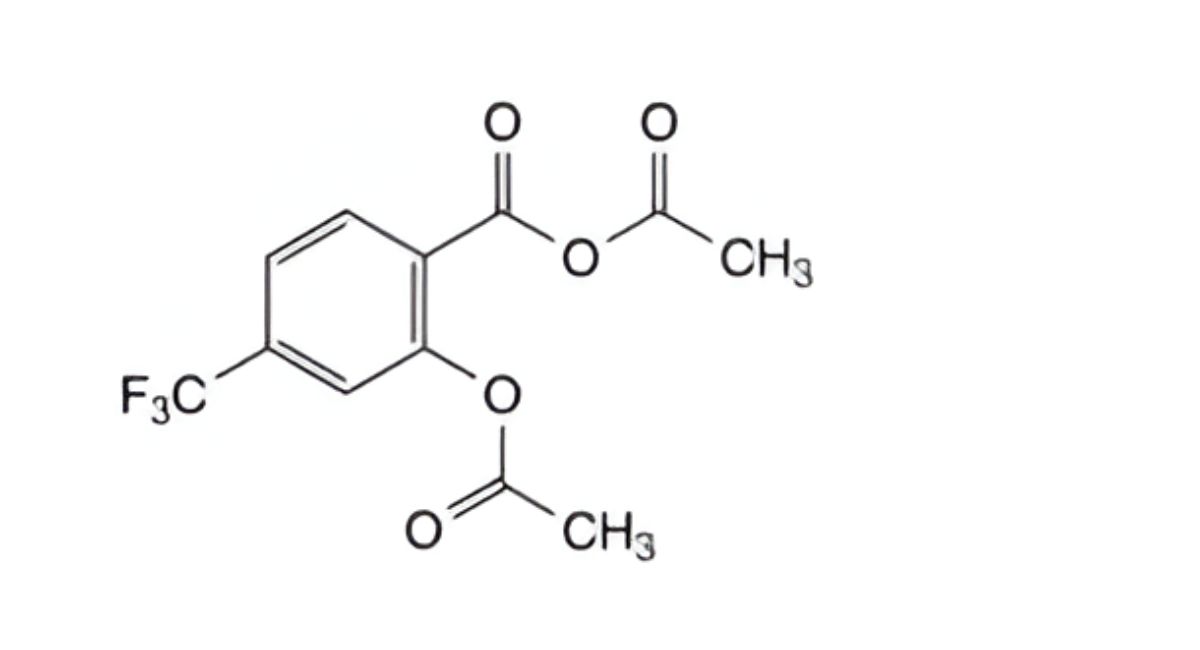
C. acetic 2-(acetyloxy)-4-(trifluoromethyl)benzoic anhydride,
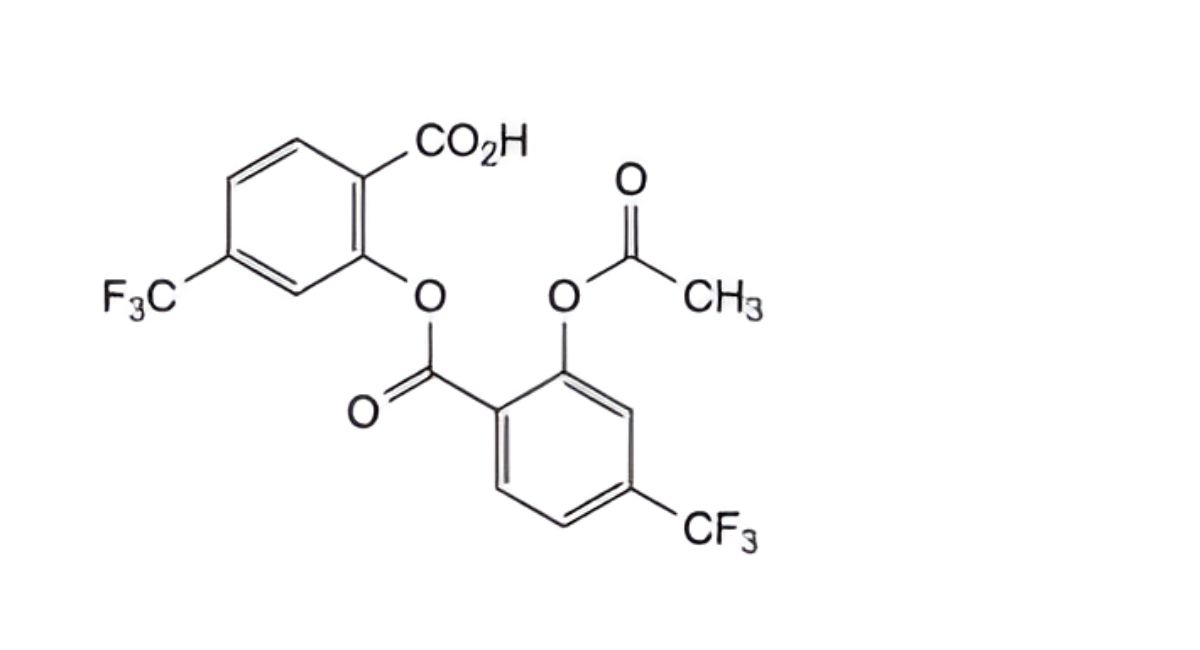
D. 2-[[2-(acetyloxy)-4-(trifluoromethyl)benzoyl]oxy]-4-(trifluoromethyl)benzoic acid.
Ph Eur