(Ph. Eur. 11.6 update)
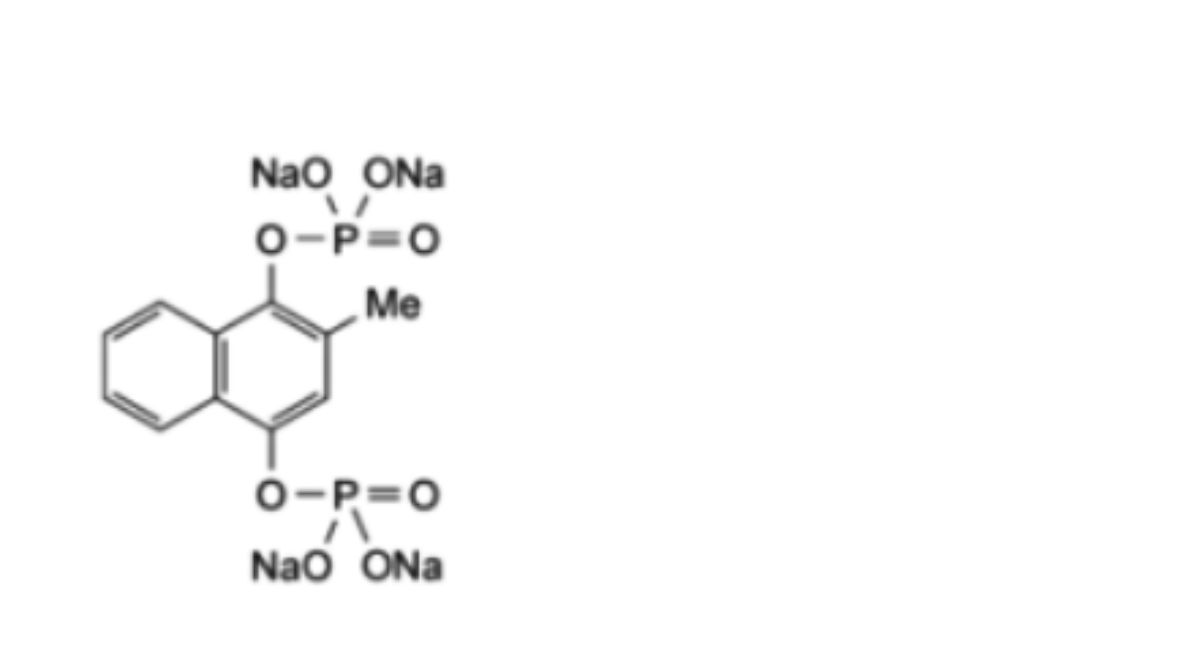
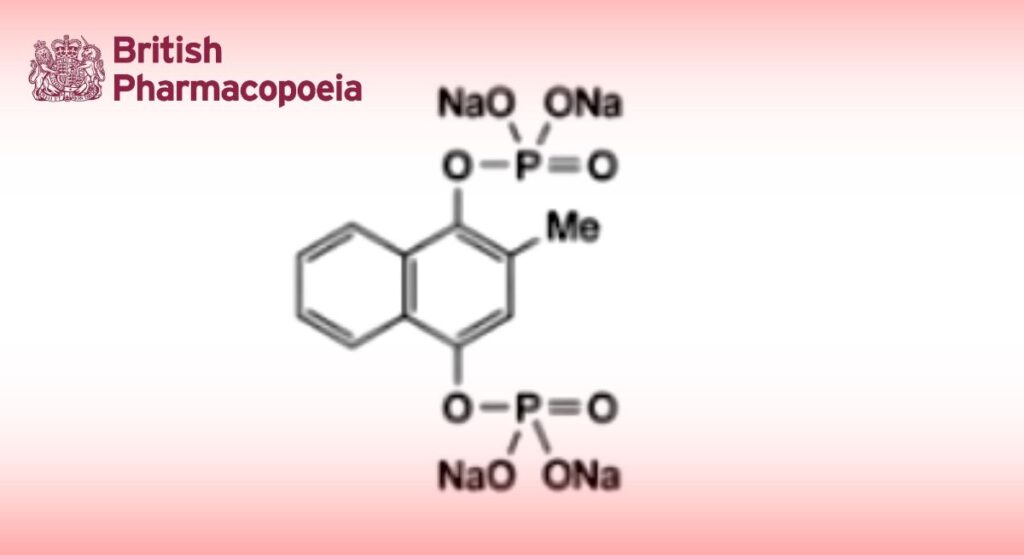
C11H8Na4O8P2,6H2O 530.2 CAS: 6700-42-1
Action and use
Vitamin K analogue.
Preparation
Menadiol Phosphate Tablets.
DEFINITION
Menadiol Sodium Phosphate is tetrasodium 2-methylnaphthalene-1,4-diyl di(orthophosphate) hexahydrate.
It contains not less than 98.0% and not more than 100.5% of C11H8Na4O8P2, calculated with reference to the anhydrous substance.
CHARACTERISTICS
A white to pink, crystalline powder; hygroscopic.
Very soluble in water; practically insoluble in ethanol (96%).
IDENTIFICATION
A. The infrared absorption spectrum, Appendix II A, is concordant with the reference spectrum of menadiol sodium phosphate (RS 213).
B. To 10 mL of a 2% w/v solution add 10 mL of 1M sulfuric acid, 10 mL of 0.1M cerium(IV) sulfate and 1 mL of hydrogen peroxide solution (20 vol) and extract with two 10 mL quantities of chloroform. Evaporate the combined chloroform extracts to dryness on a water bath and dry the residue at 40° at a pressure not exceeding 0.7 kPa. The infrared absorption spectrum of the residue, Appendix II A, is concordant with the reference spectrum of menadione.
C. Dissolve 40 mg in 2 mL of water, heat gently with 2 mL of sulfuric acid until white fumes are evolved. Add nitric acid dropwise until digestion is complete and cool. Add 2 mL of water, heat until white fumes are evolved again, cool, add a further 10 mL of water and neutralise to litmus paper with 5M ammonia. The solution yields reaction A characteristic of sodium salts and reaction B characteristic of phosphates, Appendix VI.
TESTS
Inorganic phosphate
Dissolve 25 mg in 10 mL of water, add 4 mL of 1M sulfuric acid, 1 mL of a 10% w/v solution of ammonium molybdate and 2 mL of methylaminophenyl-sulfite reagent. Allow to stand for 15 minutes. The absorbance of the solution at 730 nm, Appendix II B, is not more than the absorbance of a solution prepared in the same manner but using 10 mL of a 0.0025% w/v solution of potassium dihydrogen orthophosphate in place of the solution of the substance being examined (0.7%).
Total phosphate
Dissolve 85 mg in 50 mL of glacial acetic acid, add 5 mL of mercury(II) acetate solution and carry out Method I for non-aqueous titration, Appendix VIII A, determining the end point potentiometrically.
Not less than 7.7 mL and not more than 8.3 mL of 0.1M perchloric acid VS is required.
Related substances
Carry out the method for thin-layer chromatography, Appendix III A, in subdued light using the following solutions in methanol (50%).
(1) 4.0% w/v of the substance being examined.
(2) 0.020% w/v of the substance being examined.
(3) 0.0080% w/v of 2-methyl-1,4-naphthaquinone.
CHROMATOGRAPHIC CONDITIONS
(a) Use as the coating silica gel GF₂₅₄.
(b) Use the mobile phase as described below.
(c) Apply 5 μL of each solution.
(d) Develop the plate to 15 cm.
(e) After removal of the plate, dry in air, examine under ultraviolet light (254 nm).
MOBILE PHASE
1.5 volumes of diethylamine, 5 volumes of butan-1-ol, 50 volumes of propan-1-ol and 50 volumes of a 2% w/v solution of ammonium chloride.
LIMITS
Any spot corresponding to 2-methyl-1,4-naphthaquinone (menadione) in the chromatogram obtained with solution (1) is not more intense than the spot in the chromatogram obtained with solution (3), and any other secondary spot is not more intense than the spot in the chromatogram obtained with solution (2).
Water
19.0 to 21.5% w/w, Appendix IX A. Use 0.25 g.
ASSAY
Dissolve 0.1 g in 25 mL of water, add 25 mL of glacial acetic acid and 25 mL of 3M hydrochloric acid and titrate with 0.02M cerium(IV) sulfate VS using platinum and calomel electrodes and determining the end point potentiometrically.
Each mL of 0.02M cerium(IV) sulfate VS is equivalent to 4.221 mg of C11H8Na4O8P2