Edition: BP 2025 (Ph. Eur. 11.6 update)
Action and use
Anthelminthic.
Preparation
Piperazine Citrate Elixir Ph Eur
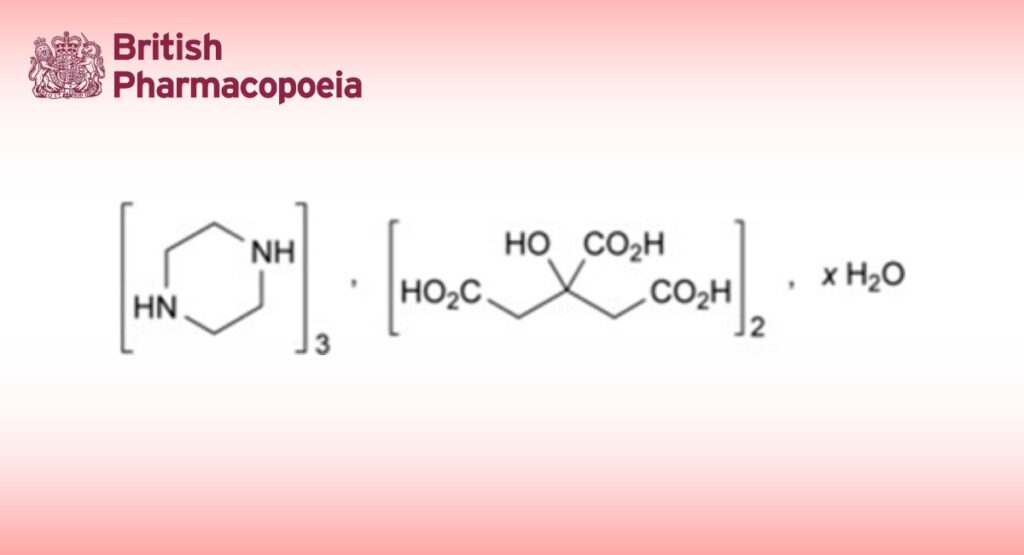
DEFINITION
Piperazine citrate contains not less than 98.0 per cent and not more than the equivalent of 101.0 per cent of tripiperazine bis(2-hydroxy-propane-1,2,3-tricarboxylate), calculated with reference to the anhydrous substance. It contains a variable quantity of water.
CHARACTERS
A white or almost white granular powder, freely soluble in water, practically insoluble in ethanol (96 per cent). After drying at 100 °C to 105 °C, it melts at about 190 °C.
IDENTIFICATION
First identification: A.
Second identification: B, C.
A. Examine by infrared absorption spectrophotometry (2.2.24), comparing with the spectrum obtained with piperazine citrate CRS. Dry the substance to be examined and the reference substance at 120 °C for 5 h, powder the substances avoiding uptake of water, prepare discs and record the spectra without delay.
B. Examine the chromatograms obtained in the test for related substances after spraying with the ninhydrin solutions. The principal spot in the chromatogram obtained with test solution (b) is similar in position, colour and size to the principal spot in the chromatogram obtained with reference solution (a).
C. Dissolve 0.5 g in water R and dilute to 5 mL with the same solvent. The solution gives the reaction of citrates (2.3.1).
TESTS
Solution S
Dissolve 1.25 g in water R and dilute to 25 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and not more intensely coloured than reference solution B8 (2.2.2, Method II).
Related substances
Examine by thin-layer chromatography (2.2.27), using a suitable silica gel as the coating substance.
Test solution (a) Dissolve 1.0 g of the substance to be examined in 6 mL of concentrated ammonia R and dilute to 10 mL with anhydrous ethanol R.
Test solution (b) Dilute 1 mL of test solution (a) to 10 mL with a mixture of 2 volumes of anhydrous ethanol R and 3 volumes of concentrated ammonia R.
Reference solution (a) Dissolve 0.1 g of piperazine citrate CRS in a mixture of 2 volumes of anhydrous ethanol R and 3 volumes of concentrated ammonia R and dilute to 10 mL with the same mixture of solvents.
Reference solution (b) Dissolve 25 mg of ethylenediamine R in a mixture of 2 volumes of anhydrous ethanol R and 3 volumes of concentrated ammonia R and dilute to 100 mL with the same mixture of solvents.
Reference solution (c) Dissolve 25 mg of triethylenediamine R in a mixture of 2 volumes of anhydrous ethanol R and 3 volumes of concentrated ammonia R and dilute to 100 mL with the same mixture of solvents.
Reference solution (d) Dissolve 12.5 mg of triethylenediamine R in 5.0 mL of test solution (a) and dilute to 50 mL with a mixture of 2 volumes of anhydrous ethanol R and 3 volumes of concentrated ammonia R.
Apply separately to the plate 5 µL of each solution. Develop over a path of 15 cm using a freshly prepared mixture of 20 volumes of concentrated ammonia R and 80 volumes of acetone R. Dry the plate at 105 °C and spray successively with a 3 g/L solution of ninhydrin R in a mixture of 3 volumes of anhydrous acetic acid R and 100 volumes of butanol R and a 1.5 g/L solution of ninhydrin R in anhydrous ethanol R. Dry the plate at 105 °C for 10 min. Any spot in the chromatogram obtained with test solution (a), apart from the principal spot, is not more intense than the spot in the chromatogram obtained with reference solution (b) (0.25 per cent). Spray the plate with 0.05 M iodine and allow to stand for about 10 min. Any spot corresponding to triethylenediamine in the chromatogram obtained with test solution (a) is not more intense than the spot in the chromatogram obtained with reference solution (c) (0.25 per cent). The test is not valid unless the chromatogram obtained with reference solution (d) shows 2 clearly separated spots. Disregard any spots remaining on the line of application.
Water (2.5.12)
10.0 per cent to 14.0 per cent, determined on 0.300 g by the semi-micro determination of water.
Sulfated ash (2.4.14)
Not more than 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.100 g in 10 mL of anhydrous acetic acid R with gentle heating and dilute to 70 mL with the same acid. Titrate with 0.1 M perchloric acid using 0.25 mL of naphtholbenzein solution R as indicator until the colour changes from
brownish-yellow to green.
1 mL of 0.1 M perchloric acid is equivalent to 10.71 mg of C24H46N6O14.
Ph Eur