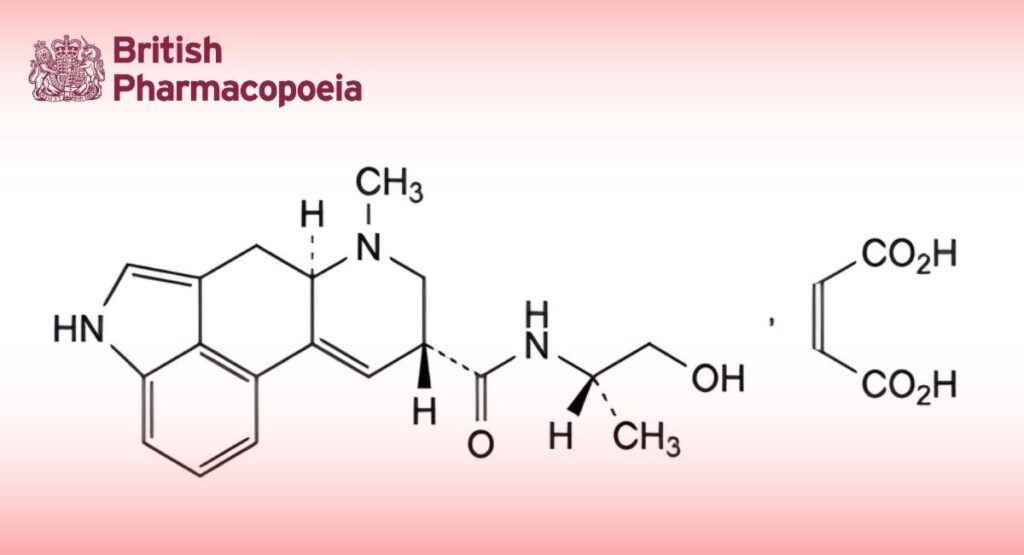
(Ph. Eur. monograph 0223)
C23H27N3O6 441.5 129-51-1
Action and use
Oxytocic.
Preparations
Ergometrine Injection
Ergometrine and Oxytocin Injection
DEFINITION
(8β)-N-[(2S)-1-Hydroxypropan-2-yl]-6-methyl-9,10-didehydroergoline-8-carboxamide (2Z)-but-2-enedioate.
Content
98.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white or slightly coloured, crystalline powder.
Solubility
Sparingly soluble in water, slightly soluble in ethanol (96 per cent).
IDENTIFICATION
First identification: A.
Second identification: B.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison: ergometrine maleate CRS.
B. Thin-layer chromatography (2.2.27). Prepare the solutions immediately before use.
Solvent mixture concentrated ammonia R, ethanol (80 per cent V/V) R (10:90 V/V).
Test solution: Dissolve 10 mg of the substance to be examined in the solvent mixture and dilute to 10 mL with the solvent mixture.
Reference solution: Dissolve 10 mg of ergometrine maleate CRS in the solvent mixture and dilute to 10 mL with the solvent mixture.
Plate: TLC silica gel G plate R.
Mobile phase: water R, methanol R, methylene chloride R (3:25:75 V/V/V).
Application: 5 μL.
Development: Over 2/3 of the plate.
Drying: In a current of cold air.
Detection: Spray with dimethylaminobenzaldehyde solution R7 and dry in a current of warm air for about 2 min.
Results: The principal spot in the chromatogram obtained with the test solution is similar in position, colour and size to the principal spot in the chromatogram obtained with the reference solution.
TESTS
Solution S
Dissolve 0.100 g, without heating and protected from light, in 9 mL of carbon dioxide-free water R and dilute to 10.0 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and not more intensely coloured than reference solution Y5 or BY5 (2.2.2, Method II).
pH (2.2.3)
3.6 to 4.4 for solution S.
Specific optical rotation (2.2.7)
+ 50 to + 56 (dried substance), determined on solution S.
Related substances
Liquid chromatography (2.2.29). Carry out the test protected from light.
Solvent mixture acetonitrile R, water R (15:85 V/V).
Test solution: Dissolve 10.0 mg of the substance to be examined in 10 mL of the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a): Dilute 1.0 mL of the test solution to 100.0 mL with water R. Dilute 1.0 mL of this solution to 10.0 mL with water R.
Reference solution (b): Dissolve the contents of a vial of ergometrine impurity mixture CRS (impurities D, E, F and I) in 1 mL of a mixture of 30 volumes of mobile phase B and 70 volumes of water R.
Column:
— size: l = 0.10 m, Ø = 4.6 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography with extended pH range R (3.5 μm);
— temperature: 30 °C.
Mobile phase:
— mobile phase A: 2 g/L solution of ammonium carbamate R;
— mobile phase B: acetonitrile R, water for chromatography R (50:50 V/V);
| Time
(min) |
Mobile phase A
(per cent V/V) |
Mobile phase B
(per cent V/V) |
| 0 – 5 | 85 | 15 |
| 5 – 10 | 85 → 65 | 15 → 35 |
| 10 – 15 | 65 | 35 |
| 15 – 20 | 65 → 20 | 35 → 80 |
| 20 – 25 | 20 | 80 |
Flow rate: 2.0 mL/min.
Detection: Spectrophotometer at 310 nm.
Injection: 20 μL.
Identification of impurities: Use the chromatogram supplied with ergometrine impurity mixture CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities D, E, F and I.
Relative retention: With reference to ergometrine (retention time = about 11.8 min): impurity D = about 1.3; impurity I = about 1.35; impurity E = about 1.43; impurity F = about 1.55.
System suitability: Reference solution (b):
— resolution: minimum 2.0 between the peaks due to impurities D and I; minimum 1.5 between the peaks due to impurities I and E.
Calculation of percentage contents:
— for each impurity, use the concentration of ergometrine maleate in reference solution (a).
Limits:
— impurity F: maximum 0.15 per cent;
— unspecified impurities: for each impurity, maximum 0.10 per cent;
— total: maximum 0.3 per cent;
— reporting threshold: 0.05 per cent.
Loss on drying (2.2.32)
Maximum 2.0 per cent, determined on 0.200 g by drying in vacuo at 80 °C for 2 h.
ASSAY
Dissolve 0.150 g in 40 mL of anhydrous acetic acid R. Titrate with 0.05 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.05 M perchloric acid is equivalent to 22.07 mg of C23H27N3O6.
STORAGE
In an airtight, glass container, protected from light, at a temperature of 2 °C to 8 °C.
IMPURITIES
Specified impurities F.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, B, C, D, E, G, H, I.
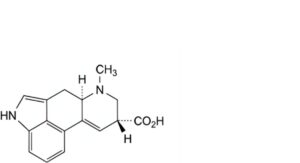
A. (8β)-6-methyl-9,10-didehydroergoline-8-carboxylic acid (lysergic acid),
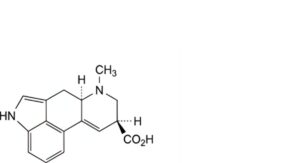
B. (8α)-6-methyl-9,10-didehydroergoline-8-carboxylic acid (isolysergic acid),
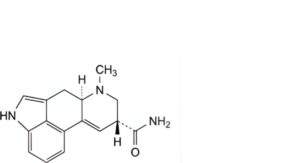
C. (8β)-6-methyl-9,10-didehydroergoline-8-carboxamide (ergine),
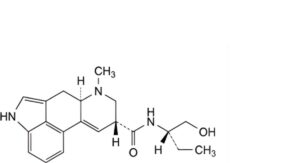
D. (8β)-N-[(2S)-1-hydroxybutan-2-yl]-6-methyl-9,10-didehydroergoline-8-carboxamide (methylergometrine),
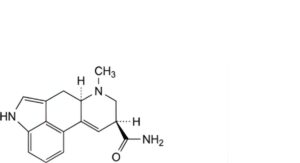
E. (8α)-6-methyl-9,10-didehydroergoline-8-carboxamide (erginine),
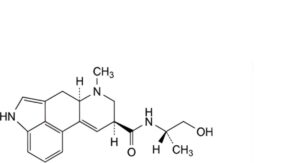
F. (8α)-N-[(2S)-1-hydroxypropan-2-yl]-6-methyl-9,10-didehydroergoline-8-carboxamide (ergometrinine),
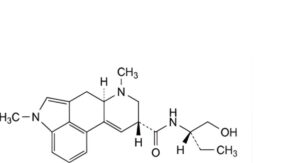
G. (8β)-N-[(2S)-1-hydroxybutan-2-yl]-1,6-dimethyl-9,10-didehydroergoline-8-carboxamide (methysergide),
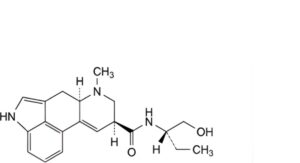
H. (8α)-N-[(2S)-1-hydroxybutan-2-yl]-6-methyl-9,10-didehydroergoline-8-carboxamide (methylergometrinine),
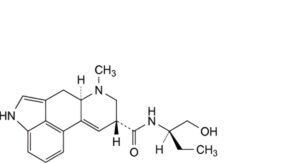
I. (8β)-N-[(2R)-1-hydroxybutan-2-yl]-6-methyl-9,10-didehydroergoline-8-carboxamide