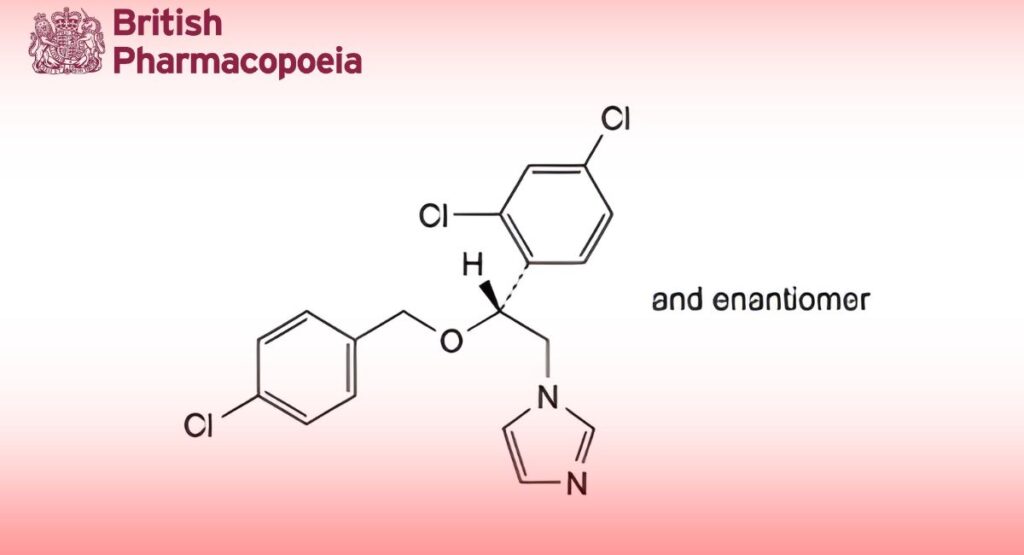
(Ph. Eur. monograph 2049)
C18H15Cl3N2O 381.7 27220-47-9
Action and use
Antifungal.
DEFINITION
1-[(2RS)-2-[(4-Chlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl]-1H-imidazole.
Content
99.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white powder.
Solubility
Practically insoluble in water, very soluble in ethanol (96 per cent) and in methylene chloride.
IDENTIFICATION
A. Melting point (2.2.14): 88 °C to 92 °C.
B. Infrared absorption spectrophotometry (2.2.24).
Comparison: econazole CRS.
TESTS
Related substances
Liquid chromatography (2.2.29).
Test solution: Dissolve 0.100 g of the substance to be examined in methanol R and dilute to 10.0 mL with the same
solvent.
Reference solution (a): Dissolve 10 mg of econazole for system suitability CRS (containing impurities A, B and C) in methanol R and dilute to 1.0 mL with the same solvent.
Reference solution (b): Dilute 1.0 mL of the test solution to 20.0 mL with methanol R. Dilute 1.0 mL of this solution to 25.0 mL with methanol R.
Column:
— size: l = 0.10 m, Ø = 4.6 mm;
— stationary phase: base-deactivated octadecylsilyl silica gel for chromatography R (3 μm);
— temperature: 35 °C.
Mobile phase:
— mobile phase A: methanol R, 0.77 g/L solution of ammonium acetate R (20:80 V/V);
— mobile phase B: methanol R, acetonitrile R (40:60 V/V);
| Time (min) |
Mobile phase A (per cent V/V) |
Mobile phase B (per cent V/V) |
| 0 – 25 | 60 → 10 | 40 → 90 |
| 25 – 27 | 10 | 90 |
Flow rate: 1.5 mL/min.
Detection: Spectrophotometer at 225 nm.
Injection: 10 μL.
Identification of impurities: Use the chromatogram supplied with econazole for system suitability CRS and the
chromatogram obtained with reference solution (a) to identify the peaks due to impurities A, B and C.
Relative retention: With reference to econazole (retention time = about 15 min): impurity A = about 0.2;
impurity B = about 0.6; impurity C = about 1.1.
System suitability: Reference solution (a):
— peak-to-valley ratio: minimum 1.5, where Hp = height above the baseline of the peak due to impurity C and Hv = height above the baseline of the lowest point of the curve separating this peak from the peak due to econazole.
Limits:
— correction factor: for the calculation of content, multiply the peak area of impurity A by 1.4;
— impurities A, B, C: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (b) (0.2 per cent);
— unspecified impurities: for each impurity, not more than 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.10 per cent);
— total: not more than 1.5 times the area of the principal peak in the chromatogram obtained with reference
solution (b) (0.3 per cent);
— disregard limit: 0.25 times the area of the principal peak in the chromatogram obtained with reference solution (b) (0.05 per cent).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in vacuo at 60 °C for 4 h.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.300 g in 75 mL of anhydrous acetic acid R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20). Carry out a blank titration.
1 mL of 0.1 M perchloric acid is equivalent to 38.17 mg of C18H15Cl3N2O.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C.

A. (1RS)-1-(2,4-dichlorophenyl)-2-(1H-imidazol-1-yl)ethanol,
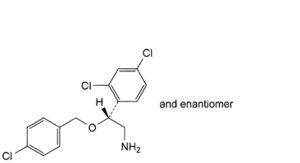
B. (2RS)-2-[(4-chlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethanamine,
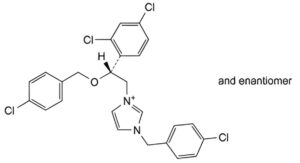
C. 1-(4-chlorobenzyl)-3-[(2RS)-2-[(4-chlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl]imidazolium.