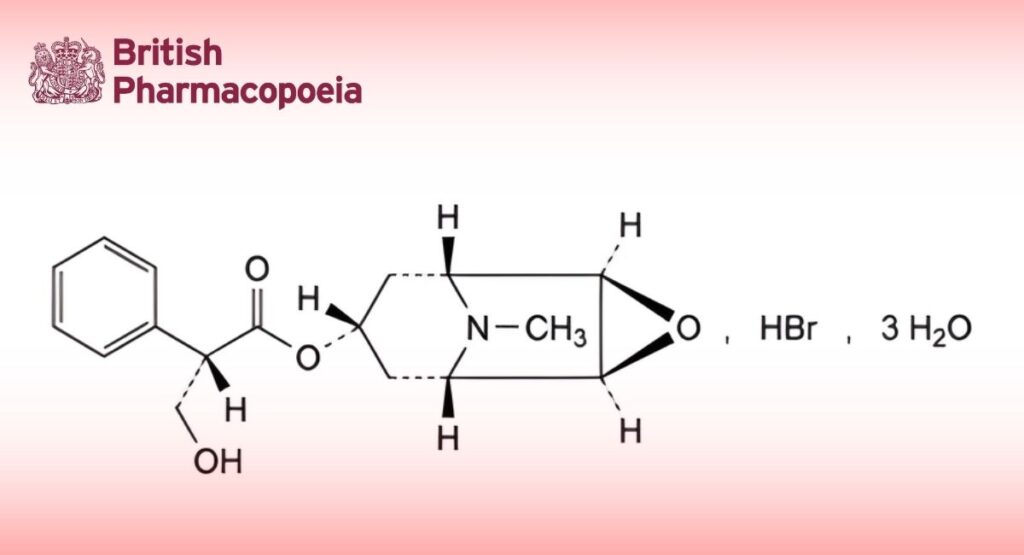
(Ph. Eur. monograph 0106)
C17H22BrNO4,3H2O 438.3 6533-68-2
Action and use
Anticholinergic.
Preparations
Hyoscine Eye Drops
Hyoscine Injection
Hyoscine Tablets
DEFINITION
(1R,2R,4S,5S,7s)-9-Methyl-3-oxa-9-azatricyclo[3.3.1.0 ]non-7-yl (2S)-3-hydroxy-2-phenylpropanoate hydrobromide trihydrate.
Content
99.0 per cent to 101.0 per cent (anhydrous substance).
CHARACTERS
Appearance
White or almost white, crystalline powder or colourless crystals, efflorescent.
Solubility
Freely soluble in water, soluble in ethanol (96 per cent).
IDENTIFICATION
First identification: B, E.
Second identification: A, C, D, E.
A. Specific optical rotation (see Tests).
B. Infrared absorption spectrophotometry (2.2.24).
Comparison: hyoscine hydrobromide CRS.
If the spectra obtained in the solid state show differences, proceed as follows: dissolve 3 mg of the substance to be examined in 1 mL of ethanol (96 per cent) R and evaporate to dryness on a water-bath; dissolve the residue in 0.5 mL of methylene chloride R and add 0.2 g of potassium bromide R and 15 mL of ether R; allow to stand for 5 min shaking frequently; decant; dry the residue on a water-bath until the solvents have evaporated; using the residue prepare a disc and dry at 100-105 °C for 3 h. Repeat the procedure with hyoscine hydrobromide CRS and record the spectra.
C. Dissolve about 50 mg in 5 mL of water R and add 5 mL of picric acid solution R dropwise and with shaking. The precipitate, washed with water R and dried at 100-105 °C for 2 h, melts (2.2.14) at 188 °C to 193 °C.
D. To about 1 mg add 0.2 mL of fuming nitric acid R and evaporate to dryness on a water-bath. Dissolve the residue in 2 mL of acetone R and add 0.1 mL of a 30 g/L solution of potassium hydroxide R in methanol R. A violet colour develops.
E. It gives reaction (a) of bromides (2.3.1).
TESTS
Solution S
Dissolve 2.50 g in carbon dioxide-free water R and dilute to 50.0 mL with the same solvent.
pH (2.2.3)
4.0 to 5.5 for solution S.
Specific optical rotation (2.2.7)
-24 to -27 (anhydrous substance), determined on solution S.
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 70.0 mg of the substance to be examined in the mobile phase and dilute to 50.0 mL with the mobile phase.
Reference solution (a): Dilute 2.0 mL of the test solution to 100.0 mL with the mobile phase. Dilute 5.0 mL of this solution to 20.0 mL with the mobile phase.
Reference solution (b): Dilute 5.0 mL of reference solution (a) to 25.0 mL with the mobile phase.
Reference solution (c): Dissolve 5.0 mg of hyoscine hydrobromide impurity B CRS in the mobile phase, add 5.0 mL of the test solution and dilute to 50.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Column:
— size: l = 0.125 m, Ø = 4.0 mm,
— stationary phase: octylsilyl silica gel for chromatography R (3 μm),
— temperature: 25 ± 1 °C.
Mobile phase: Mix 330 mL of acetonitrile R with 670 mL of a 2.5 g/L solution of sodium dodecyl sulfate R previously adjusted to pH 2.5 with a 345 g/L solution of phosphoric acid R.
Flow rate: 1.5 mL/min.
Detection: Spectrophotometer at 210 nm.
Injection: 5 μL.
Run time: 3 times the retention time of hyoscine.
Relative retention: With reference to hyoscine (retention time = about 5.0 min): impurity D = about 0.2; impurity B = about 0.9; impurity A = about 1.3; impurity C = about 2.4.
System suitability: Reference solution (c):
— resolution: minimum 1.5 between the peaks due to impurity B and hyoscine,
— symmetry factor: maximum 2.5 for the peak due to hyoscine.
Limits:
— correction factors: for the calculation of contents, multiply the peak areas of the following impurities by the corresponding correction factor: impurity D = 0.3; impurity C = 0.6;
— impurity B: not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.5 per cent);
— impurities A, C, D: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.1 per cent);
— any other impurity: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.1 per cent);
— total: not more than 1.4 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.7 per cent); disregard any peak due to the bromide ion which appears close to the solvent peak;
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Water (2.5.12)
10.0 per cent to 13.0 per cent, determined on 0.20 g.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.300 g in a mixture of 5.0 mL of 0.01 M hydrochloric acid and 50 mL of ethanol (96 per cent) R. Carry out a potentiometric titration (2.2.20), using 0.1 M sodium hydroxide free from carbonate.
Read the volume added between the 2 points of inflexion.
1 mL of 0.1 M sodium hydroxide is equivalent to 38.43 mg of C17H22BrNO4.
STORAGE
In a well-filled, airtight container of small capacity, protected from light.
IMPURITIES
Specified impurities A, B, C, D.
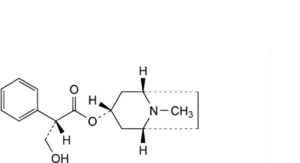
A. (1R,3r,5S)-8-methyl-8-azabicyclo[3.2.1]oct-3-yl (2S)-3-hydroxy-2-phenylpropanoate (hyoscyamine),
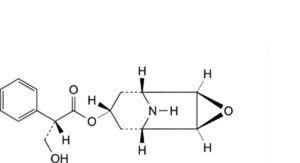
B. (1R,2R,4S,5S,7s)-3-oxa-9-azatricyclo[3.3.1.0 ]non-7-yl (2S)-3-hydroxy-2-phenylpropanoate (norhyoscine),
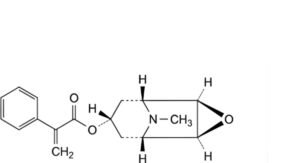
C. (1R,2R,4S,5S,7s)-9-methyl-3-oxa-9-azatricyclo[3.3.1.0 ]non-7-yl 2-phenylprop-2-enoate (apohyoscine),
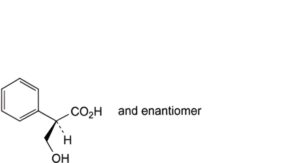
D. (2RS)-3-hydroxy-2-phenylpropanoic acid (DL-tropic acid).