(Ph. Eur. monograph 2010)
Al2Mg6C2O20H14,4H2O 630 66827-12-1
Action and use
Antacid.
DEFINITION
Hydrated aluminium magnesium hydroxycarbonate.
Content
— aluminium: 15.0 per cent to 17.0 per cent (calculated as Al2O3),
— magnesium: 36.0 per cent to 40.0 per cent (calculated as MgO),
— carbonic acid: 12.5 per cent to 14.5 per cent (calculated as CO2).
CHARACTERS
Appearance
White or almost white, fine, crystalline powder.
Solubility
Practically insoluble in water, in ethanol (96 per cent) and in methylene chloride. It dissolves with effervescence and heating in dilute mineral acids.
IDENTIFICATION
A. Infrared absorption spectrophotometry (2.2.24).
Comparison Ph. Eur. reference spectrum of almagate.
B. Dissolve 0.15 g in dilute hydrochloric acid R and dilute to 20 mL with the same acid. 2 mL of the solution gives the reaction of aluminium (2.3.1).
C. 2 mL of the solution prepared under identification test B gives the reaction of magnesium (2.3.1).
TESTS
pH (2.2.3)
9.1 to 9.7.
Disperse 4.0 g in 100 mL of carbon dioxide-free water R, stir for 2 min and filter.
Neutralising capacity
Carry out the test at 37 °C Disperse 0.5 g in 100 mL of water R, heat, add 100.0 mL of 0.1 M hydrochloric acid, previously heated and stir continuously; the pH (2.2.3) of the solution between 5 min and 20 min is not less than 3.0 and not greater than 4.5. Add 10.0 mL of 0.5 M hydrochloric acid, previously heated, stir continuously for 1 h and titrate with 0.1 M sodium hydroxide to pH 3.5; not more than 20.0 mL of 0.1 M sodium hydroxide is required.
Chlorides (2.4.4)
Maximum 0.1 per cent.
Dissolve 0.33 g in 5 mL of dilute nitric acid R and dilute to 100 mL with water R. Prepare simultaneously the standard by diluting 0.7 mL of dilute nitric acid R to 5 mL with water R and adding 10 mL of chloride standard solution (5 ppm Cl) R.
Sulfates (2.4.13)
Maximum 0.4 per cent.
Dissolve 0.25 g in 5 mL of dilute hydrochloric acid R and dilute to 100 mL with distilled water R. Prepare simultaneously the standard by adding 0.8 mL of dilute hydrochloric acid R to 15 mL of sulfate standard solution (10 ppm SO4) R.
Sodium
Maximum 150 ppm.
Atomic absorption spectrometry (2.2.23, Method I).
Test solution Dissolve 0.25 g in 50 mL of a 103 g/L solution of hydrochloric acid R.
Reference solutions Prepare the reference solutions using sodium standard solution (200 ppm Na) R, diluted as necessary with a 103 g/L solution of hydrochloric acid R.
Loss on ignition
43.0 per cent to 49.0 per cent, determined on 1.000 g by ignition at 900 ± 50 °C.
Microbial contamination
TAMC: acceptance criterion 10 CFU/g (2.6.12).
TYMC: acceptance criterion 10 CFU/g (2.6.12).
Absence of Escherichia coli (2.6.13).
Absence of Pseudomonas aeruginosa (2.6.13).
ASSAY
Aluminium
Dissolve 1.000 g in 5 mL of hydrochloric acid R, heating if necessary. Allow to cool to room temperature and dilute to 100.0 mL with water R (solution A). Introduce 10.0 mL of solution A into a 250 mL conical flask, add 25.0 mL of 0.05 M sodium edetate, 20 mL of buffer solution pH 3.5 R, 40 mL of anhydrous ethanol R and 2 mL of a freshly prepared 0.25 g/L
solution of dithizone R in anhydrous ethanol R. Titrate the excess of sodium edetate with 0.05 M zinc sulfate until the colour changes from greenish-violet to pink.
1 mL of 0.05 M sodium edetate is equivalent to 2.549 mg of Al2O3
Magnesium
Introduce 10.0 mL of solution A prepared in the assay of aluminium into a 500 mL conical flask, add 200 mL of water R, 20 mL of triethanolamine R with shaking, 10 mL of ammonium chloride buffer solution pH 10.0 R and 50 mg of mordant black 11 triturate R. Titrate with 0.05 M sodium edetate until the colour changes from violet to pure blue.
1 mL of 0.05 M sodium edetate is equivalent to 2.015 mg of MgO.
Carbonic acid
12.5 per cent to 14.5 per cent.
Test sample Place 7.00 mg of the substance to be examined in a tin capsule. Seal the capsule.
Reference sample Place 7.00 mg of almagate CRS in a tin capsule. Seal the capsule.
Introduce separately the test sample and the reference sample into a combustion chamber of a CHN analyser purged with helium for chromatography R and maintained at a temperature of 1020 °C. Simultaneously, introduce oxygen R at a pressure of 40 kPa and a flow rate of 20 mL/min and allow complete combustion of the sample. Sweep the combustion gases through a reduction reactor and separate the gases formed by gas chromatography (2.2.28).
Column:
— size: l = 2 m, Ø = 4 mm;
— stationary phase: ethylvinylbenzene-divinylbenzene copolymer R.
Carrier gas helium for chromatography R.
Flow rate 100 mL/min.
Temperature:
— column: 65 °C;
— detector: 190 °C.
Detection Thermal conductivity.
Run time 16 min.
System suitability:
— average percentage of carbon in 5 reference samples must be within ± 0.2 per cent of the value assigned to the CRS; the difference between the upper and the lower values of the percentage of carbon in these samples must be below 0.2 per cent.
Calculate the percentage content of carbonic acid in the test sample according to the following formula:
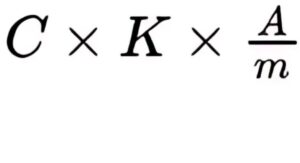
C = percentage content of carbonic acid in the reference sample;
K = mean value for the 5 reference samples of the ratio of the mass in milligrams to the area of the peak due to carbonic acid;
A = area of the peak due to carbonic acid in the chromatogram obtained with the test sample;
m = sample mass, in milligrams.
STORAGE
In an airtight container.






