(Ph. Eur. monograph 1545)
C6H12N4 140.2 100-97-0
Action and use
Anti-infective.
DEFINITION
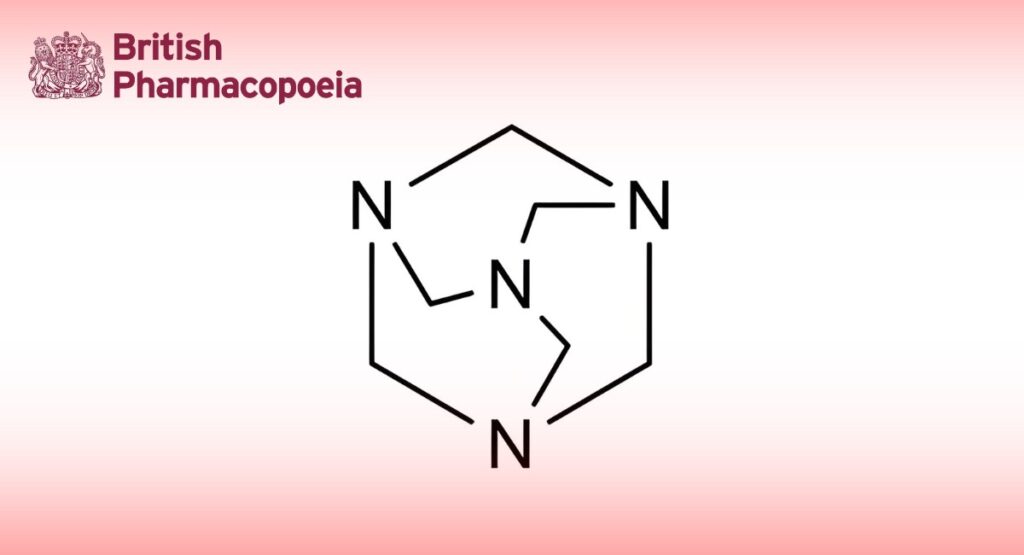
1,3,5,7-Tetraazotricyclo[3.3.1.13,7]decane.
Content
99.0 per cent to 100.5 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder or colourless crystals.
Solubility
Freely soluble in water, soluble in ethanol (96 per cent) and in methylene chloride.
IDENTIFICATION
First identification: A.
Second identification: B, C, D.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison methenamine CRS.
B. To 1 mL of solution S (see Tests) add 1 mL of sulfuric acid R and immediately heat to boiling. Allow to cool. To 1 mL of the solution add 4 mL of water R and 5 mL of acetylacetone reagent R1. Heat on a water-bath for 5 min. An intense yellow colour develops.
C. To 1 mL of solution S add 1 mL of dilute sulfuric acid R and immediately heat to boiling. The solution gives the reaction of ammonium salts and salts of volatile bases (2.3.1).
D. Dissolve 10 mg in 5 mL of water R and acidify with dilute hydrochloric acid R. Add 1 mL of potassium iodobismuthate solution R. An orange precipitate is formed immediately.
TESTS
Solution S
Dissolve 10.0 g in carbon dioxide-free water R prepared from distilled water R and dilute to 100 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and colourless (2.2.2, Method II).
Acidity or alkalinity
To 5 mL of solution S add 0.1 mL of phenolphthalein solution R. Not more than 0.2 mL of 0.1 M hydrochloric acid or 0.1 M sodium hydroxide is required to change the colour of the indicator.
Free formaldehyde
Maximum 50 ppm.
Dissolve 0.8 g in water R and dilute to 8 mL with the same solvent. Add 2 mL of ammoniacal silver nitrate solution R. After 5 min, any grey colour in the solution is not more intense than that in a standard prepared at the same time and in the same manner with a mixture of 8 mL of freshly prepared formaldehyde standard solution (5 ppm CH2O) R and 2 mL of ammoniacal silver nitrate solution R.
Chlorides (2.4.4)
Maximum 100 ppm.
Dilute 5 mL of solution S to 15 mL with water R.
Sulfates (2.4.13)
Maximum 100 ppm, determined on solution S.
Ammonium (2.4.1)
Maximum 50 ppm.
Dilute 2 mL of freshly prepared solution S to 13 mL with water R. Add 2 mL of dilute sodium hydroxide solution R.
Loss on drying (2.2.32)
Maximum 2.0 per cent, determined on 1.000 g by drying in a desiccator.
ASSAY
Dissolve 0.100 g in 30 mL of methanol R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 14.02 mg of C6H12N4.
STORAGE
Protected from light.