Action and use
Antiseptic.
DEFINITION
Chlorhexidine Gluconate Dental Gel contains Chlorhexidine Gluconate Solution in a suitable water-miscible basis.
The gel complies with the requirements stated under Topical Semi-solid Preparations and with the following requirements.
Content of chlorhexidine gluconate, C22H30Cl2N10,2C6H12O7
90.0 to 105.0% of the stated amount.
IDENTIFICATION
In the Assay, record the UV spectrum of the principal peak in the chromatograms obtained with solutions (1) and (2) with a diode array detector in the range of 210 to 400 nm.
The UV spectrum of the principal peak in the chromatogram obtained with solution (1) is concordant with that of the peak in the chromatogram obtained with solution (2);
the retention time of the principal peak in the chromatogram obtained with solution (1) is similar to that of the peak in the chromatogram obtained with solution (2).
TESTS
Acidity or alkalinity
pH, 5.0 to 7.0, Appendix V L.
Related substances
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
- Dilute a quantity of the gel containing the equivalent of 10 mg of chlorhexidine gluconate to 50 mL with mobile phase A, stir and filter through a regenerated cellulose filter (Spartan 30 is suitable).
- Dilute 1 volume of solution (1) to 100 volumes with mobile phase A.
- Dilute 1 volume of a solution containing 0.1% w/v of chloroaniline (impurity P) in acetonitrile (50%) to 250 volumes with mobile phase A. Dilute 1 volume of this solution to 10 volumes with mobile phase A.
- Dilute 1 volume of a solution containing 0.2% w/v of chlorhexidine impurity standard BPCRS and 0.001% w/v of chloroaniline in acetonitrile (50%) to 10 volumes with mobile phase A.
- 0.02% w/v of chlorhexidine for system suitability EPCRS in mobile phase A.
- Dilute 1 volume of solution (2) to 10 volumes with mobile phase A.
CHROMATOGRAPHIC CONDITIONS
- Use a stainless steel column (25 cm x 4.6 mm) packed with end-capped octadecylsilyl silica gel for chromatography (5 μm) (Reprosil-Pur Basic C18 is suitable).
- Use gradient elution and the mobile phase described below.
- Use a flow rate of 1 mL per minute.
- Use a column temperature of 40°.
- Use a detection wavelength of 250 nm.
- Inject 25 μL of each solution.
MOBILE PHASE
Solvent A 10% v/v of trifluoroacetic acid in acetonitrile.
Mobile phase A 1.9 volumes of solvent A, 180 volumes of acetonitrile and 820 volumes of water.
Mobile phase B 0.2 volume of solvent A, 200 volumes of water and 800 volumes of acetonitrile.
| Time (Minutes) | Mobile phase A (% v/v) | Mobile phase B (% v/v) | Comment |
| 0-10 | 100 | 0 | isocratic |
| 10-14 | 100->85 | 0->15 | linear gradient |
| 14-43 | 85->75 | 15->25 | linear gradient |
| 43-54 | 75->0 | 25->100 | linear gradient |
| 54-60 | 0 | 100 | isocratic |
| 60-62 | 0->100 | 100->0 | linear gradient |
| 62-70 | 100 | 0 | re-equilibration |
SYSTEM SUITABILITY
The test is not valid unless:
in the chromatogram obtained with solution (4), the resolution between the peaks due to impurity 2 and impurity P is at least 1.5;
in the chromatogram obtained with solution (6), the signal-to-noise ratio for the peak due to chlorhexidine is at least 20.
CALCULATION OF IMPURITIES
For impurity P, use the concentration of chloroaniline in solution (3).
For each other impurity, use the concentration of chlorhexidine gluconate in solution (2).
For the reporting threshold, use the concentration of chlorhexidine gluconate in solution (6).
For peak identification, use solutions (3), (4) and (5).
Chlorhexidine retention time: about 26 minutes.
Relative retention: impurity 1, about 0.14; impurity G, about 0.30; impurity 2, about 0.33; impurity P, about 0.36; impurity N, about 0.5; impurity F, about 0.78; impurity A, about 0.79; impurity H, about 0.81; impurity O, about 0.9; impurity J, about 1.2; impurity K, about 1.3.
Correction factors: impurity 1, multiply by 0.5; impurity G, multiply by 1.3; impurity 2, multiply by 0.6; impurity N, multiply by 1.9, impurity F, multiply by 0.3; impurity A, multiply by 1.5; impurity O, multiply by 1.8; impurity J, multiply by 1.4.
LIMITS
— impurity N: not more than 2.0%;
- impurity H: not more than 1.0%;
- impurities J and K: not more than 0.7% of each;
- impurity G: not more than 0.6%;
- impurity F: not more than 0.5%;
- impurities A and O: not more than 0.4% of each;
- impurity P: not more than 0.2%;
- unspecified impurities: for each impurity, not more than 0.2%;
- total impurities, excluding impurity N: not more than 2.0%;
- reporting threshold: 0.1%.
ASSAY
Carry out the method for liquid chromatography, Appendix III D, using the following solutions.
- Dilute a quantity of the gel containing the equivalent of 10 mg of chlorhexidine gluconate to 50 mL with mobile phase A, stir and filter through a regenerated cellulose filter (Spartan 30 is suitable).
- 0.0144% w/v of chlorhexidine acetate BPCRS in mobile phase A.
- Dilute 1 volume of a solution containing 0.2% w/v of chlorhexidine impurity standard BPCRS and 0.001% w/v of chloroaniline in acetonitrile (50%) to 10 volumes with mobile phase A.
CHROMATOGRAPHIC CONDITIONS
The chromatographic conditions described under Related substances may be used.
SYSTEM SUITABILITY
The test is not valid unless, in the chromatogram obtained with solution (3), the resolution between the peaks due to impurity 2 and impurity P is at least 1.5.
DETERMINATION OF CONTENT
Calculate the content of C22H30Cl2N10,2C6H12O7 in the gel from the declared content of C22H30Cl2N10 in chlorhexidine acetate BPCRS. Each mg of C22H30Cl2N10 is equivalent to 1.776 mg of C22H30Cl2N10,2C6H12O7.
IMPURITIES
The impurities limited by the requirements of this monograph include impurities A, B, F, G, H, J, K, N, O and P listed under Chlorhexidine Gluconate Solution and:
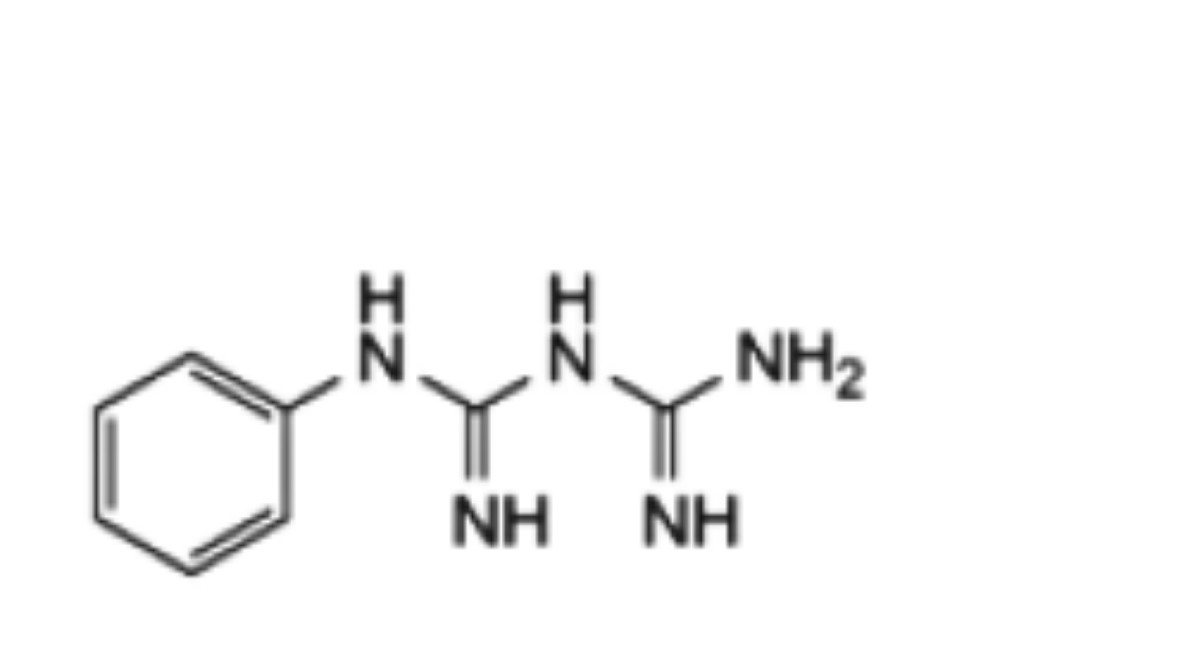
1. 1-phenylbiguanidine;
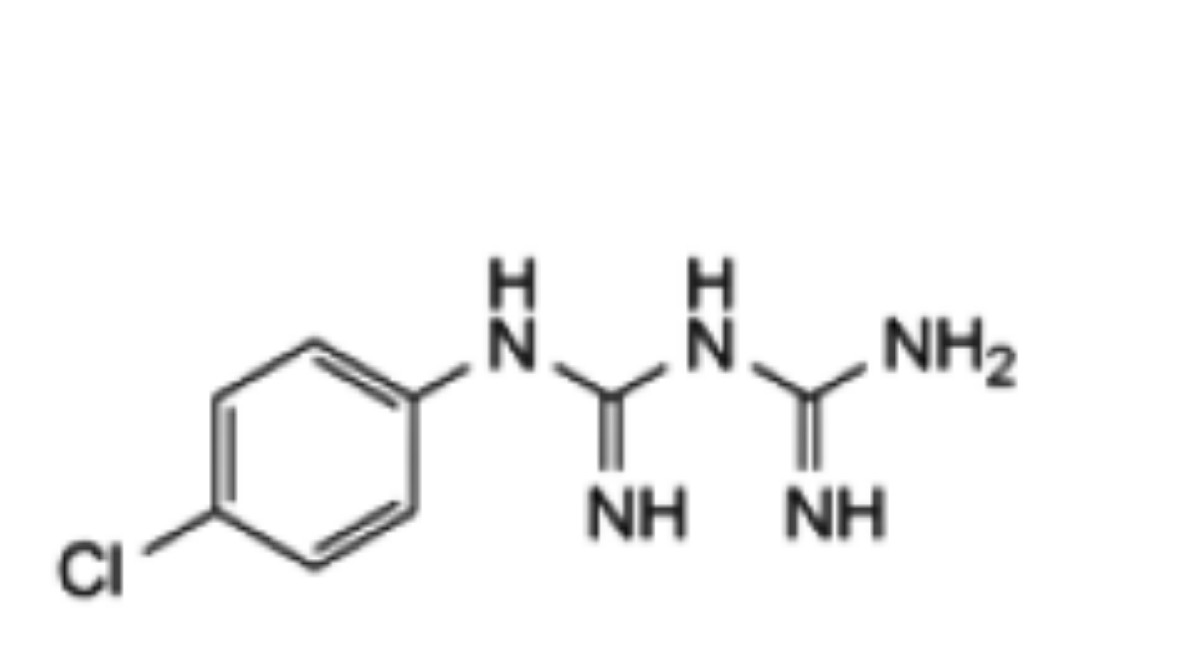
2. 4-chlorophenyl biguanide.






