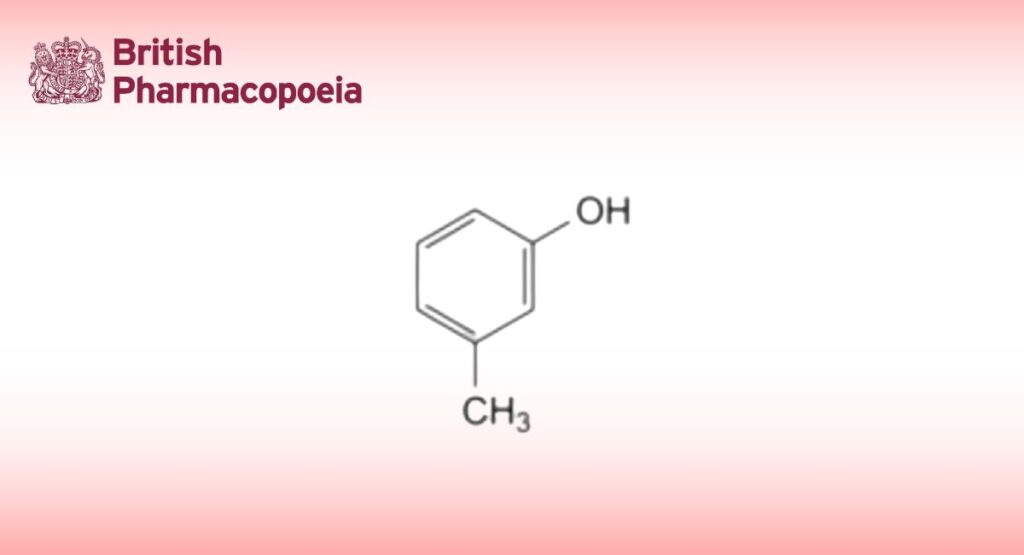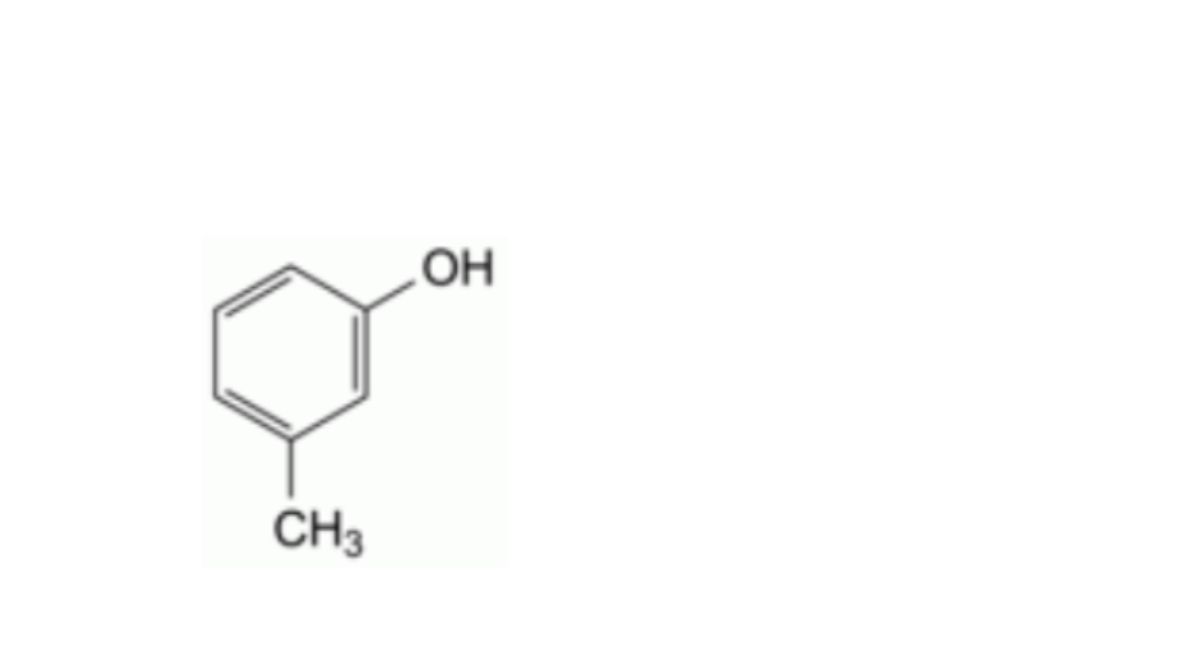
C7H8O 108.1 108-39-4
Action and use
Antiseptic; antimicrobial preservative.
DEFINITION
3-Methylphenol.
CHARACTERS
Appearance
Colourless or yellowish liquid.
Solubility
Sparingly soluble in water, miscible with ethanol (96 per cent) and with methylene chloride.
Relative density
About 1.03.
mp
About 11 °C.
bp
About 202 °C.
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24).
Comparison Ph. Eur. reference spectrum of metacresol.
TESTS
Solution S
Dissolve 1.5 g in carbon dioxide-free water R and dilute to 100 mL with the same solvent.
Appearance of solution
Freshly prepared solution S is not more opalescent than reference suspension III (2.2.1) and not more intensely coloured than reference solution BY7 (2.2.2, Method II).
Acidity
To 25 mL of solution S add 0.15 mL of methyl red solution R. The solution is red. Not more than 0.5 mL of 0.01 M sodium hydroxide is required to change the colour of the indicator to yellow.
Related substances
Gas chromatography (2.2.28): use the normalisation procedure.
Test solution Dissolve 1.00 g of the substance to be examined in methanol R and dilute to 100.0 mL with the same solvent.
Reference solution (a) Dissolve 0.10 g of cresol R, 0.10 g of p-cresol R and 0.10 g of the substance to be examined in methanol R and dilute to 20.0 mL with the same solvent.
Reference solution (b) Dilute 1.0 mL of the test solution to 100.0 mL with methanol R. Dilute 1.0 mL of this solution to 20.0 mL with methanol R.
Column:
— material: fused silica,
— size: l = 25 m, Ø = 0.25 mm,
— stationary phase: cyanopropyl(25)phenyl(25)methyl(50)polysiloxane R (0.2 μm).
Carrier gas helium for chromatography R.
Flow rate 1.8 mL/min.
Split ratio 1:30.
Temperature:
| Time (min) |
Temperature (°C) |
|
| Column | 0 – 35 | 100 |
| 35 – 40 | 100 → 150 | |
| 40 – 50 | 150 | |
| Injection port | 200 | |
| Detector | 200 |
Detection Flame ionisation.
Injection 1.0 μL.
Relative retention With reference to metacresol (retention time = about 28 min): impurity B = about 0.75; impurity C = about 0.98.
System suitability Reference solution (a):
— resolution: minimum 1.4 between the peaks due to impurity C and metacresol.
Limits:
— impurities B, C: for each impurity, not more than 0.5 per cent,
— any other impurity: for each impurity, not more than 0.1 per cent,
— total: not more than 1.0 per cent.
— disregard limit: the area of the peak due to metacresol in the chromatogram obtained with reference solution (b) (0.05 per cent).
Residue on evaporation
Maximum 0.1 per cent.
Evaporate 2.0 g to dryness on a water-bath in a fume cupboard and dry at 100-105 °C for 1 h. The residue weighs a maximum of 2 mg.
STORAGE
In an airtight container, protected from light.
IMPURITIES
Specified impurities B, C.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10.
Control of impurities in substances for pharmaceutical use) A, D, E, F, G, H, I, J, K, L, M.
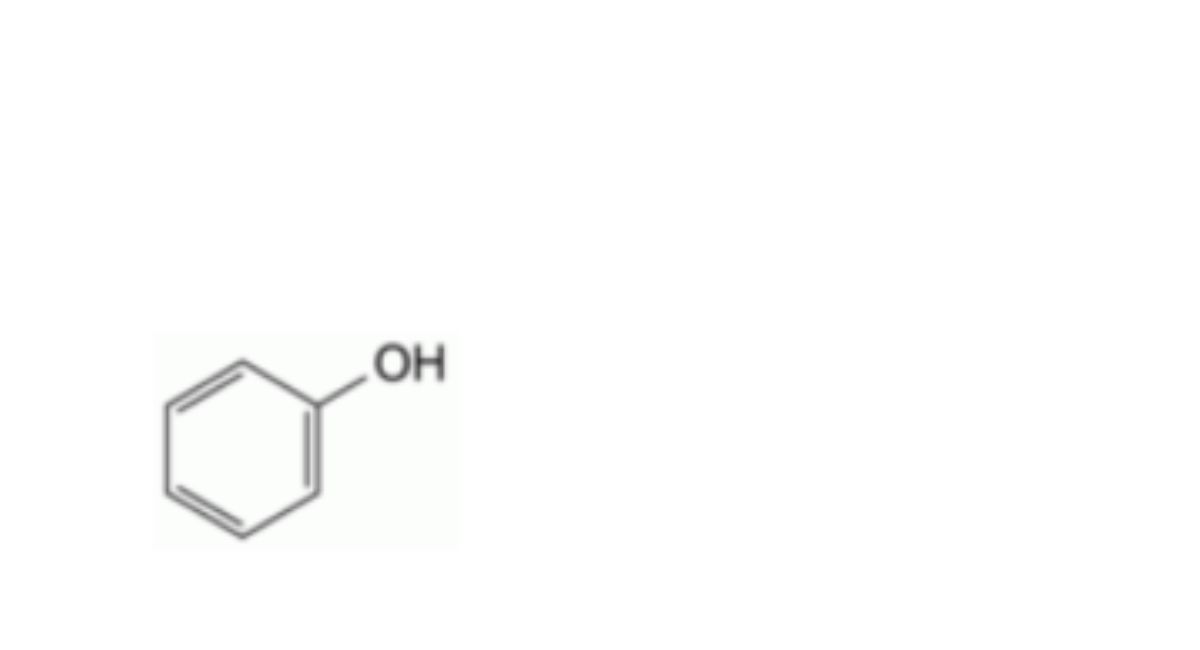
A. phenol,
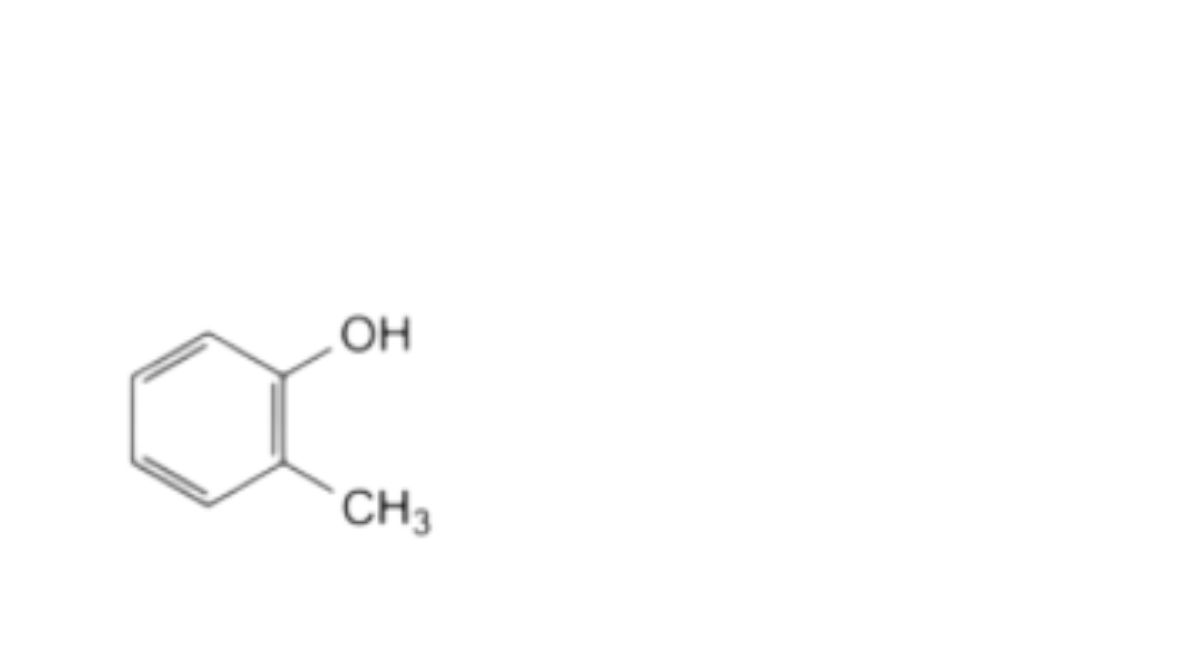
B. 2-methylphenol (o-cresol, cresol),
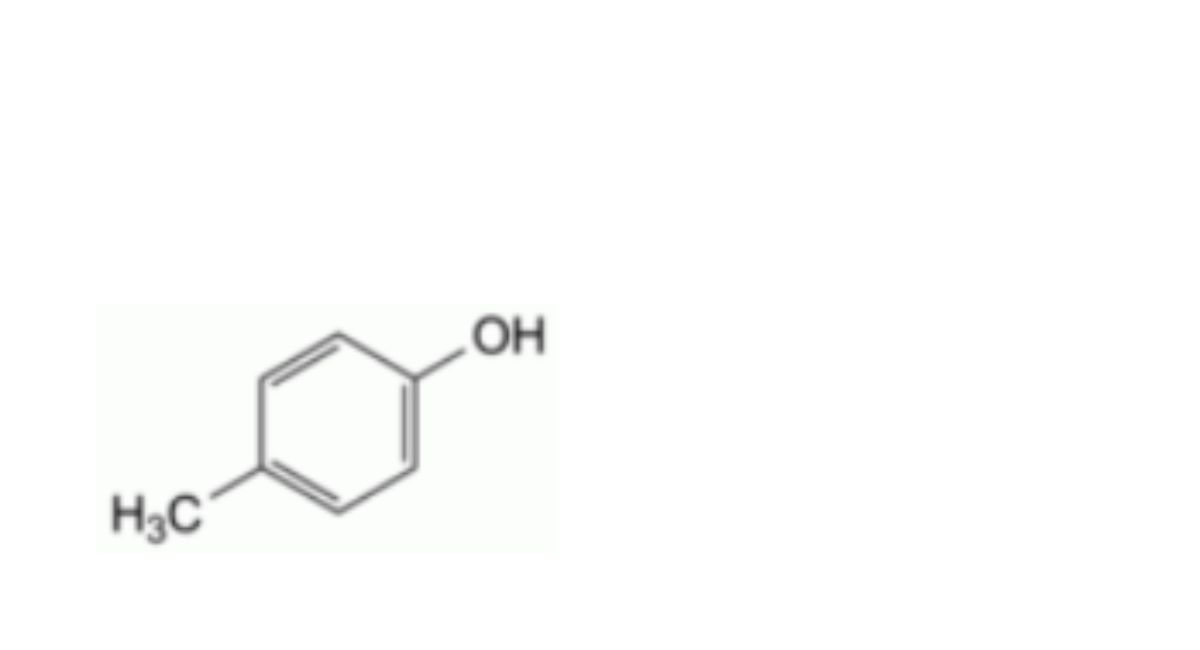
C. 4-methylphenol (p-cresol),
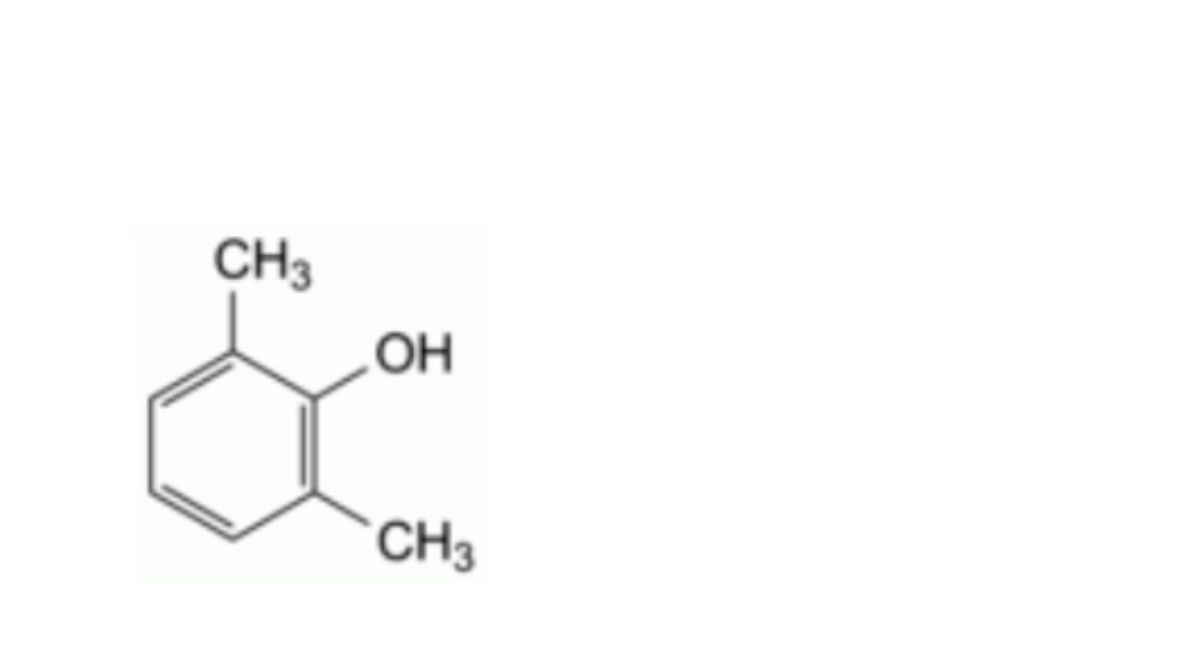
D. 2,6-dimethylphenol (2,6-xylenol),
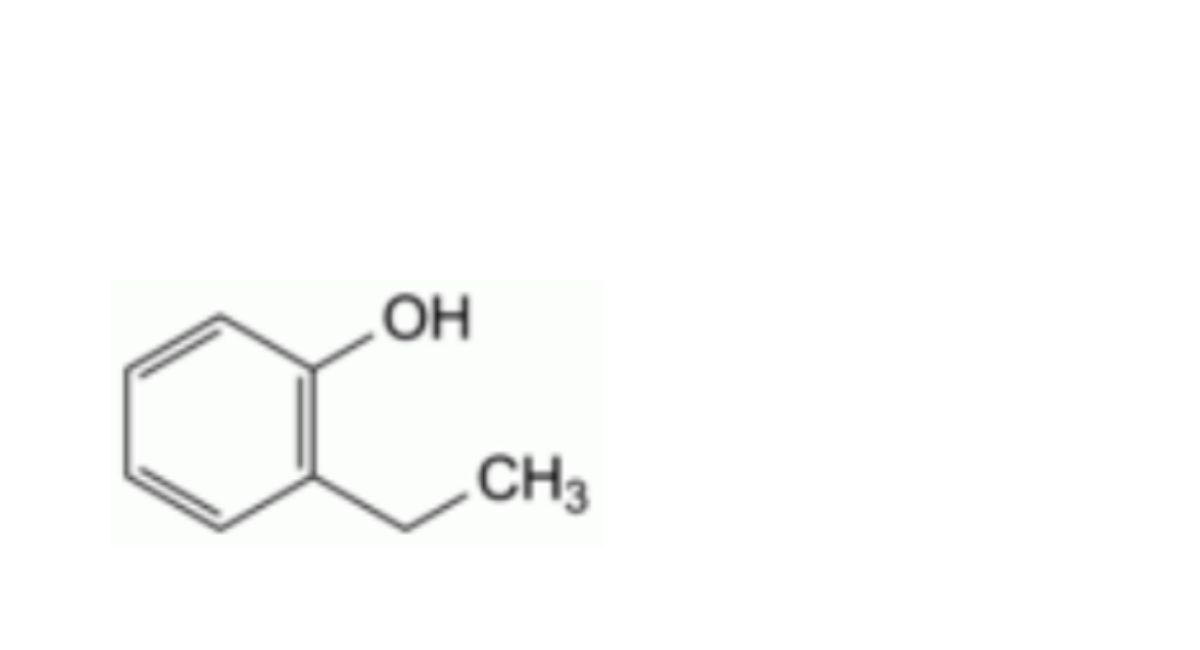
E. 2-ethylphenol (o-ethylphenol),
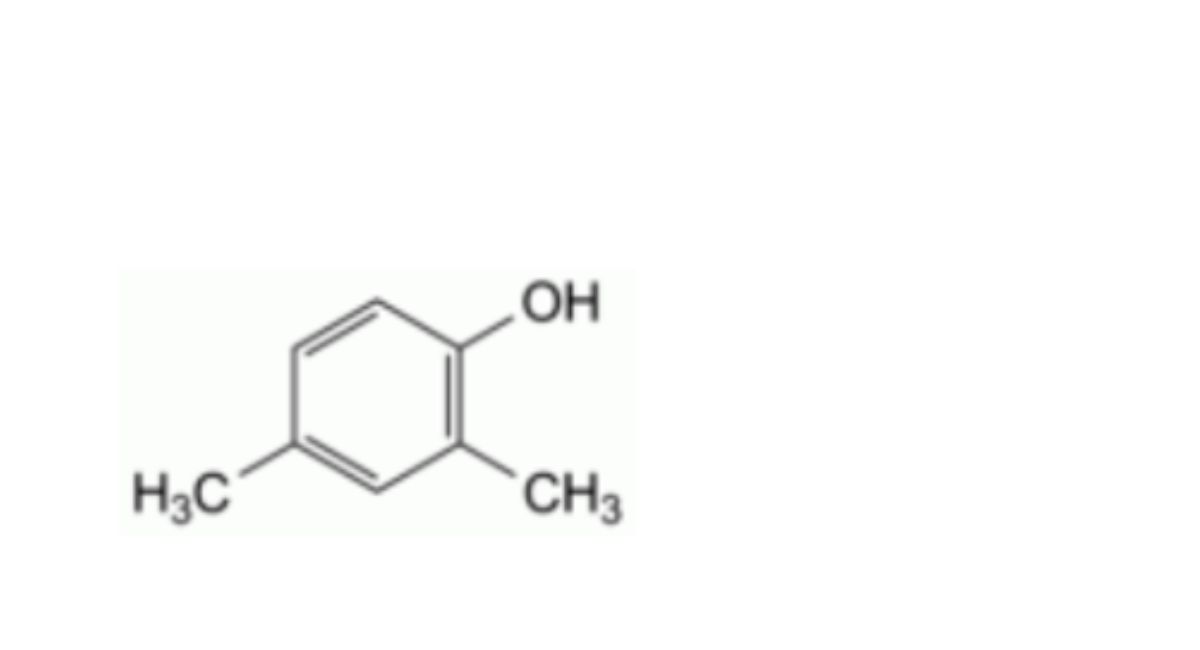
F. 2,4-dimethylphenol (2,4-xylenol),
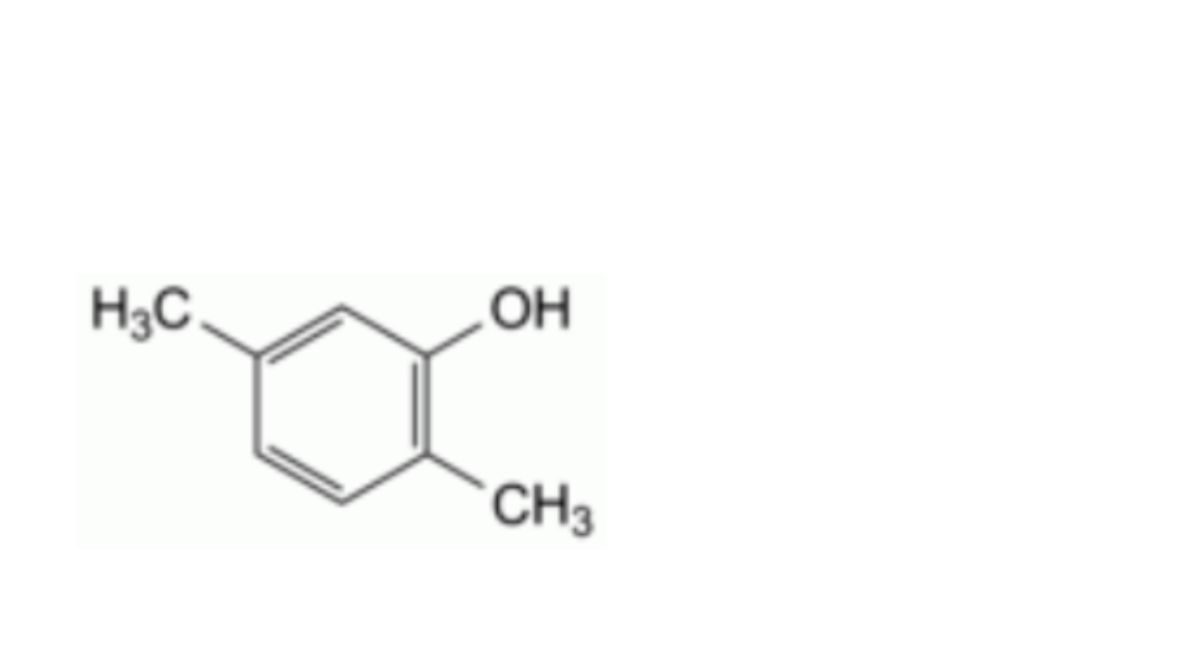
G. 2,5-dimethylphenol (2,5-xylenol),
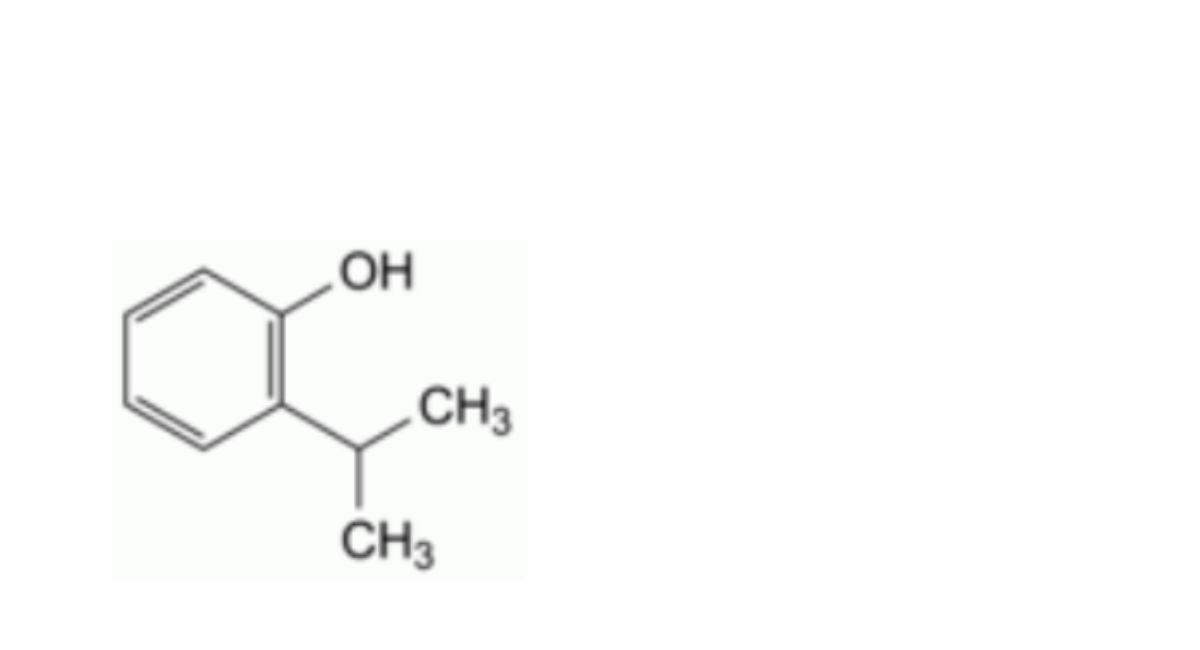
H. 2-(1-methylethyl)phenol,
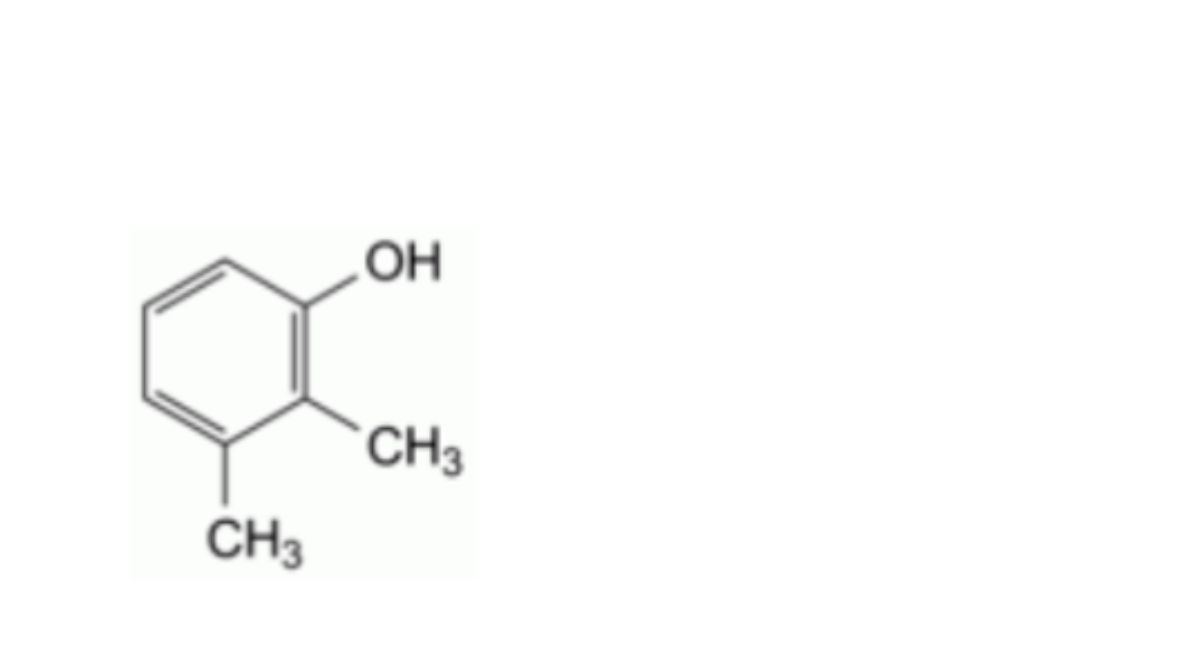
I. 2,3-dimethylphenol (2,3-xylenol),
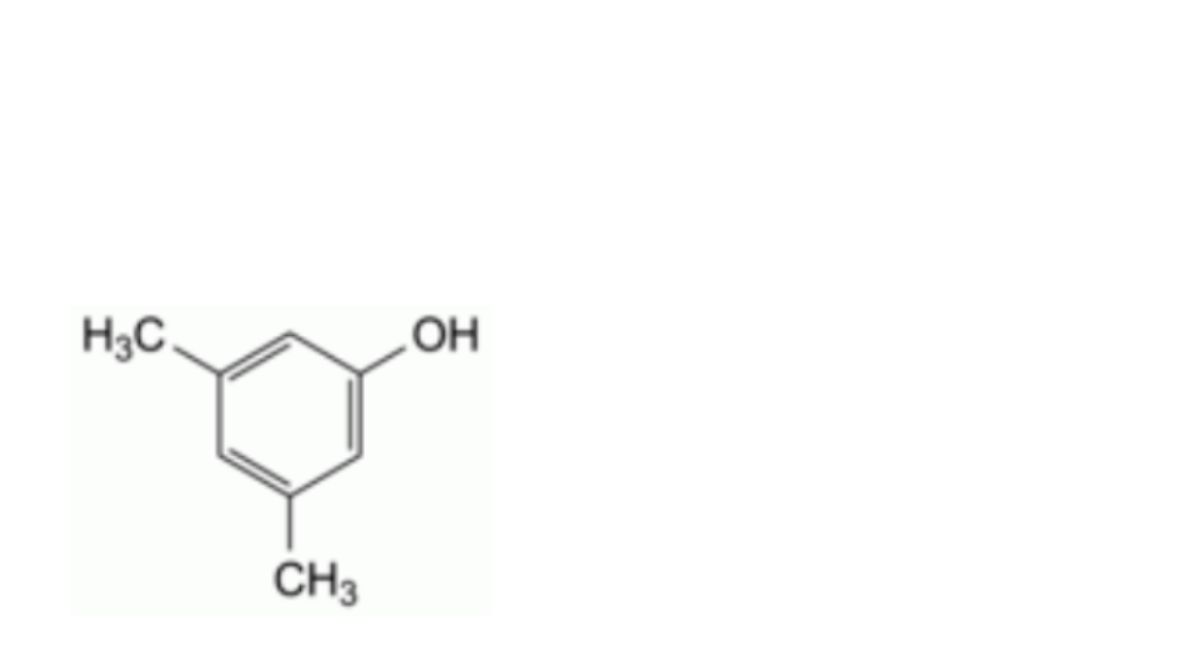
J. 3,5-dimethylphenol (3,5-xylenol),
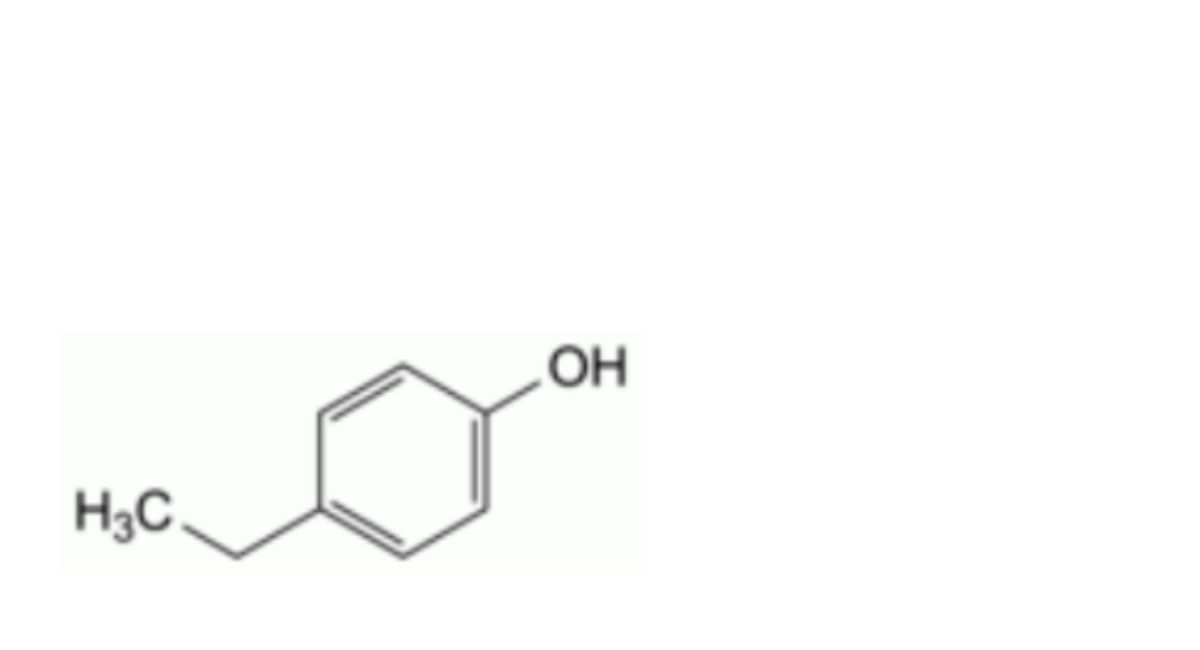
K. 4-ethylphenol (p-ethylphenol),
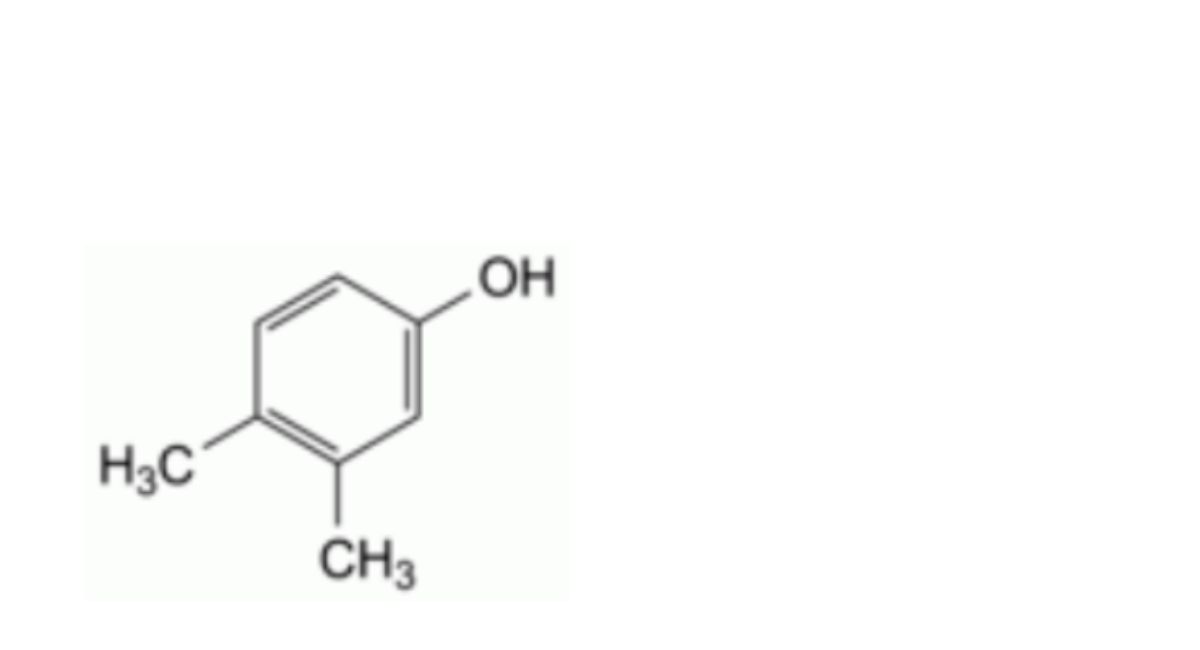
L. 3,4-dimethylphenol (3,4-xylenol),
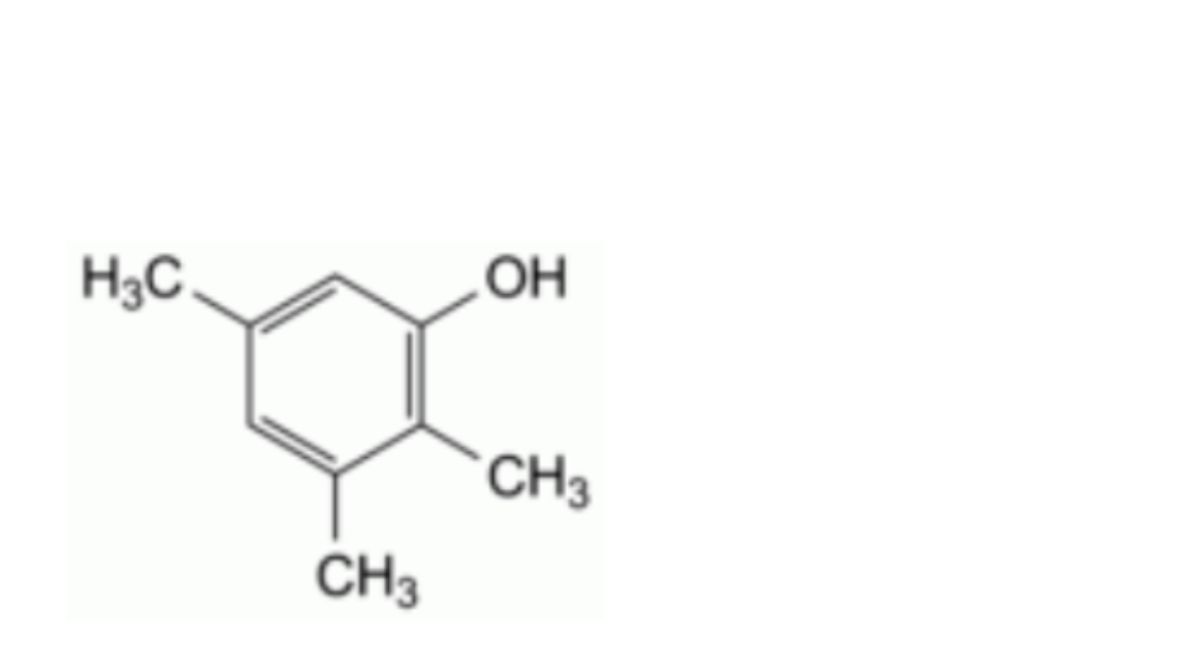
M. 2,3,5-trimethylphenol.