Edition: BP 2025 (Ph. Eur. 11.6 update)
Action and use
Treatment of influenza.
Preparation
Paediatric Oseltamivir Oral Solution Ph Eur
DEFINITION
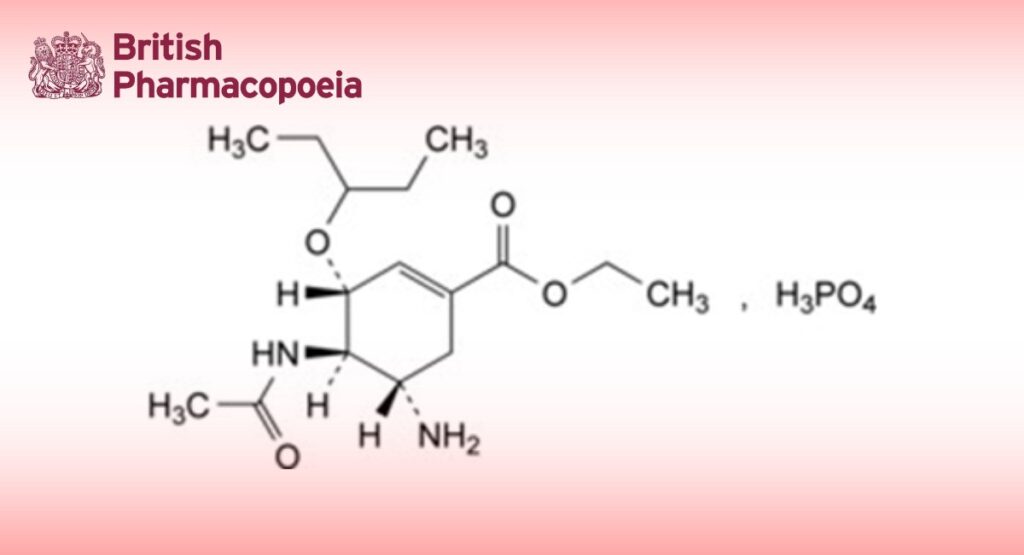
Ethyl (3R,4R,5S)-4-acetamido-5-amino-3-(1-ethylpropoxy)cyclohex-1-ene-1-carboxylate phosphate.
Content
98.0 per cent to 102.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white powder.
Solubility
Freely soluble in water and in methanol, practically insoluble in methylene chloride. It shows polymorphism (5.9).
IDENTIFICATION
A. Specific optical rotation (see Tests).
B. Infrared absorption spectrophotometry (2.2.24).
Comparison oseltamivir phosphate (impurity B-free) CRS.
If the spectra obtained show differences, dissolve the substance to be examined and the reference substance separately in methanol R, evaporate to dryness and record new spectra using the residues.
C. Dissolve 200 mg in 10 mL of water R. It gives reaction (b) of phosphates (2.3.1).
TESTS
Specific optical rotation (2.2.7)
-30.7 to -32.6 (anhydrous substance), measured at 25 °C.
Dissolve 0.50 g in water R and dilute to 50.0 mL with the same solvent.
Impurity B
Liquid chromatography (2.2.29) coupled with mass spectrometry (2.2.43).
Test solution Dissolve 0.100 g of the substance to be examined in water for chromatography R and dilute to 10.0 mL with the same solvent.
Reference solution (a) Dissolve 2.5 mg of oseltamivir impurity B CRS in 5.0 mL of anhydrous ethanol R and dilute to 50.0 mL with water for chromatography R. Dilute 2.0 mL of the solution to 100.0 mL with water for chromatography R.
Reference solution (b) Dissolve 50.0 mg of oseltamivir phosphate (impurity B-free) CRS in reference solution (a) and dilute to 5.0 mL with the same solution.
Column:
— size: l = 0.05 m, Ø = 3.0 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R (5 µm);
— temperature: 40 °C.
Mobile phase Mix 10 volumes of a 1.54 g/L solution of ammonium acetate R in water for chromatography R, 30 volumes of acetonitrile R1 and 60 volumes of water for chromatography R.
Flow rate 1.5 mL/min.
Post-column split ratio Use a split ratio suitable for the mass detector (e.g. 1:3).
Detection:
— mass detector: the following settings have been found to be suitable and are given as examples; if the detector has different setting parameters, adjust the detector settings so as to comply with the system suitability criterion:
— ionisation: ESI-positive;
— detection m/z: 356.2;
— dwell: 580 ms;
— gain EMV: 1;
— fragmentator voltage: 120 V;
— gas temperature: 350 °C;
— drying gas flow: 13 L/min,
— nebuliser pressure: 345 kPa;
— capillary voltage (Vcap): 3 kV.
Injection 1 µL of the test solution and reference solution (b).
Run time 3 min.
System suitability Reference solution (b):
— repeatability: maximum relative standard deviation of 15 per cent determined on 6 injections.
Limit:
— impurity B: not more than the area of the corresponding peak in the chromatogram obtained with reference solution (b) (100 ppm).
Impurity H
Gas chromatography (2.2.28).
Silylation reagent Mix 1.0 mL of chlorotrimethylsilane R, 2.0 mL of hexamethyldisilazane R and 10.0 mL of anhydrous pyridine R.
Test solution Introduce 15.0 mg of the substance to be examined into a 2 mL vial and add 1.0 mL of the silylation reagent. Close the vial, shake and heat at 60 °C for 20 min. Centrifuge and discard the precipitate.
Reference solution Introduce 15.0 mg of oseltamivir impurity H CRS into a 2 mL vial and add 1.0 mL of anhydrous pyridine R. Close the vial and shake (solution A). (Note: impurity H is hygroscopic.) Introduce
15.0 mg of the substance to be examined into another 2 mL vial and add 1.0 mL of the silylation reagent. Close the vial, shake and heat at 60 °C for 20 min.
Centrifuge and discard the precipitate (solution B). Introduce 10.0 µL of solution A and 10.0 µL of solution B into a volumetric flask and dilute to 10.0 mL with anhydrous pyridine R.
Column:
— material: fused silica;
— size: l = 30 m, Ø = 0.32 mm;
— stationary phase: methylpolysiloxane R (film thickness 0.25 µm).
Carrier gas helium for chromatography R. Flow rate 1.2 mL/min.
Split ratio 1:50.
Temperature:
| Time (min) | Temperature (°C) | |
| Column | 0 – 2 | 180 |
| 2 – 11 | 180 → 250 | |
| 11 – 21 | 250 | |
| Injection port | 260 | |
| Detector | 260 | |
Detection Flame ionisation.
Injection 1 µL.
Relative retention With reference to oseltamivir phosphate (retention time = about 10 min): impurity H = about 0.5.
System suitability Reference solution:
— repeatability: maximum relative standard deviation of 5 per cent for the peak due to impurity H after 6 injections.
Limit:
— impurity H: not more than 1.5 times the area of the corresponding peak in the chromatogram obtained with the reference solution (0.15 per cent).
Related substances
Liquid chromatography (2.2.29).
Solvent mixture acetonitrile R1, methanol R2, water for chromatography R (135:245:620 V/V/V).
Test solution Dissolve 50.0 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Reference solution (b) Dissolve 5 mg of oseltamivir impurity A CRS and 5.0 mg of oseltamivir impurity C CRS in the solvent mixture and dilute to 50.0 mL with the solvent mixture. Dilute 1.0 mL of the solution to 10.0 mL with the solvent mixture.
Reference solution (c) Dissolve 50.0 mg of oseltamivir phosphate (impurity B-free) CRS in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: end-capped octylsilyl silica gel for chromatography R (5 µm);
— temperature: 50 °C.
Mobile phase Mix 135 volumes of acetonitrile R1, 245 volumes of methanol R2 and 620 volumes of a 6.8 g/L solution of potassium dihydrogen phosphate R in water for chromatography R, adjusted to pH 6.0 with a 1 M potassium hydroxide solution prepared from potassium hydroxide R.
Flow rate 1.2 mL/min.
Detection Spectrophotometer at 207 nm.
Injection 15 µL of the test solution and reference solutions (a) and (b).
Run time Twice the retention time of oseltamivir phosphate.
Relative retention With reference to oseltamivir phosphate (retention time = about 17 min): impurity A = about 0.16; impurity C = about 0.17.
System suitability Reference solution (b):
— resolution: minimum 1.5 between the peaks due to impurities A and C.
Limits:
— impurity C: not more than 0.3 times the area of the corresponding peak in the chromatogram obtained with reference solution (b) (0.3 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
— total: not more than 7 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.7 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Water (2.5.12)
Maximum 0.5 per cent, determined on 0.500 g.
ASSAY
Liquid chromatography (2.2.29) as described in the test for related substances with the following modification.
Injection Test solution and reference solution (c).
Calculate the percentage content of C16H31N2O8P from the declared content of oseltamivir phosphate (impurity B-free) CRS.
STORAGE
Protected from light.
IMPURITIES
Specified impurities B, C, H.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, D, E, F, G.
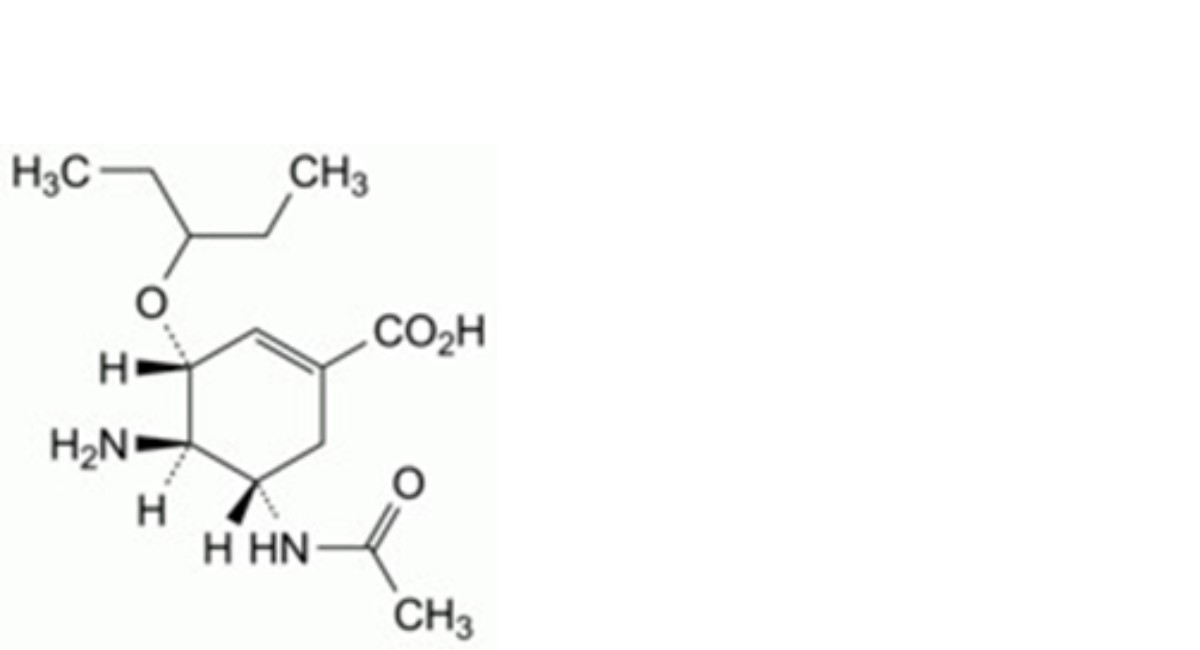
A. (3R,4R,5S)-5-acetamido-4-amino-3-(1-ethylpropoxy)cyclohex-1-ene-1-carboxylic acid,
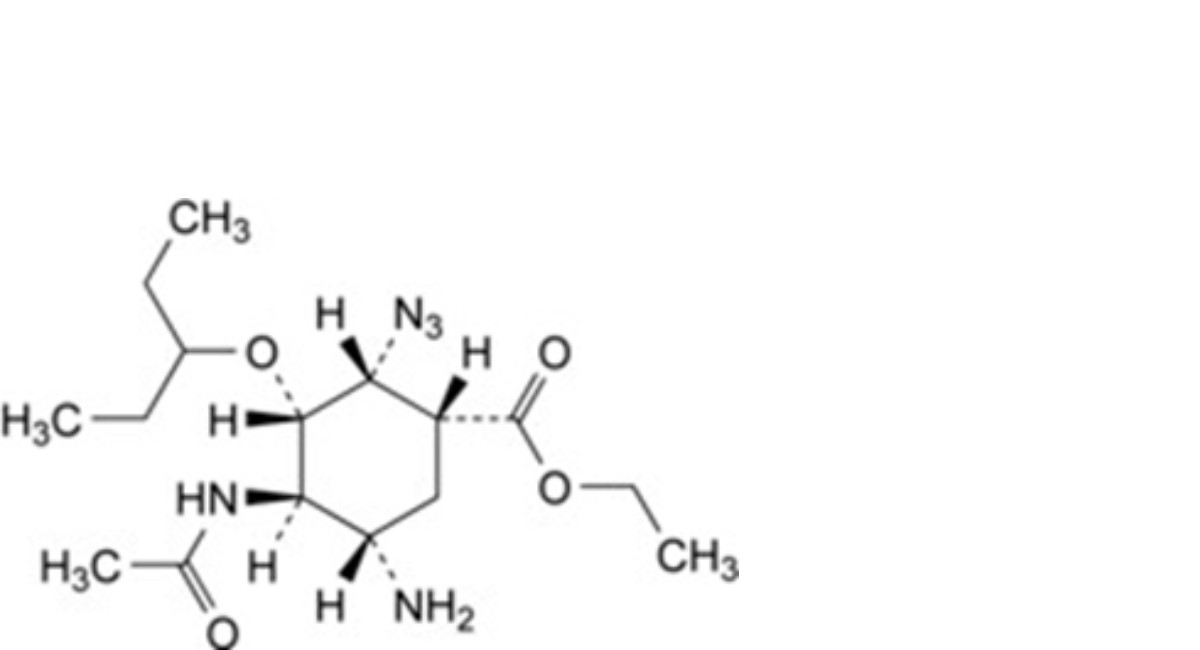
B. ethyl (1R,2R,3S,4R,5S)-4-acetamido-5-amino-2-azido-3(1-ethylpropoxy)cyclohexanecarboxylate,
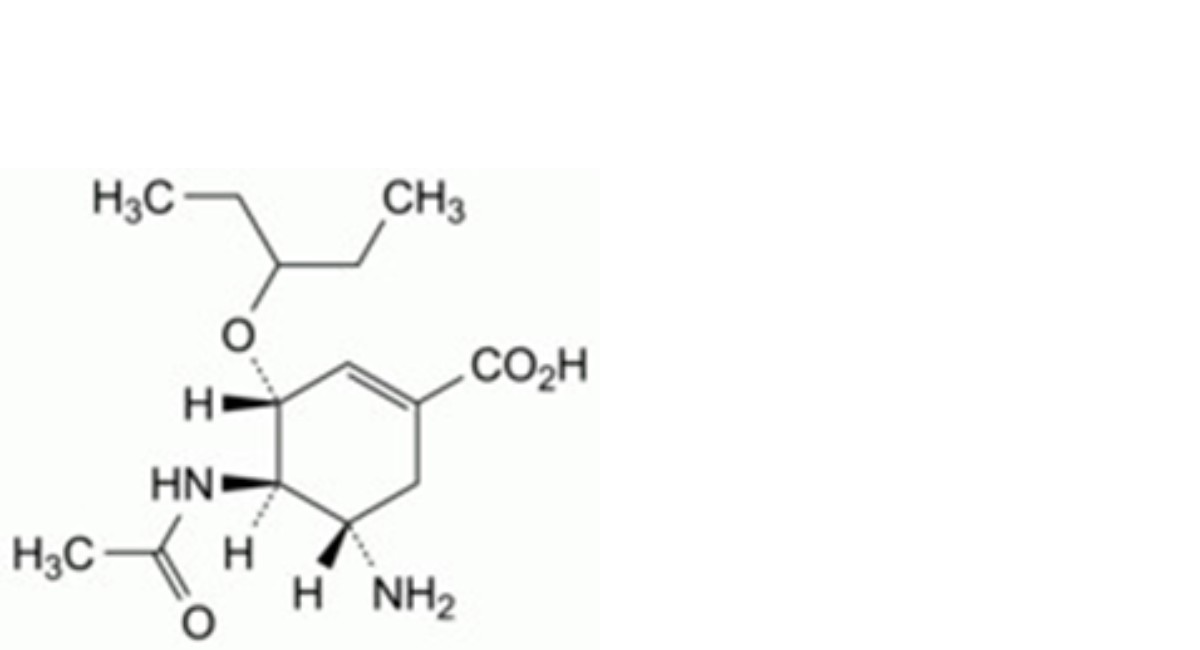
C. (3R,4R,5S)-4-acetamido-5-amino-3-(1-ethylpropoxy)cyclohex-1-ene-1-carboxylic acid,
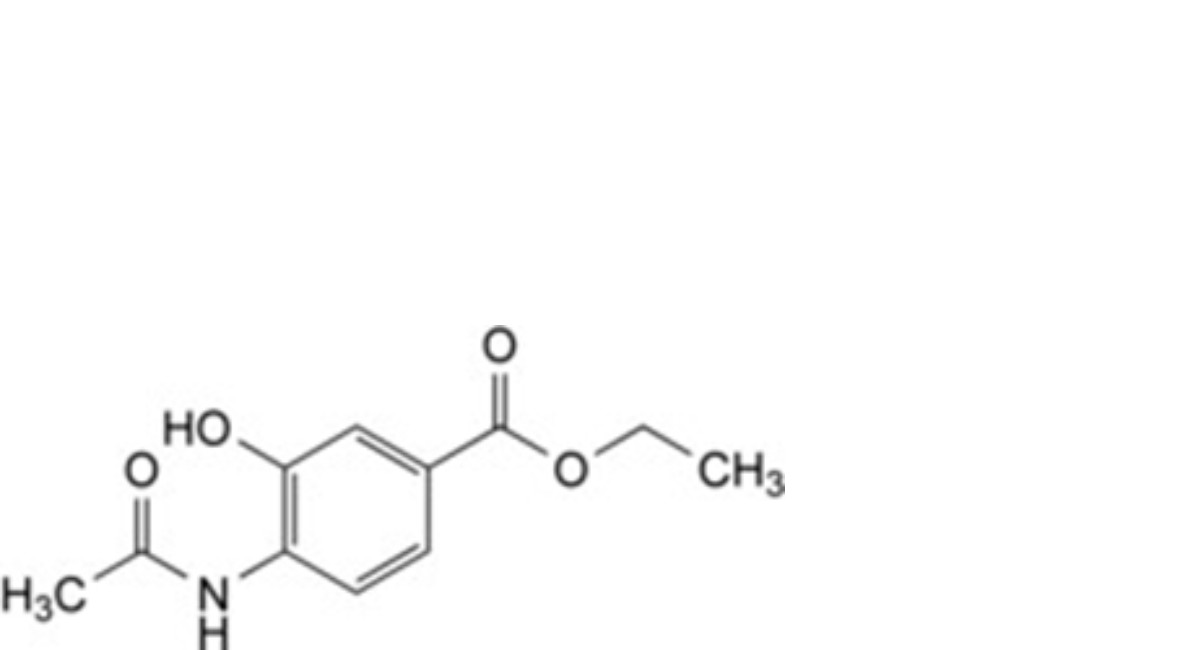
D. ethyl 4-acetamido-3-hydroxybenzoate,
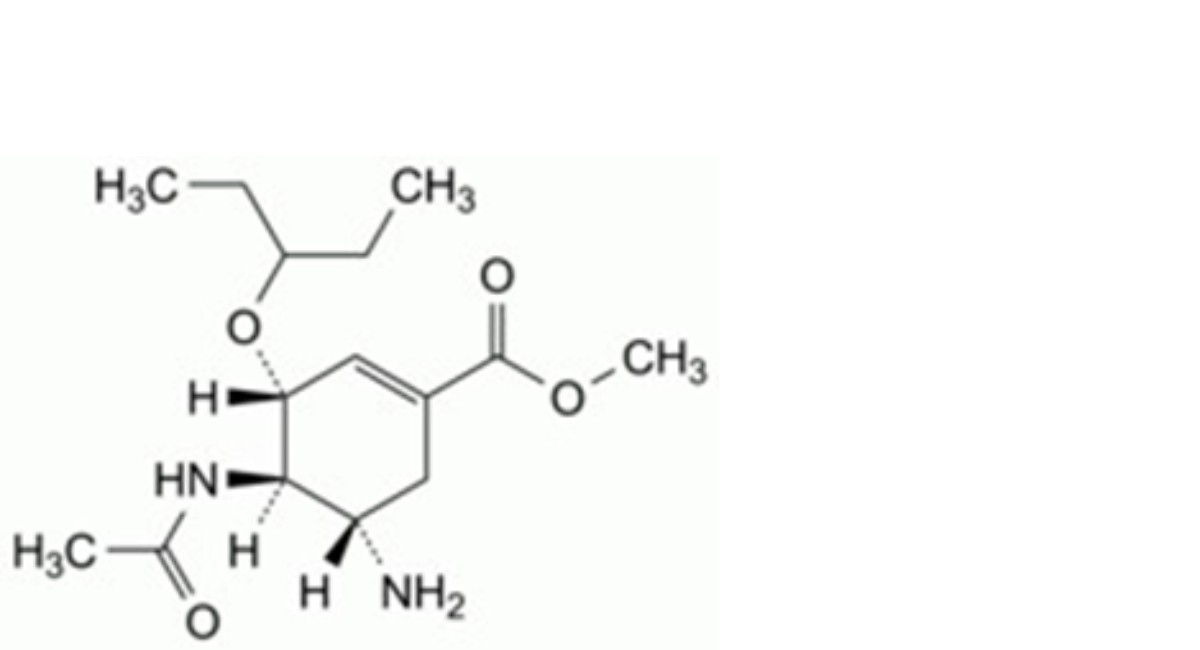
E. methyl (3R,4R,5S)-4-acetamido-5-amino-3-(1-ethylpropoxy)cyclohex-1-ene-1-carboxylate,
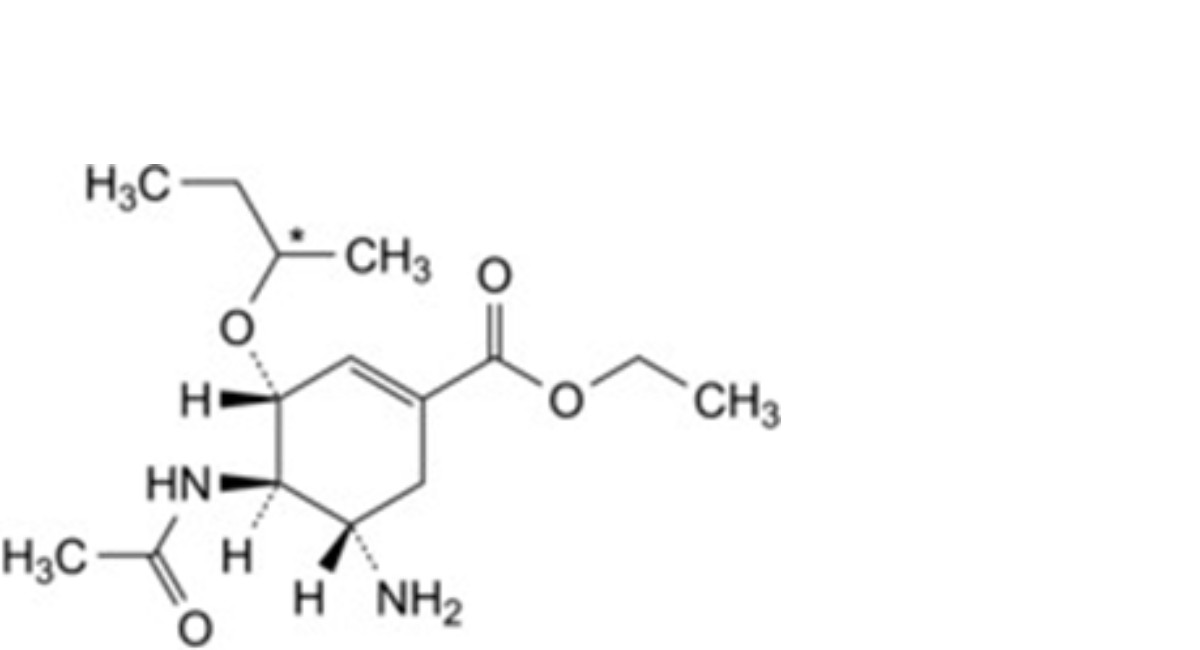
F. ethyl (3R,4R,5S)-4-acetamido-5-amino-3-(1-methylpropoxy)cyclohex-1-ene-1-carboxylate,
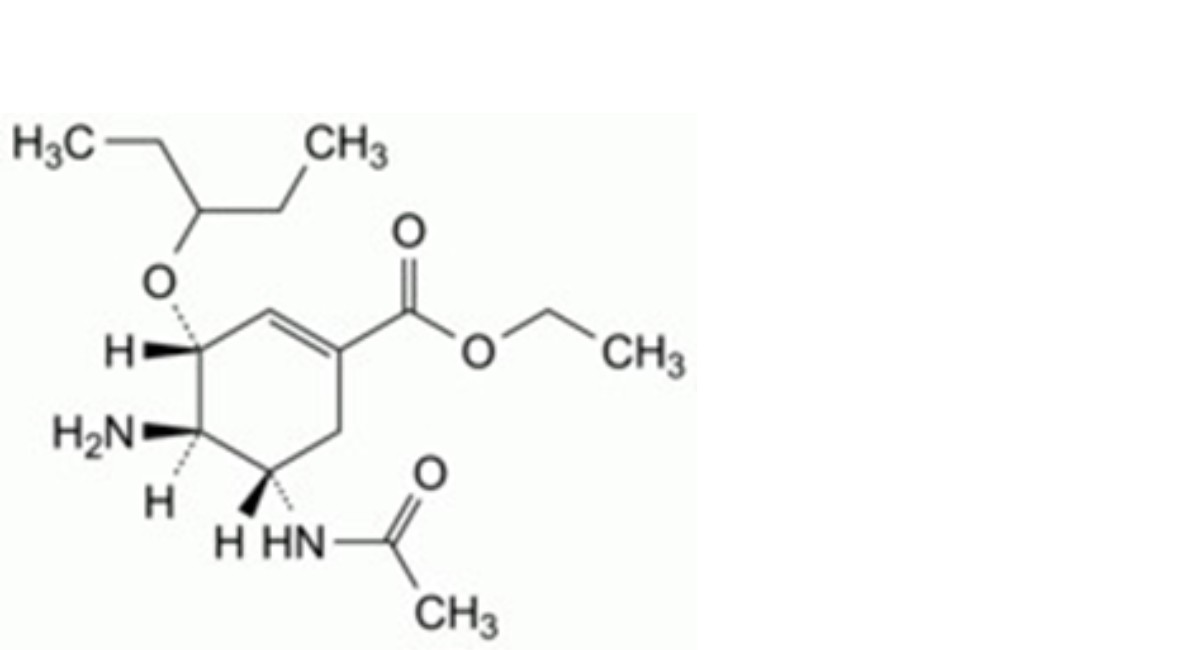
G. ethyl (3R,4R,5S)-5-acetamido-4-amino-3-(1-ethylpropoxy)cyclohex-1-ene-1-carboxylate,
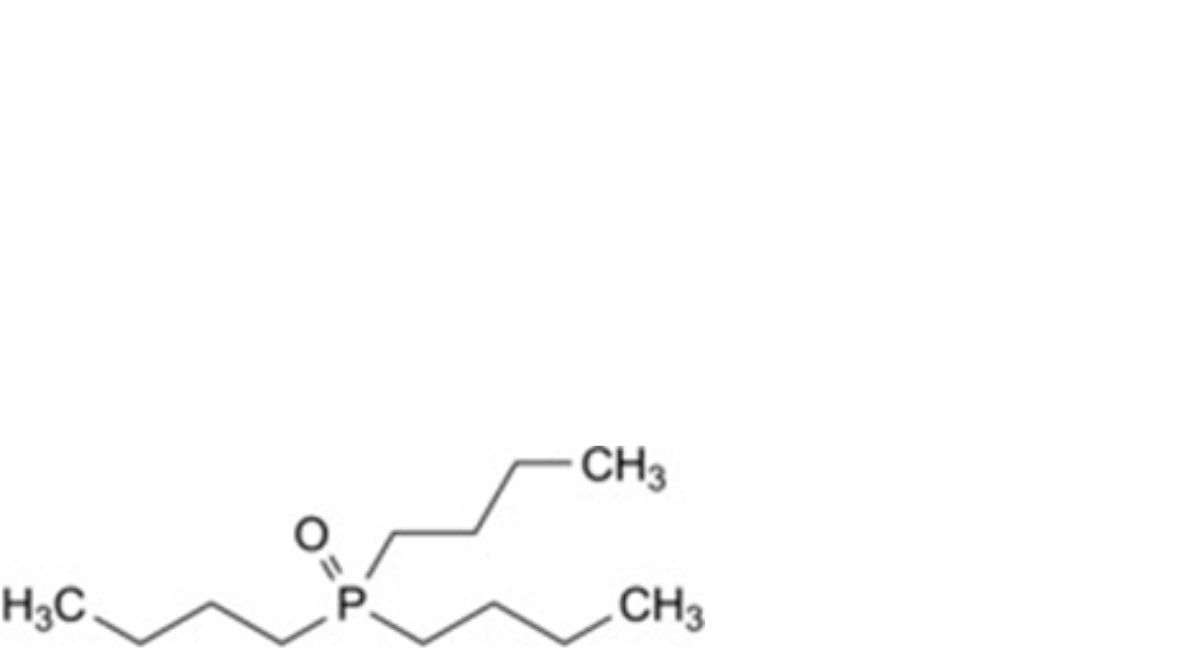
H. tributylphosphane oxide.
Ph Eur