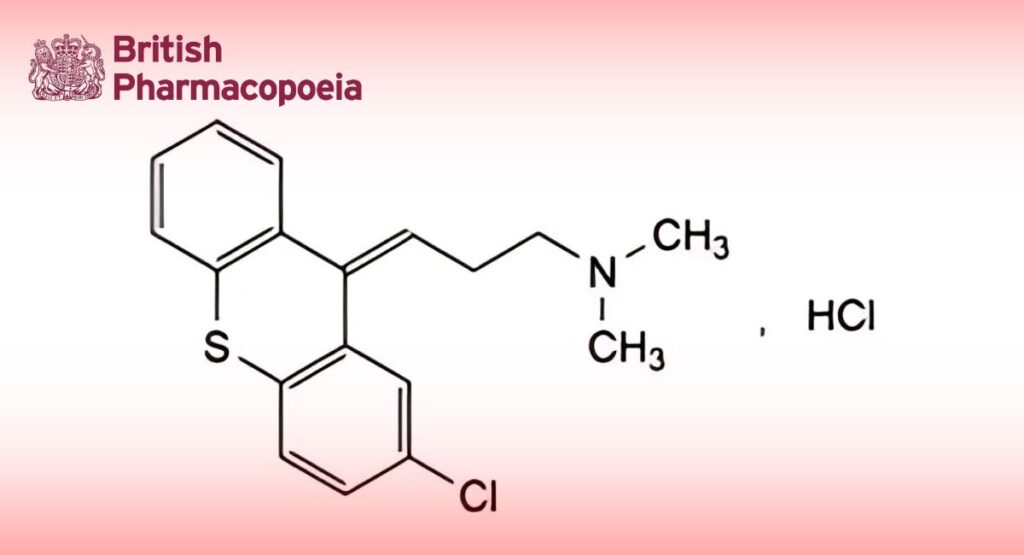
(Ph. Eur. monograph 0815)
C18H19Cl2NS 352.3 6469-93-8
Action and use
Dopamine receptor antagonist; neuroleptic.
DEFINITION
(3Z)-3-(2-Chloro-9H-thioxanthen-9-ylidene)-N,N-dimethylpropan-1-amine hydrochloride.
Content
99.0 per cent to 101.0 per cent (dried substance).
CHARACTERS
Appearance
White or almost white, crystalline powder.
Solubility
Soluble in water and in ethanol (96 per cent), slightly soluble in methylene chloride.
mp
About 220 °C.
IDENTIFICATION
First identification: A, E.
Second identification: B, C, D, E.
A. Infrared absorption spectrophotometry (2.2.24).
Preparation Dissolve 25 mg in 1 mL of water R, add 0.1 mL of dilute sodium hydroxide solution R and shake with 2 mL of methylene chloride R; separate the organic layer and wash with 0.5 mL of water R; evaporate the organic layer to dryness and dry the residue at 40-50 °C; examine the residue as a disc.
Comparison: Repeat the operations using 25 mg of chlorprothixene hydrochloride CRS.
B. Dissolve 0.2 g in a mixture of 5 mL of dioxan R and 5 mL of a 1.5 g/L solution of sodium nitrite R. Add 0.8 mL of nitric acid R. After 10 min, add the solution to 20 mL of water R. Filter the precipate formed after 1 h. The filtrate is used immediately for identification test C. Dissolve the precipitate by warming in about 15 mL of ethanol (96 per cent) R and add the solution to 10 mL of water R. Filter and dry the precipitate at 100-105 °C for 2 h. The melting point (2.2.14) is 152 °C to 154 °C.
C. To 1 mL of the filtrate obtained in identification test B, add 0.2 mL of a suspension of 50 mg of fast red B salt R in 1 mL of ethanol (96 per cent) R. Add 1 mL of 0.5 M alcoholic potassium hydroxide. A dark red colour is produced. Carry out a blank test.
D. Dissolve about 20 mg in 2 mL of nitric acid R. A red colour is produced. Add 5 mL of water R and examine in ultraviolet light at 365 nm. The solution shows green fluorescence.
E. Dissolve 20 mg in 2 mL of water R, acidify with dilute nitric acid R and allow to stand for 5 min. Centrifuge. The supernatant gives reaction (a) of chlorides (2.3.1) starting from ‘add 0.4 mL of silver nitrate solution R1′.
TESTS
Solution S
Dissolve 0.25 g in carbon dioxide-free water R and dilute to 25 mL with the same solvent.
Appearance of solution
Solution S is clear (2.2.1) and colourless (2.2.2, Method II).
pH (2.2.3)
4.4 to 5.2 for solution S.
Related substances
Liquid chromatography (2.2.29). Carry out the test protected from bright light.
Test solution: Dissolve 20.0 mg of the substance to be examined in the mobile phase and dilute to 20.0 mL with the mobile phase.
Reference solution (a): Dilute 1.0 mL of the test solution to 100.0 mL with the mobile phase. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (b): Dissolve the contents of a vial of chlorprothixene for system suitability CRS (containing impurities C and F) in 1 mL of the mobile phase.
Column:
— size: l = 0.10 m, Ø = 4.0 mm;
— stationary phase: base-deactivated end-capped octadecylsilyl silica gel for chromatography R (3 μm).
Mobile phase: Solution containing 6.0 g/L of potassium dihydrogen phosphate R, 2.9 g/L of sodium laurilsulfate R and 9 g/L of tetrabutylammonium bromide R in a mixture of 50 volumes of methanol R, 400 volumes of acetonitrile R and 550 volumes of water for chromatography R.ư
Flow rate: 1.0 mL/min.
Detection: Spectrophotometer at 254 nm.
Equilibration: For about 30 min with the mobile phase.
Injection: 20 μL.
Run time: Twice the retention time of chlorprothixene.
Identification of impurities: Use the chromatogram supplied with chlorprothixene for system suitability CRS and the chromatogram obtained with reference solution (b) to identify the peaks due to impurities C and F.
Relative retention: With reference to chlorprothixene (retention time = about 10 min): impurity C = about 1.25; impurity F = about 1.33.
System suitability: Reference solution (b):
— resolution: minimum 3.0 between the peaks due to chlorprothixene and impurity C.
Limits:
— impurity F: not more than 5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.5 per cent);
— unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
— total: not more than 8 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.8 per cent);
— disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Loss on drying (2.2.32)
Maximum 0.5 per cent, determined on 1.000 g by drying in vacuo at 60 °C for 3 h.
Sulfated ash (2.4.14)
Maximum 0.1 per cent, determined on 1.0 g.
ASSAY
Dissolve 0.300 g in a mixture of 5.0 mL of 0.01 M hydrochloric acid and 50 mL of ethanol (96 per cent) R. Carry out a potentiometric titration (2.2.20), using 0.1 M sodium hydroxide. Read the volume added between the 2 points of inflexion.
1 mL of 0.1 M sodium hydroxide is equivalent to 35.23 mg of C18H19Cl2NS.
STORAGE
Protected from light.
IMPURITIES
Specified impurities F.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use) A, B, C, D,
E.
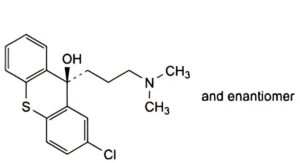
A. (3RS)-2-chloro-9-[3-(dimethylamino)propyl]-9H-thioxanthen-9-ol,
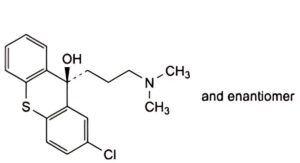
B. N,N-dimethyl-3-(9H-thioxanthen-9-ylidene)propan-1-amine,
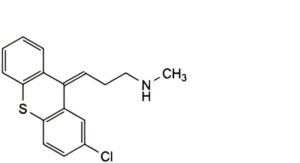
C. (3Z)-3-(2-chloro-9H-thioxanthen-9-ylidene)-N-methylpropan-1-amine,
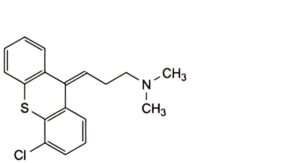
D. (3Z)-3-(4-chloro-9H-thioxanthen-9-ylidene)-N,N-dimethylpropan-1-amine,
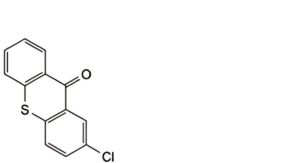
E. 2-chloro-9H-thioxanthen-9-one,
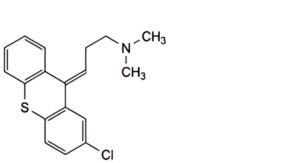
F. (3E)-3-(2-chloro-9H-thioxanthen-9-ylidene)-N,N-dimethylpropan-1-amine ((E)-chlorprothixene).