(Ph. Eur. monograph 1721)
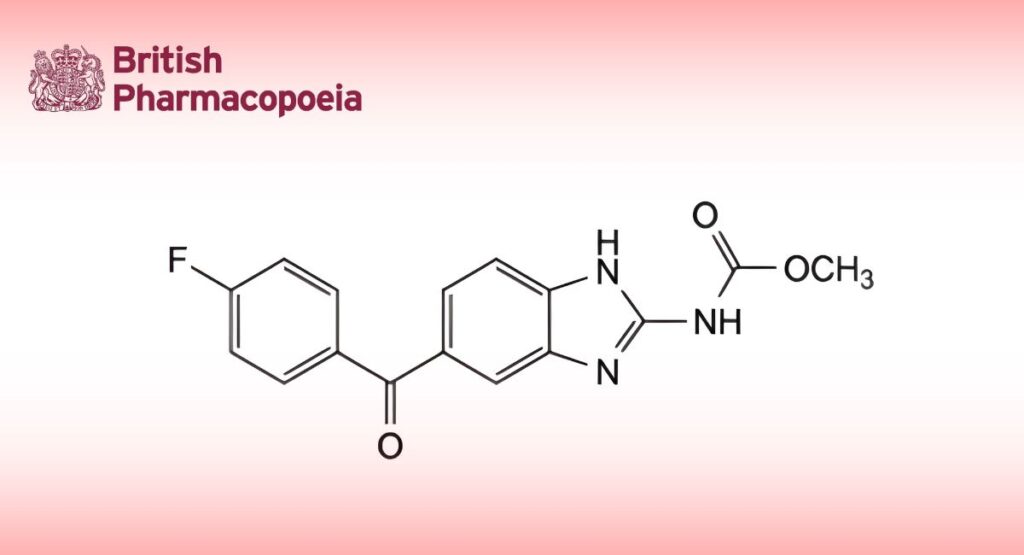
C16H12FN3O3 313.3 31430-15-6
Action and use
Benzimadazole antihelminthic.
DEFINITION
Methyl [5-(4-fluorobenzoyl)-1H-benzimidazol-2-yl]carbamate.
Content
99.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white powder.
Solubility
Practically insoluble in water, in ethanol (96 per cent) and in methylene chloride.
It shows polymorphism (5.9).
IDENTIFICATION
Infrared absorption spectrophotometry (2.2.24), without recrystallisation.
Comparison flubendazole CRS.
TESTS
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 0.100 g of the substance to be examined in dimethylformamide R and dilute to 100.0 mL with the same solvent.
Reference solution (a): Dissolve 5 mg of flubendazole for system suitability CRS in dimethylformamide R and dilute to 5.0 mL with the same solvent.
Reference solution (b): Dilute 1.0 mL of the test solution to 100.0 mL with dimethylformamide R. Dilute 5.0 mL of this solution to 20.0 mL with dimethylformamide R.
Column:
— size: l = 0.10 m, Ø = 4.6 mm,
— stationary phase: base-deactivated octadecylsilyl silica gel for chromatography R (3 μm),
— temperature: 40 °C.
Mobile phase:
— mobile phase A: 7.5 g/L solution of ammonium acetate R,
— mobile phase B: acetonitrile R,
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 15 | 90 → 75 | 10 → 25 |
| 15 – 30 | 75 → 45 | 25 → 55 |
| 30 – 32 | 45 → 10 | 55 → 90 |
| 32 – 37 | 10 | 90 |
Flow rate 1.2 mL/min.
Detection: Spectrophotometer at 250 nm.
Injection 10 μL.
System suitability Reference solution (a):
— the chromatogram obtained is similar to the chromatogram supplied with flubendazole for system suitability CRS.
Limits:
— correction factors: for the calculation of contents, multiply the peak areas of the following impurities by the corresponding correction factor: impurity A = 1.4; impurity C = 1.3; impurity D = 1.3; impurity G = 1.4,
— impurities A, B, C, D, E, G: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.25 per cent),
— impurity F: not more than twice the area of the principal peak in the chromatogram obtained with reference solution (b) (0.5 per cent),
— any other impurity with a relative retention between 1.2 and 1.3: not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.25 per cent),
— total: not more than 6 times the area of the principal peak in the chromatogram obtained with reference solution (b) (1.5 per cent),
— disregard limit: 0.2 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C, for 4 h.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.250 g in 3 mL of anhydrous formic acid R and add 50 mL of a mixture of 1 volume of anhydrous acetic acid R and 7 volumes of methyl ethyl ketone R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 31.33 mg of C16H12FN3O3.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D, E, F, G.
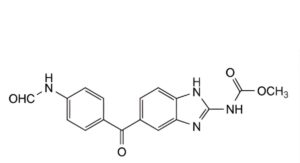
A. methyl [5-[4-(formylamino)benzoyl]-1H-benzimidazol-2-yl]carbamate,
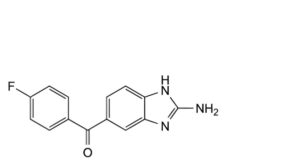
B. (2-amino-1H-benzimidazol-5-yl)(4-fluorophenyl)methanone,
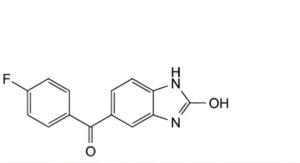
C. (4-fluorophenyl)(2-hydroxy-1H-benzimidazol-5-yl)methanone,
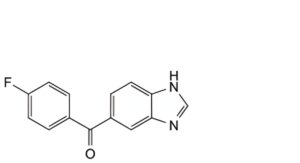
D. (1H-benzimidazol-5-yl)(4-fluorophenyl)methanone,
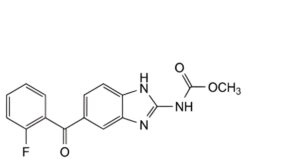
E. methyl [5-(2-fluorobenzoyl)-1H-benzimidazol-2-yl]carbamate,
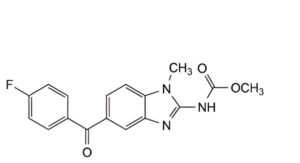
F. methyl [5-(4-fluorobenzoyl)-1-methyl-1H-benzimidazol-2-yl]carbamate,
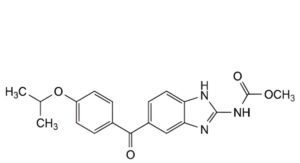
G. methyl [5-[4-(1-methylethoxy)benzoyl]-1H-benzimidazol-2-yl]carbamate.