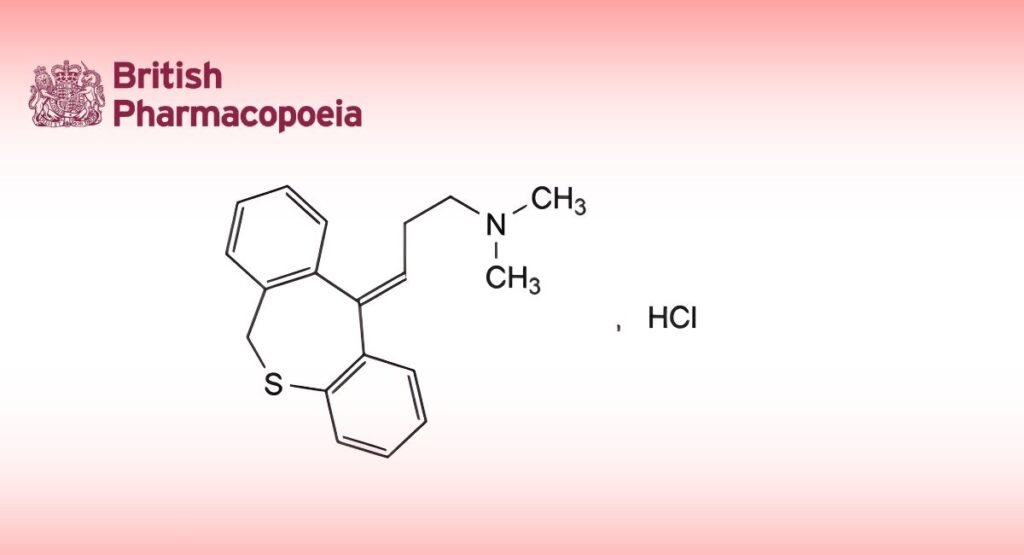
(Ph. Eur. monograph 1096)
C19H22ClNO 315.8 1229-29-4
Action and use
Monoamine reuptake inhibitor; tricyclic antidepressant.
Preparation
Doxepin Capsules
DEFINITION
(E)-3-(Dibenzo[b,e]oxepin-11(6H)-ylidene)-N,N-dimethylpropan-1-amine hydrochloride.
Content
98.0 per cent to 101.0 per cent of C19H22ClNO (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Freely soluble in water, in ethanol (96 per cent) and in methylene chloride.
IDENTIFICATION
First identification: C, E.
Second identification: A, B, D, E.
A. Melting point (2.2.14): 185 °C to 191 °C.
B. Ultraviolet and visible absorption spectrophotometry (2.2.25).
Test solution Dissolve 50.0 mg in a 1 g/L solution of hydrochloric acid R in methanol R and dilute to 100.0 mL with the same acid solution. Dilute 5.0 mL to 50.0 mL with a 1 g/L solution of hydrochloric acid R in methanol R.
Spectral range 230-350 nm.
Absorption maximum At 297 nm.
Specific absorbance at the absorption maximum 128 to 142.
C. Infrared absorption spectrophotometry (2.2.24).
Comparison doxepin hydrochloride CRS.
D. Dissolve about 5 mg in 2 mL of sulfuric acid R. A dark red colour is produced.
E. Solution S (see Tests) gives reaction (a) of chlorides (2.3.1).
TESTS
Solution S
Dissolve 1.5 g in carbon dioxide-free water R and dilute to 30 mL with the same solvent.
Appearance of solution
Dilute 10 mL of solution S to 25 mL with water R. The solution is clear (2.2.1) and colourless (2.2.2, Method II).
Acidity
To 10 mL of solution S add 0.1 mL of methyl red solution R. Not more than 0.1 mL of 0.1 M sodium hydroxide is required to change the colour of the indicator to yellow.
Related substances
Liquid chromatography (2.2.29). Prepare the solutions immediately before use and protect them from light.
Phosphate buffer solution: Dissolve 1.42 g of anhydrous disodium hydrogen phosphate R in water R, adjust to pH 7.7 with dilute phosphoric acid R and dilute to 1000 mL with water R.
Solvent mixture: Mix 1 volume of 1 M sodium hydroxide and 250 volumes of the mobile phase.
Test solution: Dissolve 50 mg of the substance to be examined in the solvent mixture and dilute to 50.0 mL with the solvent mixture.
Reference solution (a): Dilute 1.0 mL of the test solution to 100.0 mL with the solvent mixture. Dilute 1.0 mL of this solution to 10.0 mL with the solvent mixture.
Reference solution (b): Dissolve the contents of a vial of doxepin for system suitability CRS (containing impurities A, B and C) in 1.0 mL of mobile phase.
Column:
— size: l = 0.25 m, Ø = 4.6 mm;
— stationary phase: end-capped octadecylsilyl silica gel for chromatography R (5 μm);
— temperature: 30 °C.
Mobile phase acetonitrile R1, phosphate buffer solution, methanol R1 (20:30:50 V/V/V).
Flow rate 1.0 mL/min.
Detection Spectrophotometer at 215 nm.
Injection 20 μL.
Run time 1.5 times the retention time of doxepin.
Identification of impurities: Use the chromatogram supplied with doxepin for system suitability CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities A, B and C.
Relative retention: With reference to doxepin (retention time = about 18 min): impurity A = about 0.5;
impurity C = about 0.6; impurity B = about 0.7; the peak due to doxepin might show a shoulder caused by the (Z)-isomer (impurity D).
System suitability Reference solution (b):
— resolution: minimum 1.5 between the peaks due to impurities A and C, and minimum 1.5 between the peaks due to impurities C and B;
—the chromatogram obtained is similar to the chromatogram supplied with doxepin for system suitability CRS.
Limits:
— correction factor: for the calculation of content, multiply the peak area of impurity B by 1.7;
— impurities A, B: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent);
— impurity C: not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
— total: not more than 3 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.3 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
(Z)-Isomer
Liquid chromatography (2.2.29).
Test solution Dissolve 20.0 mg of the substance to be examined in the mobile phase and dilute to 20.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Column:
— size: l = 0.12 m, Ø = 4 mm;
— stationary phase: spherical octylsilyl silica gel for chromatography R (5 μm) with a specific surface area of 220 m /g and a pore size of 80 nm;
— temperature: 50 °C.
Mobile phase Mix 30 volumes of methanol R and 70 volumes of a 30 g/L solution of sodium dihydrogen phosphate R previously adjusted to pH 2.5 with phosphoric acid R.
Flow rate 1 mL/min.
Detection Spectrophotometer at 254 nm.
Injection 20 μL.
System suitability:
— resolution: minimum 1.5 between the peaks due to the (E)-isomer (1 peak) and to the (Z)-isomer (2 peak).
Results:
— calculate the ratio of the area of the peak due to the (E)-isomer to the area of the peak due to the (Z)-isomer: this ratio is 4.4 to 6.7 (13.0 per cent to 18.5 per cent of the (Z)-isomer).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.250 g in a mixture of 5 mL of anhydrous acetic acid R and 35 mL of acetic anhydride R. Using 0.2 mL of crystal violet solution R as indicator, titrate with 0.1 M perchloric acid until the colour changes from blue to green.
1 mL of 0.1 M perchloric acid is equivalent to 31.58 mg of C19H22ClNO.
STORAGE
Protected from light.
IMPURITIES
Specified impurities A, B, C, D.
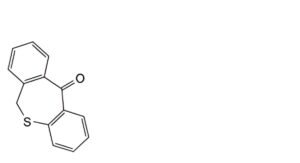
A. dibenzo[b,e]oxepin-11(6H)-one (doxepinone),
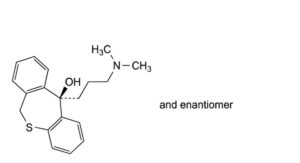
B. (11RS)-11-[3-(dimethylamino)propyl]-6,11-dihydrodibenzo[b,e]oxepin-11-ol (doxepinol),
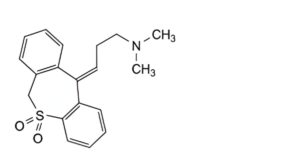
C. (E)-3-(dibenzo[b,e]oxepin-11(6H)-ylidene)-N-methylpropan-1-amine (desmethyldoxepin),
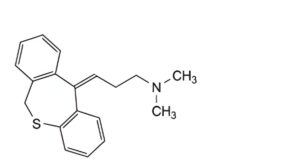
D. (Z)-3-(dibenzo[b,e]oxepin-11(6H)-ylidene)-N,N-dimethylpropan-1-amine.